Search results
Search for "acid" in Full Text gives 2759 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
N-Sulfenylsuccinimide/phthalimide: an alternative sulfenylating reagent in organic transformations
Beilstein J. Org. Chem. 2023, 19, 1471–1502, doi:10.3762/bjoc.19.106

- -catalyzed sulfenylation by N-(sulfenyl)succinimides/phthalimides In 2012, Chen and co-workers found that in the reaction of N-(organothio)succinimides 1 and sodium sulfinates 2 using a Lewis acid in ionic liquids (ILs) and water as a green solvent system leads to the formation of thiosulfonates 3 (Scheme 2
- ) [44]. Among different Lewis acid catalysts, such as Cu(OTf)2, Mg(OTf)2, Zn(OTf)2, Sc(OTf)3, Eu(OTf)3, and Yb(OTf)3, it was found that Sc(OTf)3 gave higher product yield. In addition, the combination of Sc(OTf)3/ILs displayed good recyclability in this transformation. In 2014, Anbarasan and Saravanan
- nucleophilic attack of TMSN3 to deliver product 11 (Scheme 7). Tian and Chang et al. could synthesize 3‑sulfenylated coumarin compounds 13 by using N-sulfanylsuccinimides 1 under a Lewis acid catalysis system (Scheme 8) [48]. Additionally, oxidation of 3-sulfenylated coumarins utilizing (diacetoxyiodo)benzene
Functions of enzyme domains in 2-methylisoborneol biosynthesis and enzymatic synthesis of non-natural analogs
Beilstein J. Org. Chem. 2023, 19, 1452–1459, doi:10.3762/bjoc.19.104
- diphosphate synthase (FPPS) and 2MIBS from Streptomyces coelicolor [26] (Scheme 1B). Crystal structures of both enzymes have been obtained [27][28] and allowed for a deep structure-based investigation of 2MIBS through site-directed mutagenesis [29]. The predicted amino acid sequences of 2MIBS homologs from
- alignment with the short 2MIBS from Longispora albida DSM 44784 (WP_018349754) the border between domains A and B in the 2MIBS from S. coelicolor was identified. Domain A spans the amino acid residues 1–115 and domain B includes the amino acid residues 115–440. The gene sequences for both domains were
α-(Aminomethyl)acrylates as acceptors in radical–polar crossover 1,4-additions of dialkylzincs: insights into enolate formation and trapping
Beilstein J. Org. Chem. 2023, 19, 1443–1451, doi:10.3762/bjoc.19.103
- . Trialkylboranes can react in a similar way with enones [3] whereas, distinctively, suitable acceptors for the reaction with dialkylzinc reagents also include α,β-unsaturated carboxylic acid derivatives such as α,β-unsaturated (di)esters [4][5], N-enoyloxazolidinones [6][7], N-enoyloxazolidines [8], or
- . Trapping of this enolate would lead to β-amino acid units, a class of compounds that has attracted a great deal of attention [19][20][21][22][23][24]. An obvious possible shortcoming that had to be considered was still that the generated zinc enolate III having a β-amino group could undergo β-elimination
- . Importantly, the protocol was found to be similarly applicable with enoates 8b (Table 3, entry 6) and 8c (entry 7) having tert-butyl and benzyl ester groups, which, as the methyl ester unit, are typical in the context of amino acid synthesis. ZnBu2 was also amenable to 1,4-addition (Table 3, entry 8), but not
Application of N-heterocyclic carbene–Cu(I) complexes as catalysts in organic synthesis: a review
Beilstein J. Org. Chem. 2023, 19, 1408–1442, doi:10.3762/bjoc.19.102
- heteroleptic Cu(I) complexes combining the malonic acid-derived anionic NHC and a neutral imidazol-2-ylidene were also obtained in a very selective manner (Scheme 12). As discussed later, many of these complexes were employed as catalysts. In 2015, Collins et al. [27] compared the stability and reactivity of
- explored the application of a thiazolylidene-based Cu(I) complex as catalyst for [3 + 2] cycloaddition reactions. They prepared an ethylene-linked bisthiazol-2-ylidene dicopper(I) complex 136 which showed high catalytic activity (Scheme 53). The activity increased upon addition of acetic acid, particularly
Functional characterisation of twelve terpene synthases from actinobacteria
Beilstein J. Org. Chem. 2023, 19, 1386–1398, doi:10.3762/bjoc.19.100
- synthase homologs from diverse actinobacteria that were selected based on a phylogenetic analysis of more than 4000 amino acid sequences were investigated for their products. For four enzymes with functions not previously reported from bacterial terpene synthases the products were isolated and their
- , Supporting Information File 1). The closest characterised terpene synthase with an amino acid sequence identity of 25% is the (1(10)E,4E,6S,7R)-germacradien-6-ol synthase from Streptomyces pratensis [33]. The recombinant enzyme efficiently converted FPP into one sesquiterpene alcohol whose electron
- . brevicatena (Table 1, entry 5) showed the highly conserved motifs with a modified aspartate-rich region (86DDHRN) and the NSE triad 227NDLHSMPKE (Figure S33, Supporting Information File 1). This enzyme is closely related to the epi-isozizaene synthase from S. coelicolor (EIZS) [24], but is with an amino acid
Consecutive four-component synthesis of trisubstituted 3-iodoindoles by an alkynylation–cyclization–iodination–alkylation sequence
Beilstein J. Org. Chem. 2023, 19, 1379–1385, doi:10.3762/bjoc.19.99
- , 63.81; H, 3.74; N, 2.96. Synthesis of 1,2,3-trisubstituted indole 8b (typical procedure) 3-Iodoindole 5a (167 mg, 0.50 mmol), (p-tolyl)boronic acid (7b, 204 mg, 1.50 mmol), Pd(PPh3)4 (28.9 mg, 25.0 μmol), and cesium carbonate (652 mg, 2.00 mmol) were placed in an oven-dried Schlenk tube with magnetic
Visible-light-induced nickel-catalyzed α-hydroxytrifluoroethylation of alkyl carboxylic acids: Access to trifluoromethyl alkyl acyloins
Beilstein J. Org. Chem. 2023, 19, 1372–1378, doi:10.3762/bjoc.19.98
- photocatalysts, providing a direct and robust access to trifluoromethyl aliphatic acyloins. Results and Discussion Initially, we commenced our exploration by choosing 4-phenylbutyric acid (1a) as the model substrate to react with N-trifluoroethoxyphthalimide (2, Table 1). On basis of the previously reported
- carboxylic acid and pivalic anhydride than aryl bromide. The halides provided versatile synthetic handles for further transformations. Substrates bearing thiophene (3k) furan (3j) and other heterocycle (3l,m) moieties were also applicable to this reaction. This protocol allowed for the coupling of not only
- slightly lower yield. To further demonstrate the amenability toward pharmaceutically active molecules, chloroambucil (1t) and dehydrocholic acid (1u) were successfully subjected to the reaction conditions, delivering the desired products in moderate yields. However, the analogous reaction with N
Synthesis of ether lipids: natural compounds and analogues
Beilstein J. Org. Chem. 2023, 19, 1299–1369, doi:10.3762/bjoc.19.96
- hypotensive effects. First, Wissner et al. reported the synthesis of racemic sn-1-deoxy-PAF 11.8 (Figure 11) [91]. First, n-octadecanoic acid chloride (11.1) reacted with tris[(trimethylsilyl)oxy]ethylene (11.2) [92] to produce, after acidic hydrolysis and subsequent decarboxylation, compound 11.3. Then, the
- whereas heating it in 75% acetic acid solution produced the deprotected compound but migration of the acyl group from the sn-2 to the sn-3 position lead to an inseparable mixture of regioisomers. A selective desilylation of 12.5 was finally achieved with BF3·Et2O producing 12.6 without migration of the
- with mCPBA produced the epoxide 13.3. Then, the addition of benzoic acid in the presence of acid catalysis produced an ester that was saponified to yield the diol 13.4. A three-step sequence is applied to produce compound 13.5 that features a secondary alcohol protected with a benzyl group. Then, the
Non-noble metal-catalyzed cross-dehydrogenation coupling (CDC) involving ether α-C(sp3)–H to construct C–C bonds
Beilstein J. Org. Chem. 2023, 19, 1259–1288, doi:10.3762/bjoc.19.94
- achieve the CDC of THF and phenol C(sp2)–H (Scheme 12) [62]. The role of Pd may be through the formation of a Pd(II) phenolic acid salt from phenol and Pd(OAc)2 to improve the reactivity of phenol. Subsequently, a more complex C(sp2)–H component was employed as a coupling substrate to functionalize the
- of ethers to obtain symmetric and asymmetric 1,1-bis-indolylmethane derivatives (Scheme 23) [84]. The reaction proceeds through the tandem oxidative coupling of the C–O bond and cleavage of the C–H bond. Fe plays a dual role in catalysing the C–C bond coupling and C–O bond cleavage as Lewis acid
- ]. This pathway involves the presence of methanesulfonic acid and a large amount of metal oxide to obtain the target product with moderate to good yield. In 2015, a MnO2-catalyzed sequential oxidative alkylation of C(sp3)–H/C(sp2)–H CDC and hydrolysis of enamides with ethers was reported by Xu et al
Metal catalyst-free N-allylation/alkylation of imidazole and benzimidazole with Morita–Baylis–Hillman (MBH) alcohols and acetates
Beilstein J. Org. Chem. 2023, 19, 1251–1258, doi:10.3762/bjoc.19.93
- , Yang et al. [18][19] have developed a catalytic system involving Pd/Ti(OiPr)4 or Pd/carboxylic acid for the direct allylation of anilines with alcohols. The synthesis of N-allylimidazole derivatives 3 has been previously carried out using acyclic MBH adducts bearing good leaving groups, such as bromide
Acetaldehyde in the Enders triple cascade reaction via acetaldehyde dimethyl acetal
Beilstein J. Org. Chem. 2023, 19, 1243–1250, doi:10.3762/bjoc.19.92
- acetaldehyde have been reported [20][21][22][23][24]. The safety and handling problems associated with acetaldehyde can be solved by synthetic equivalents that can be generated in situ through different paths. Some examples are represented by vinyl acetate [25], silyl vinyl ethers [26], ethanol, pyruvic acid
- , (E)-3-chloroacrylic acid, 2,4,6-trimethyl-1,3,5-trioxane (paraldehyde) [24][27], and acetaldehyde dimethyl acetal (6) [17][18][19]. On the basis of a long-term project based on masked reagents, our group has previously demonstrated the feasibility of the addition of a masked acetaldehyde 6 to
- acetaldehyde, which is hydrolyzed in situ using Amberlyst-15 as an acid catalyst, instead of directly using acetaldehyde allows for higher yields and fewer byproducts. Using mild reaction conditions, it was possible to obtain a variety of functionalized cyclohexene carbaldehydes in good yields and very high
Radical ligand transfer: a general strategy for radical functionalization
Beilstein J. Org. Chem. 2023, 19, 1225–1233, doi:10.3762/bjoc.19.90
- the mechanism. In 2015, the Groves group reported their manganese porphin-based catalyst V and related species being capable of participating in decarboxylation reactions (Scheme 4) [42]. The activated Mn(V) species is proposed to perform HAT carboxylic acid O–H bond, directly forming a carboxyl
- ) azide complex, reducing the iron catalyst back to the starting Fe(II) state. Organic azides can be formed in moderate to high enantioselectivity using this approach; however, the scope is largely limited to benzylic products, a result in line with Groves’ finding that benzylic acid substrates perform
- beautiful decarboxylative azidation examples, combining iron-mediated photodecarboxylation via LMCT and azide RLT (Scheme 5) [11]. Irradiating a substoichiometric amount of iron(III) nitrate hydrate III in the presence of carboxylic acid, TMS azide, and sodium carbonate allows for direct synthesis of alkyl
Unravelling a trichloroacetic acid-catalyzed cascade access to benzo[f]chromeno[2,3-h]quinoxalinoporphyrins
Beilstein J. Org. Chem. 2023, 19, 1216–1224, doi:10.3762/bjoc.19.89

- ]quinoxalinoporphyrins in good yields via a sequential reaction of copper(II) 2,3-diamino-5,10,15,20-tetraarylporphyrins, 2-hydroxynaphthalene-1,4-dione, aromatic aldehydes, and dimedone in the presence of a catalytic amount of trichloroacetic acid in chloroform at 65 °C. Further, the newly prepared copper(II
- ; multicomponent synthesis; one-pot reaction; trichloroacetic acid; Introduction π-Conjugated porphyrin macrocycles are known for their applications in numerous areas ranging from oxygen transport, photosynthesis, catalysis and medicine [1][2][3]. In the past several years, diverse organic scaffolds have been
- present study discloses an easy and first synthetic approach to build highly π-conjugated copper(II) benzo[f]chromeno[2,3-h]quinoxalinoporphyrins through a trichloroacetic acid-catalyzed one-pot four-component reaction of 2,3-diamino-5,10,15,20-tetraarylporphyrins, 2-hydroxynaphthalene-1,4-dione, aromatic
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
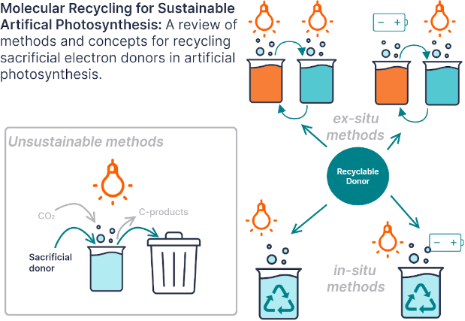
- reduction consumed the sacrificial donor methanol to form formic acid and formaldehyde [56]. This system is interesting for a number of reasons. Rather than intermediate redox mediators shuttling charge between two photocatalytic assemblies, Ishitani, Domen, and co-workers covalently connected the catalytic
- photocatalytic or electrochemical carbon dioxide reduction. The oxidation product, formaldehyde, can be re-reduced. However, separation of formaldehyde and the carbon dioxide reduction product formic acid would be difficult. Therefore, a logical route for sustainably sourcing methanol would be using this system
Cyanothioacetamides as a synthetic platform for the synthesis of aminopyrazole derivatives
Beilstein J. Org. Chem. 2023, 19, 1191–1197, doi:10.3762/bjoc.19.87
- upon addition of hydrochloric acid. This is probably due to the protonation of the dimethylamino moiety or/and that dimethylamine hydrochloride is a better leaving group than the free base. The structures of compounds 6a–f were confirmed by 1H and 13C NMR spectroscopy and HRMS, as well as X-ray
- significantly higher yield (72–94%, Scheme 4). We have noticed that 1-sulfonylpyrazole 7b is unstable when heated in ethanol in the presence of hydrochloric acid and converts into 3-aminopyrazole-4-carbothioamide 5b. It is worth noting that the two-stage method of obtaining 3-aminopyrazole-4-carbothioamide 5b
Exploring the role of halogen bonding in iodonium ylides: insights into unexpected reactivity and reaction control
Beilstein J. Org. Chem. 2023, 19, 1171–1190, doi:10.3762/bjoc.19.86

- potentials of 0.075 e and 0.048 e. The cyclic dimedone-derived ylide I-12 showed similar potentials of 0.080 e and 0.052 e, as did the Meldrum’s acid-derived ylide I-13 (0.085 e and 0.057 e). Whether the ylide was cyclic or acyclic, the σ-hole opposite the β-dicarbonyl motif was consistently greater in
- decomposition [1][102]. They discovered that an iodonium ylide (e.g., 6/I-12) reacts with hydrochloric acid to produce a chlorinated β-dicarbonyl 7, opposite their expectation that the C–I bond with the lower electron density would be cleaved. While both of these reaction pathways are viable, the iodonium ylide
- iodobenzene then generated a strained, cyclopropane-fused bicycle 49 that rearranged into the isolated C–H insertion product 46a. In addition to Neiland’s disclosure of the reactions between iodonium ylides and hydrochloric acid (Scheme 1) [1][102], others have also reported that both strong acids (e.g., TsOH
Two new lanostanoid glycosides isolated from a Kenyan polypore Fomitopsis carnea
Beilstein J. Org. Chem. 2023, 19, 1161–1169, doi:10.3762/bjoc.19.84
- Chemical exploration of solid-state cultures of the polypore Fomitopsis carnea afforded two new C31 lanostane-type triterpenoid glycosides, forpiniosides B (1) and C (2) together with two known derivatives, namely 3-epipachymic acid (3) and (3α,25S)-3-O-malonyl-23-oxolanost-8,24(31)-dien-26-oic acid (4
- elucidation of two new C31 lanostane-type triterpenoid glycosides (compounds 1 and 2 in Figure 1) together with two known derivatives, namely 3-epipachymic acid (3α-acetoxy-16α-hydroxy-5α-lanost-8,24(31)-dien-21-oic acid (3)) [24] and (3α,25S)-3-O-malonyl-23-oxolanost-8,24(31)-dien-26-oic acid (4) [25
- lanostan-8,24(31)-diene-21-oic acid skeleton supported by 2D NMR cross peaks in the 1H,1H COSY, HMBC, and HSQC spectra, suggesting a closely related structure to forpinioside A [23][28]. The C-5, C-10, C-13, and C-14 configurations were assigned not only from the biogenetic considerations, but also from
New one-pot synthesis of 4-arylpyrazolo[3,4-b]pyridin-6-ones based on 5-aminopyrazoles and azlactones
Beilstein J. Org. Chem. 2023, 19, 1155–1160, doi:10.3762/bjoc.19.83
- few (Scheme 1). To obtain 4-arylpyrazolo[3,4-b]pyridin-6-ones, the only known one-step method is most often used, including the acid-catalyzed condensation of aminopyrazoles with ketoesters [1][16][18] (method A). Its significant disadvantage is the low yields of the target products (11–60%). Yields
- are also low in two-stage synthesis methods. The first of them is based on the three-component condensation of aminopyrazoles, Meldrum's acid, and aromatic aldehydes, followed by the oxidation of the intermediate with DDQ [13][16][19] (method B). The second one includes the reaction of an aromatic
- aldehyde with thioglycolic acid and aminopyrazole, followed by the extrusion of sulfur from the resulting thiazepine [20] (method C). The three-stage synthesis of 4-arylpyrazolo[3,4-b]pyridin-6-ones, involving the preparation of 3-aryl-N-(1H-pyrazol-5-yl)propiolamides (method D), also leads to the
Selective and scalable oxygenation of heteroatoms using the elements of nature: air, water, and light
Beilstein J. Org. Chem. 2023, 19, 1146–1154, doi:10.3762/bjoc.19.82
- maximal industrial relevance, the focus was placed on additives that are non-toxic, easy to separate, inexpensive, and readily available. Three classes of compounds have been exploited: acid and bases, salts, and aromatic additives. Acid and bases As can be seen from Figure 1A, in the presence of strong
- acids, an increase in reaction rate was observed. For example, when adding 1 equiv trifluoroacetic acid, a full conversion was achieved in 40 minutes instead of 60 minutes. Contrarily, the addition of a strong base substantially slowed down the reaction rate, e.g., the addition of 1 equiv NaOH resulted
Photoredox catalysis harvesting multiple photon or electrochemical energies
Beilstein J. Org. Chem. 2023, 19, 1055–1145, doi:10.3762/bjoc.19.81
- products, demonstrating a clear preference for addition to alkenes even in the presence of alkyne functionalities. Due to basic conditions of the reductive quenching (RQ) route, the formation of lactone side product 30h could be observed with a carboxylic acid functionality. In the absence of Et3N, the
Synthesis of imidazo[4,5-e][1,3]thiazino[2,3-c][1,2,4]triazines via a base-induced rearrangement of functionalized imidazo[4,5-e]thiazolo[2,3-c][1,2,4]triazines
Beilstein J. Org. Chem. 2023, 19, 1047–1054, doi:10.3762/bjoc.19.80
- [2,3-c][1,2,4]triazines was synthesized via a cascade sequence of hydrolysis and skeletal rearrangement of imidazo[4,5-e]thiazolo[2,3-c][1,2,4]triazin-7(8H)-ylidene)acetic acid esters in methanol upon treatment with excess KOH. Imidazo[4,5-e]thiazolo[3,2-b][1,2,4]triazin-6(7H)-ylidene)acetic acid
- -aminocephalosporanic acid (7-ACA), which is a key fragment of broad-spectrum cephalosporin antibiotics [13][14]. Condensed 1,2,4-triazines attract attention of researchers due to their diverse biological activities [15] and also their application as starting materials for the constructing of new heterocyclic systems
- prepare the corresponding carboxylic acids 4 using an aqueous KOH solution. Heating esters 1a,b in an aqueous solution of KOH and subsequent addition of hydrochloric acid led to the corresponding acids 4a,b as the main products. Acids 4a,b were isolated from the mixtures in 17 and 38% yield, respectively
The effect of dark states on the intersystem crossing and thermally activated delayed fluorescence of naphthalimide-phenothiazine dyads
Beilstein J. Org. Chem. 2023, 19, 1028–1046, doi:10.3762/bjoc.19.79

- synthesized by a modified literature method [43]. Compound NI-PTZ-F (36 mg, 0.074 mmol) was dissolved in glacial acetic acid (5 mL), H2O2 (1.5 mL, 30%, 1.184 mmol) was added dropwise and the mixture was stirred at 40 °C for 1 h. Then, the reaction mixture was poured into water and the pH of the mixture was
CO2 complexation with cyclodextrins
Beilstein J. Org. Chem. 2023, 19, 1021–1027, doi:10.3762/bjoc.19.78
- using a pressure cell and a UV–vis competition assay with an azo-dye (4-((4-hydroxyphenyl)azo)-1-naphthalenesulfonic acid (7) Figure 4) [15] as an indicator. The UV–vis spectrum of 7 changes on binding to cyclodextrins and we can thereby indirectly monitor the binding of CO2 to the CD by observing the
- change in spectrum of 7 provided 7 and CO2 compete for the binding site. To avoid problems with formation of hydrogencarbonate the experiments were conducted in a buffer at pH 3 where only a minor fraction of the carbonic acid (pKa1 = 3.6) is dissociated and since the hydration constant of CO2 is small
Copper-catalyzed N-arylation of amines with aryliodonium ylides in water
Beilstein J. Org. Chem. 2023, 19, 1008–1014, doi:10.3762/bjoc.19.76
- pharmaceutical agents containing the arylamine moiety as an integral part of the structural framework includes molecule such as retigabine (I) [5], an anticonvulsant used as an adjunctive agent in the treatment of partial seizures, folic acid (II) [6], a type of vitamin B12 displaying an important role in
- metabolism, cell growth and during pregnancy, ofloxacin (III) [7], an antibacterial agent, mefenamic acid (IV) [8], an anti-inflammatory agent used to treat mild pain, linezolid (V) [9], an antibacterial agent, repaglinide (VI) [10], used to treat diabetes mellitus type 2, and tolfenamic acid (VII) [11], an
Synthesis of tetrahydrofuro[3,2-c]pyridines via Pictet–Spengler reaction
Beilstein J. Org. Chem. 2023, 19, 991–997, doi:10.3762/bjoc.19.74
- the condensation of easily accessibly 2-(5-methylfuran-2-yl)ethanamine with commercially available aromatic aldehydes followed by acid-catalyzed Pictet–Spengler cyclization. Using this approach, we synthesized a range of 4-substituted tetrahydrofuro[3,2-c]pyridines in reasonable yields. The reactivity
- of some of the products was investigated and selected synthetic transformations of the obtained tetrahydrofuro[3,2-c]pyridines were shown. Keywords: acid hydrolysis; 1,4-diketone; tetrahydrofuro[3,2-c]pyridines; Paal–Knorr reaction; Pictet–Spengler reaction; Introduction Hydrogenated furo[3,2-c
- acetic and hydrochloric acid previously well-proven for furan chemistry [44][45][46][47][48]. Under these conditions at room temperature for 1 hour, the product yield was 18% (Table 1, entry 11). An increase of the reaction time up to 48 h leads to a significant increase of the yield (Table 1, entries 12





































































































































































































































































































































































































































































































































































