Search results
Search for "one-pot reaction" in Full Text gives 183 result(s) in Beilstein Journal of Organic Chemistry.
Tying a knot between crown ethers and porphyrins
Beilstein J. Org. Chem. 2023, 19, 1630–1650, doi:10.3762/bjoc.19.120

- notable distinction as they could readily undergo oxidation to yield the corresponding crownphyrins 28–33, incorporating a dipyrrin unit. The synthetic pathway towards crownphyrins is straightforward and relies on a one-pot reaction between diformyldipyrromethane 20a/b and a diamine which introduces the
One-pot nucleophilic substitution–double click reactions of biazides leading to functionalized bis(1,2,3-triazole) derivatives
Beilstein J. Org. Chem. 2023, 19, 1399–1407, doi:10.3762/bjoc.19.101
- substituent undergo a one-pot reaction with reasonable efficiency and furnished the expected 1,2,3-triazole derivative 7 in 61% yield under the approved conditions. Next, we turned our attention to the generation of divalent systems, starting with the reactions of 1,3- (8) and 1,2-bis(bromomethyl)benzene (11
- . One-pot reaction employing enantiopure alkynyl-substituted 1,2-oxazin-4-one derivative 6 leading to 1,2,3-triazole 7. One-pot reactions of dihalides 8 and 11 with sodium azide and alkyne 2 leading to symmetric divalent bis(1,2,3-triazoles) 9 and 12 as major products. One-pot reactions employing
- enantiopure alkynyl-substituted 1,2-oxazin-4-one derivative 6 leading to bis(1,2,3-triazoles) 14 and 17 as major products. One-pot reaction employing enantiopure alkynyl-substituted 1,2-oxazin-4-ol derivative 19 leading to bis(1,2,3-triazoles) 20 and 21. Reductive ring-openings of 1,2-oxazine derivatives 19
Unravelling a trichloroacetic acid-catalyzed cascade access to benzo[f]chromeno[2,3-h]quinoxalinoporphyrins
Beilstein J. Org. Chem. 2023, 19, 1216–1224, doi:10.3762/bjoc.19.89

- ; multicomponent synthesis; one-pot reaction; trichloroacetic acid; Introduction π-Conjugated porphyrin macrocycles are known for their applications in numerous areas ranging from oxygen transport, photosynthesis, catalysis and medicine [1][2][3]. In the past several years, diverse organic scaffolds have been
Photoredox catalysis harvesting multiple photon or electrochemical energies
Beilstein J. Org. Chem. 2023, 19, 1055–1145, doi:10.3762/bjoc.19.81
- products (e.g., 7) whereas irradiation with blue light (λ = 455 nm) provided disubstituted products 8 (Figure 5A). Additionally, adding a different trapping reagent before switching from green to blue light allows for a sequential and controlled substitution in a one-pot reaction (Figure 5B). 2,4,6
An efficient metal-free and catalyst-free C–S/C–O bond-formation strategy: synthesis of pyrazole-conjugated thioamides and amides
Beilstein J. Org. Chem. 2023, 19, 231–244, doi:10.3762/bjoc.19.22
- for the construction of biologically interesting highly diversified pyrazole-linked thioamide and amide conjugates has been developed. The pyrazole C-3/4/5-tethered thioamide conjugates were prepared via a one-pot reaction between highly diversified pyrazole carbaldehydes, cyclic secondary amines, and
Sequential hydrozirconation/Pd-catalyzed cross coupling of acyl chlorides towards conjugated (2E,4E)-dienones
Beilstein J. Org. Chem. 2023, 19, 176–185, doi:10.3762/bjoc.19.17
- substituents in the one-pot reaction and showed that regardless of the substitution pattern, the reactions lead to the stereoselective formation (≥95% (2E,4E)) of the respective dienones under mild conditions. It was found that enynes with alkyl chains gave higher yields than the corresponding aryl-substituted
A novel bis-triazole scaffold accessed via two tandem [3 + 2] cycloaddition events including an uncatalyzed, room temperature azide–alkyne click reaction
Beilstein J. Org. Chem. 2022, 18, 1636–1641, doi:10.3762/bjoc.18.175
- intermediate before the click reaction was established by 1H NMR analysis of the reaction mixture). However, the product of this two-step, one-pot reaction (19) was isolated in respectable 61% yield. The structure of compound 19 was confirmed by the single-crystal X-ray analysis which demonstrated that the
One-pot double annulations to confer diastereoselective spirooxindolepyrrolothiazoles
Beilstein J. Org. Chem. 2022, 18, 1607–1616, doi:10.3762/bjoc.18.171
- (Table 1, entries 2–4) superior to 86% yield for 3 h (Table 1 entry 1), and followed by decarboxylative [3 + 2] cycloaddition with the second equivalent of compound 1a and olefinic oxindole 4a under reflux heating for 12 h. It indicates that the one-pot reaction process with EtOH and iPrOH afforded the
- spirooxindolepyrrolothiazole analogues 5a–d with 49–70% isolated yield (Scheme 3) under the optimized reaction conditions (Table 1, entry 7). Compounds 5b–d with using heteroaromatic aldehydes resulted in lower yield than 5a. In addition, according to the one-pot reaction process (Table 1, entry 5) with two operational steps
- (0.5 M), 90 °C, 9 h (Table 1, entry 6). iii) 2.2:1.1:1.0 of 1a/2/4a, EtOH (0.5 M), 90 °C, 9 h (Table 1, entry 7). Optimization of reaction conditions for double annulations of cysteine.a One-pot reaction for the synthesis of compound 7. Green metrics (AE, AEf, CE, RME, OE and MP) analysis for processes
Synthesis of novel alkynyl imidazopyridinyl selenides: copper-catalyzed tandem selenation of selenium with 2-arylimidazo[1,2-a]pyridines and terminal alkynes
Beilstein J. Org. Chem. 2022, 18, 863–871, doi:10.3762/bjoc.18.87
- activity of the compounds obtained via this synthesis route. Conclusion In this study, the synthesis route for novel alkynyl imidazopyridinyl selenides using the Cu-catalyzed one-pot reaction of Se powder with imidazo[1,2-a]pyridines and terminal alkynes was developed. A variety of desired compounds was
- reactions. Transformation from 4aa. One-pot reaction of Se powder with 1a and 3aa. One-pot two-step synthesis of alkynyl imidazopyridinyl selenides 4a. Supporting Information Supporting Information File 76: Characterization data of all new compounds, synthetic procedures for compounds 6–8, X-ray
- -pot reaction, both selanyl groups from the diselenide transferred to the product 4aa. When no base was added or the amount of the base was reduced, the yield of 4aa decreased significantly (Table 1, entries 9 and 10). Moreover, increasing the amount of the alkyne 1a or Na2CO3 did not affect the
Copper-catalyzed multicomponent reactions for the efficient synthesis of diverse spirotetrahydrocarbazoles
Beilstein J. Org. Chem. 2022, 18, 796–808, doi:10.3762/bjoc.18.80
- underwent a [4 + 2] cycloaddition reaction (reaction 1 in Scheme 1) [69][70][71][72][73][74]. This metal-catalyzed one-pot reaction not only combined the advantages of a traditional Diels–Alder reaction and the recently developed multicomponent reactions, but also meets the goal of green and sustainable
Heteroleptic metallosupramolecular aggregates/complexation for supramolecular catalysis
Beilstein J. Org. Chem. 2022, 18, 597–630, doi:10.3762/bjoc.18.62

- advantages of utilizing self-assembly to construct discrete nanocages with predetermined geometry and function is the use of a one-pot reaction employing complementary organic linkers with inorganic metal ions. Although the one-pot synthesis of homoleptic metallacages has been thoroughly investigated over
- -bonding, which would lead to catalytic quenching, a unique design strategy was applied. Instead of using a cis-blocked palladium(II) unit for self-assembly, Pd(NO3)2 was employed along with the triazole-based 0° clip 13. In a one-pot reaction, 12 and 13 in DMSO were treated with Pd(NO3)2 in a 1:1:1 ratio
- reside inside the sphere (Figure 7). In order to vary the local gold concentration inside the cavity, heteroleptic cages were assembled from a multicomponent one-pot reaction of Pd(II) with 30 and the analogous non-functionalized ligand 31. By controlling the ratio of 30 and 31, spheres with varying
Syntheses of novel pyridine-based low-molecular-weight luminogens possessing aggregation-induced emission enhancement (AIEE) properties
Beilstein J. Org. Chem. 2022, 18, 580–587, doi:10.3762/bjoc.18.60

- . pyrido[1,2-a]pyrrolo[3,4-d]pyrimidines 3a,b and N-methyl-4-((pyridin-2-yl)amino)maleimides 4a–e, were selectively prepared by a one-pot reaction between a functionalized maleimide and 2-aminopyridines with electron-donating or electron-withdrawing groups at position 5 and were investigated
- the electronic nature of the compounds with and without AIEE properties. Keywords: acceptor–donor–acceptor; AIEE; low molecular weight; one-pot reaction; ((pyridin-2-yl)amino)maleimide; TD-DFT calculation; Introduction Fluorescent compounds have attracted considerable attention as functional
- study, we used 1-methyl-4-(methylsulfanyl)-2,5-dioxo-2,5-dihydro-1H-pyrrole-3-carbonitrile (1) with a methylsulfanyl group as a good leaving group. As shown in Scheme 1, the one-pot reaction of 1 with 2-aminopyridine (2a) proceeded by refluxing in ethanol for 2 h to produce the ring-fused pyridine
Tosylhydrazine-promoted self-conjugate reduction–Michael/aldol reaction of 3-phenacylideneoxindoles towards dispirocyclopentanebisoxindole derivatives
Beilstein J. Org. Chem. 2022, 18, 469–478, doi:10.3762/bjoc.18.49
- and operational simplicity through one pot reaction. Keywords: chemoselective conjugate reduction; dispirocyclopentanebisoxindole scaffolds; metal-free; one-pot operation; reductive cyclization; Introduction There is a vast demand of the structurally complex spirooxindole scaffold which is an
Recent developments and trends in the iron- and cobalt-catalyzed Sonogashira reactions
Beilstein J. Org. Chem. 2022, 18, 262–285, doi:10.3762/bjoc.18.31
- cross-coupling reactions Homogeneous green protocols Tsai et al. discussed an efficient, simple and environmentally friendly method for the coupling of arylynols 3 with an aryl halide [21]. This strategy discloses a one pot reaction catalyzed by FeCl3 in an aqueous medium associated with the cationic
Bifunctional thiourea-catalyzed asymmetric [3 + 2] annulation reactions of 2-isothiocyanato-1-indanones with barbiturate-based olefins
Beilstein J. Org. Chem. 2022, 18, 25–36, doi:10.3762/bjoc.18.3
- the basic reaction conditions (Scheme 5b). A one-pot reaction of three available starting materials was tested using CH2Cl2 as the solvent. The one-pot reaction of 1,3-dimethylbarbituric acid (6), benzaldehyde (7), and 2-isothiocyanato-1-indanone (1a) proceeded smoothly to provide the desired product
- 3aa in 80% yield with 95% ee and >20:1 dr (Scheme 6a). In addition, the one-pot reaction of 1,3-dimethylbarbituric acid (6), m-bromobenzaldehyde (8), and 2-isothiocyanato-1-indanone (1a) was also investigated, and the reaction yield (80%) was lower than before, but the stereoselectivity (>20:1 dr, >99
N-Sulfinylpyrrolidine-containing ureas and thioureas as bifunctional organocatalysts
Beilstein J. Org. Chem. 2021, 17, 2629–2641, doi:10.3762/bjoc.17.176
- after each step prompted us to apply a Mitsunobu and Staudinger reaction for the preparation of amine 2 (Scheme 1) [33]. This one-pot reaction gave the desired amine 2 in 56% yield. Then, the corresponding isothiocyanate 3a was prepared by reaction of amine 2 with CS2 and DCC according to the reported
Efficient synthesis of polyfunctionalized carbazoles and pyrrolo[3,4-c]carbazoles via domino Diels–Alder reaction
Beilstein J. Org. Chem. 2021, 17, 2425–2432, doi:10.3762/bjoc.17.159
- diastereoisomers of tetrahydropyrrolo[3,4-c]carbazoles, which can be dehydrogenated by DDQ oxidation in acetonitrile at room temperature to give the aromatized pyrrolo[3,4-c]carbazoles in high yields. On the other hand, the one-pot reaction of 3-(indol-3-yl)-1,3-diphenylpropan-1-ones with chalcones or
- scope of this domino Diels–Alder reaction, another kind of 3-vinylindoles was employed in the one-pot reaction. First, the 3-(indol-3-yl)-1,3-diphenylpropan-1-ones prepared through Friedel–Crafts alkylation of indole with chalcones, were oxidized by DDQ in acetonitrile to generate in situ the expected
Recent advances in the syntheses of anthracene derivatives
Beilstein J. Org. Chem. 2021, 17, 2028–2050, doi:10.3762/bjoc.17.131
- 2012, Singh and co-workers performed green syntheses of oxa-aza-benzo[a]anthracene and oxa-aza-phenanthrene derivatives 151 and 152 via a sequential one-pot reaction in an aqueous micellar system (Scheme 34) [68]. This methodology comprised reactions of isoquinoline (147), phenacyl bromides 148 bearing
Synthetic accesses to biguanide compounds
Beilstein J. Org. Chem. 2021, 17, 1001–1040, doi:10.3762/bjoc.17.82

- apparition of side-products and a drop of the yields (Scheme 40). Recently, a one-pot reaction was reported by Yavari and Nematpour that includes the formation of an hexasubstituted biguanide from TMG, and a copper-catalyzed N-arylation [81]. Using TMG and DIC/DCC as reagents, and 10 mol % of CuI
Application of the Meerwein reaction of 1,4-benzoquinone to a metal-free synthesis of benzofuropyridine analogues
Beilstein J. Org. Chem. 2021, 17, 977–982, doi:10.3762/bjoc.17.79
- to hydroquinone 12 with N,N-diethylhydroxylamine (N,N-DEHA) and cyclized via intramolecular nucleophilic aromatic substitution to isolate 6-hydroxybenzofuro[2,3-b]pyridine (13) with 82% yield. Conveniently, the synthesis of 13 was achieved in a one-pot reaction from 11 with no significant differences
Microwave-assisted multicomponent reactions in heterocyclic chemistry and mechanistic aspects
Beilstein J. Org. Chem. 2021, 17, 819–865, doi:10.3762/bjoc.17.71

[2 + 1] Cycloaddition reactions of fullerene C60 based on diazo compounds
Beilstein J. Org. Chem. 2021, 17, 630–670, doi:10.3762/bjoc.17.55
- stage, viz, it becomes necessary to preliminarily halogenate the cyclopropanating agent. As a rule, a hardly separable mixture of mono- and dihalo-containing products is formed. The Hirsch-modified Bingel method makes it possible to isolate methanofullerenes by a one-pot reaction using simultaneously
Synthetic strategies of phosphonodepsipeptides
Beilstein J. Org. Chem. 2021, 17, 461–484, doi:10.3762/bjoc.17.41
- the N-Cbz-1-aminophosphonic monomethyl esters 12b,d and hydroxy esters 106 followed by the selective demethylation with TMSBr in a one-pot reaction. The method provides a mild route to prepare phosphonodepsipeptides. The yields were insensitive to the steric encumbrance of both reactants being coupled
- directly. The multicomponent condensation reaction was applied as a direct synthetic method for phosphonodepsipeptides via the formation of 1-aminoalkylphosphonic acids and simultaneous construction of the phosphonate bond. A series of phosphonodepsipeptides 158 was prepared in good yields in a one-pot
- reaction directly from the simple and commercially available chemicals, benzyl carbamate (154), aldehydes 155, and methyl dichlorophosphite (156), followed by the alcoholysis with the hydroxy esters 157. The current strategy is a highly efficient and convergent synthesis of phosphonodepsipeptides that does
Using multiple self-sorting for switching functions in discrete multicomponent systems
Beilstein J. Org. Chem. 2020, 16, 2831–2853, doi:10.3762/bjoc.16.233

- BPh4− at the cage [Co6(10')4]12+ (SelfSORT-I). Double self-sorting (only structural) Hahn et al. demonstrated for the first time that poly-NHC ligands furnish metallosupramolecular assemblies through narcissistic self-sorting [53]. The one-pot reaction of the tris-NHC ligands 13–15 with different
One-pot synthesis of oxazolidinones and five-membered cyclic carbonates from epoxides and chlorosulfonyl isocyanate: theoretical evidence for an asynchronous concerted pathway
Beilstein J. Org. Chem. 2020, 16, 1805–1819, doi:10.3762/bjoc.16.148
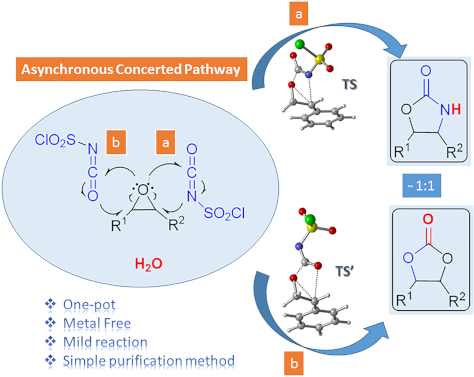
- Technology, Oltu Vocational School, Atatürk University, 25400 Oltu, Erzurum, Turkey Department of Chemistry, Faculty of Arts and Sciences, Marmara University, Goztepe Campus, 34722 Istanbul, Turkey 10.3762/bjoc.16.148 Abstract The one-pot reaction of chlorosulfonyl isocyanate (CSI) with epoxides having
- advantageous: being a one-pot reaction with metal-free reagent, having shorter reaction times, good yields and a very simple purification method. Moreover, using the density functional theory (DFT) method at the M06-2X/6-31+G(d,p) level of theory the mechanism of the cycloaddition reactions has been elucidated
- using a safe, inexpensive, metal-free reagent, a simple purification method and shorter reaction times via a one-pot reaction. The study presents a useful method for one-pot conversion of epoxides to protected 1,2-diols and 2-amino alcohols in one reaction. In the computational part of the study, the



















































































































































































































































































































































































































































































































































































































