Search results
Search for "alcohol" in Full Text gives 1179 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Trifluoromethylated hydrazones and acylhydrazones as potent nitrogen-containing fluorinated building blocks
- Zhang Dongxu
Beilstein J. Org. Chem. 2023, 19, 1741–1754, doi:10.3762/bjoc.19.127
- indispensable role in drug discovery and design since the hydrogen atom can act as lipophilic hydrogen-bond donor and act as a bioisostere for the alcohol moiety [81][82][83]. Thus, many effective difluoromethylation strategies have been developed in recent years. Difluoroacetohydrazonoyl bromides were chosen

Graphical Abstract

Scheme 1: Synthesis of trifluoromethylpyrazoles from trifluoroacetaldehyde hydrazones.

Scheme 2: Synthesis of polysubstituted pyrazolidines and pyrazolines.

Scheme 3: Asymmetric synthesis of 3-trifluoromethyl-1,4-dihydropyridazines reported by Rueping et al. [39].

Scheme 4: Synthesis of 3-trifluoromethyl-1,4-dihydropyridazine with Brønsted acid-assisted Lewis base catalys...

Scheme 5: Synthesis of CF3-pyrazoles and CF3-1,6-dihydropyridazines.

Scheme 6: Asymmetric reactions of trifluoromethylimines with organometallic reagents.

Scheme 7: Mannich-type reaction of trifluoroacetaldehyde hydrazones.

Scheme 8: Synthesis of trifluoromethylated hydrazonoyl halides.

Scheme 9: Early work of trifluoromethylated hydrazonoyl halides.

Scheme 10: [3 + 2]/[3 + 3] Cycloadditions of trifluoromethylated hydrazonoyl halides.

Scheme 11: Substrate scope for [3 + 2] cycloadditions with trifluoroacetonitrile imines reported by Jasiński’s...

Scheme 12: Synthesis of trifluoromethylated 1,2,4-triazole and 1,2,4-triazine derivatives.

Scheme 13: [3 + 2] Cycloadditions of difluoromethylated hydrazonoyl halides.

Scheme 14: Preparation and early applications of trifluoromethylated acylhydrazones.

Scheme 15: 1,2-Nucleophilic addition reactions of trifluoromethylated acylhydrazones.

Scheme 16: Cascade oxidation/cyclization reactions of trifluoromethylated homoallylic acylhydrazines.

Scheme 17: Synthesis of trifluoromethylated cyanohydrazines and 3-trifluoromethyl-1,2,4-triazolines.

Scheme 18: N-Arylation and N-alkylation of trifluoromethyl acylhydrazones.

Scheme 19: [3 + 2]-Cycladditions of trifluoromethyl acylhydrazones.
A series of perylene diimide cathode interlayer materials for green solvent processing in conventional organic photovoltaics
- Kathryn M. Wolfe,
- Shahidul Alam,
- Eva German,
- Fahad N. Alduayji,
- Maryam Alqurashi,
- Frédéric Laquai and
- Gregory C. Welch
Beilstein J. Org. Chem. 2023, 19, 1620–1629, doi:10.3762/bjoc.19.119

- film, but use of such alcohol-based solvents limits the types of organic materials to be used as CILs. With ethyl acetate as a processing solvent a wider range of organic materials can be developed and studied as CILs. Herein, we report on the design, synthesis, and application of four new CILs using

Graphical Abstract

Figure 1: a) Select cathode interlayers used to make high-performance OPVs previously reported in the literat...

Figure 2: Evolution of the dimer tPDI2N-R [23] as an NFA to PDIN-B/PDIN-FB monomers to serve as CILs, where the a...

Scheme 1: Synthesis schematic for PDIN-FB, PDIN-B, CN-PDIN-FB, and CN-PDIN-B.

Figure 3: a) Structures of PDIN-FB, PDIN-B, CN-PDIN-FB, and CN-PDIN-B. b) Normalized solution UV–visible spec...

Figure 4: a) PDIN-FB, PDIN-B, CN-PDIN-FB, and CN-PDIN-B structures with their respective HOMO/LUMO level ener...

Figure 5: a) Chemical structures of BHJ donor material PM6 and acceptor material Y6, b) conventional OPV devi...

Figure 6: a) Current density-voltage (I–V) characterization under illumination, b) spectral response of the s...
Radical chemistry in polymer science: an overview and recent advances
- Zixiao Wang,
- Feichen Cui,
- Yang Sui and
- Jiajun Yan
Beilstein J. Org. Chem. 2023, 19, 1580–1603, doi:10.3762/bjoc.19.116
- dissolved initiators in the liquid phase during the process. A suspending agent, e.g., polyvinyl alcohol (PVA), is added to the system to prevent coalescence. The viscosity in suspension polymerization is low throughout the process which brings good heat transfer and temperature control, and therefore well

Graphical Abstract

Scheme 1: Oxidation of catechol and subsequent cross-linking. Scheme 1 redrawn from [3].

Scheme 2: (A) Structure of typical urushiol in Chinese lacquer, and (B) schematic process of laccase-catalyze...

Scheme 3: A) Primary amino acid sequence of mfp-1, mfp-3, and mfp-5 (Y: DOPA, K: lysine). B) Scheme showing e...

Scheme 4: Activation–deactivation equilibrium in nitroxide-mediated polymerizations. Bicomponent initiating s...

Scheme 5: Mechanism of a transition metal complex-mediated ATRP. Scheme 5 redrawn from [14].

Scheme 6: Mechanism of RAFT polymerization. Scheme 6 redrawn from [68].

Scheme 7: Degenerative transfer (a) and reversible termination (b) mechanism of OMRP. Scheme 7 redrawn from [70].

Scheme 8: Simplified mechanism of a RITP. Scheme 8 redrawn from [21].

Scheme 9: (A) Structures of π-conjugated conductive polymers. (B) Examples of conductive polymer synthesis vi...

Scheme 10: Possible regiochemical couplings in PATs. Scheme 10 redrawn from [79].

Scheme 11: General thiol-ene photopolymerization process. Scheme 11 redrawn from [81].

Scheme 12: (a) Three generations of Grubbs catalysts. (b) Proposed mechanism for photo-ROMP via a reductive qu...

Scheme 13: Pyrylium and thiopyrylium salts studied by Boydston et al. Scheme 13 redrawn from [91].

Scheme 14: A general illustration of post-polymerization modification by thiol–ene chemistry.

Scheme 15: Introduction of functionalities by nitroxide radical coupling of HO-TEMPO derivatives.

Scheme 16: Chemical reaction process scheme of DCP-induced crosslinking of LDPE. Scheme 16 redrawn from [126].

Scheme 17: A probable mechanism of radical-induced hydrosilylation.

Scheme 18: Polymer surface modification by homolytic dediazonation of diazonium salts.

Scheme 19: Photoinduced polymer surface modification or surface grafting using benzophenone.

Scheme 20: Depolymerization mechanism of common photoresists. (a) A possible mechanism of radiation decomposit...

Scheme 21: Proposed mechanisms of photooxidative depolymerization of polystyrene. (a) Scheme 21a was reprinted with perm...
N-Sulfenylsuccinimide/phthalimide: an alternative sulfenylating reagent in organic transformations
- Fatemeh Doraghi,
- Seyedeh Pegah Aledavoud,
- Mehdi Ghanbarlou,
- Bagher Larijani and
- Mohammad Mahdavi
Beilstein J. Org. Chem. 2023, 19, 1471–1502, doi:10.3762/bjoc.19.106

- method, diverse β-alkoxy sulfides were synthesized without the need to any catalyst, or additive. The reaction proceeded through the formation of carbonium ion intermediate I, which underwent electrophilic addition of alcohol to provide product 152. In the meantime, N-(arylthio)succinimide 1 as a

Graphical Abstract

Scheme 1: Sulfur-containing bioactive molecules.

Scheme 2: Scandium-catalyzed synthesis of thiosulfonates.

Scheme 3: Palladium-catalyzed aryl(alkyl)thiolation of unactivated arenes.

Scheme 4: Catalytic cycle for Pd-catalyzed aryl(alkyl)thiolation of unactivated arenes.

Scheme 5: Iron- or boron-catalyzed C–H arylthiation of substituted phenols.

Scheme 6: Iron-catalyzed azidoalkylthiation of alkenes.

Scheme 7: Plausible mechanism for iron-catalyzed azidoalkylthiation of alkenes.

Scheme 8: BF3·Et2O‑mediated electrophilic cyclization of aryl alkynoates.

Scheme 9: Tentative mechanism for BF3·Et2O‑mediated electrophilic cyclization of aryl alkynoates.

Scheme 10: Construction of 6-substituted benzo[b]thiophenes.

Scheme 11: Plausible mechanism for construction of 6-substituted benzo[b]thiophenes.

Scheme 12: AlCl3‑catalyzed cyclization of N‑arylpropynamides with N‑sulfanylsuccinimides.

Scheme 13: Synthetic utility of AlCl3‑catalyzed cyclization of N‑arylpropynamides with N‑sulfanylsuccinimides.

Scheme 14: Sulfenoamination of alkenes with sulfonamides and N-sulfanylsuccinimides.

Scheme 15: Lewis acid/Brønsted acid controlled Pd-catalyzed functionalization of aryl C(sp2)–H bonds.

Scheme 16: Possible mechanism for Lewis acid/Brønsted acid controlled Pd-catalyzed functionalization of aryl C...

Scheme 17: FeCl3-catalyzed carbosulfenylation of unactivated alkenes.

Scheme 18: Copper-catalyzed electrophilic thiolation of organozinc halides.

Scheme 19: h-BN@Copper(II) nanomaterial catalyzed cross-coupling reaction of sulfoximines and N‑(arylthio)succ...

Scheme 20: AlCl3‑mediated cyclization and sulfenylation of 2‑alkyn-1-one O‑methyloximes.

Scheme 21: Lewis acid-promoted 2-substituted cyclopropane 1,1-dicarboxylates with sulfonamides and N-(arylthio...

Scheme 22: Lewis acid-mediated cyclization of β,γ-unsaturated oximes and hydrazones with N-(arylthio/seleno)su...

Scheme 23: Credible pathway for Lewis acid-mediated cyclization of β,γ-unsaturated oximes with N-(arylthio)suc...

Scheme 24: Synthesis of 4-chalcogenyl pyrazoles via chalcogenation/cyclization of α,β-alkynic hydrazones.

Scheme 25: Controllable synthesis of 3-thiolated pyrroles and pyrrolines.

Scheme 26: Possible mechanism for controllable synthesis of 3-thiolated pyrroles and pyrrolines.

Scheme 27: Co-catalyzed C2-sulfenylation and C2,C3-disulfenylation of indole derivatives.

Scheme 28: Plausible catalytic cycle for Co-catalyzed C2-sulfenylation and C2,C3-disulfenylation of indoles.

Scheme 29: C–H thioarylation of electron-rich arenes by iron(III) triflimide catalysis.

Scheme 30: Difunctionalization of alkynyl bromides with thiosulfonates and N-arylthio succinimides.·

Scheme 31: Suggested mechanism for difunctionalization of alkynyl bromides with thiosulfonates and N-arylthio ...

Scheme 32: Synthesis of thioesters, acyl disulfides, ketones, and amides by N-thiohydroxy succinimide esters.

Scheme 33: Proposed mechanism for metal-catalyzed selective acylation and acylthiolation.

Scheme 34: AlCl3-catalyzed synthesis of 3,4-bisthiolated pyrroles.

Scheme 35: α-Sulfenylation of aldehydes and ketones.

Scheme 36: Acid-catalyzed sulfetherification of unsaturated alcohols.

Scheme 37: Enantioselective sulfenylation of β-keto phosphonates.

Scheme 38: Organocatalyzed sulfenylation of 3‑substituted oxindoles.

Scheme 39: Sulfenylation and chlorination of β-ketoesters.

Scheme 40: Intramolecular sulfenoamination of olefins.

Scheme 41: Plausible mechanism for intramolecular sulfenoamination of olefins.

Scheme 42: α-Sulfenylation of 5H-oxazol-4-ones.

Scheme 43: Metal-free C–H sulfenylation of electron-rich arenes.

Scheme 44: TFA-promoted C–H sulfenylation indoles.

Scheme 45: Proposed mechanism for TFA-promoted C–H sulfenylation indoles.

Scheme 46: Organocatalyzed sulfenylation and selenenylation of 3-pyrrolyloxindoles.

Scheme 47: Organocatalyzed sulfenylation of S-based nucleophiles.

Scheme 48: Conjugate Lewis base Brønsted acid-catalyzed sulfenylation of N-heterocycles.

Scheme 49: Mechanism for activation of N-sulfanylsuccinimide by conjugate Lewis base Brønsted acid catalyst.

Scheme 50: Sulfenylation of deconjugated butyrolactams.

Scheme 51: Intramolecular sulfenofunctionalization of alkenes with phenols.

Scheme 52: Organocatalytic 1,3-difunctionalizations of Morita–Baylis–Hillman carbonates.

Scheme 53: Organocatalytic sulfenylation of β‑naphthols.

Scheme 54: Acid-promoted oxychalcogenation of o‑vinylanilides with N‑(arylthio/arylseleno)succinimides.

Scheme 55: Lewis base/Brønsted acid dual-catalytic C–H sulfenylation of aryls.

Scheme 56: Lewis base-catalyzed sulfenoamidation of alkenes.

Scheme 57: Cyclization of allylic amide using a Brønsted acid and tetrabutylammonium chloride.

Scheme 58: Catalytic electrophilic thiocarbocyclization of allenes with N-thiosuccinimides.

Scheme 59: Suggested mechanism for electrophilic thiocarbocyclization of allenes with N-thiosuccinimides.

Scheme 60: Chiral chalcogenide-catalyzed enantioselective hydrothiolation of alkenes.

Scheme 61: Proposed mechanism for chalcogenide-catalyzed enantioselective hydrothiolation of alkenes.

Scheme 62: Organocatalytic sulfenylation for synthesis a diheteroatom-bearing tetrasubstituted carbon centre.

Scheme 63: Thiolative cyclization of yne-ynamides.

Scheme 64: Synthesis of alkynyl and acyl disulfides from reaction of thiols with N-alkynylthio phthalimides.

Scheme 65: Oxysulfenylation of alkenes with 1-(arylthio)pyrrolidine-2,5-diones and alcohols.

Scheme 66: Arylthiolation of arylamines with (arylthio)-pyrrolidine-2,5-diones.

Scheme 67: Catalyst-free isothiocyanatoalkylthiation of styrenes.

Scheme 68: Sulfenylation of (E)-β-chlorovinyl ketones toward 3,4-dimercaptofurans.

Scheme 69: HCl-promoted intermolecular 1, 2-thiofunctionalization of aromatic alkenes.

Scheme 70: Possible mechanism for HCl-promoted 1,2-thiofunctionalization of aromatic alkenes.

Scheme 71: Coupling reaction of diazo compounds with N-sulfenylsuccinimides.

Scheme 72: Multicomponent reactions of disulfides with isocyanides and other nucleophiles.

Scheme 73: α-Sulfenylation and β-sulfenylation of α,β-unsaturated carbonyl compounds.
Application of N-heterocyclic carbene–Cu(I) complexes as catalysts in organic synthesis: a review
- Nosheen Beig,
- Varsha Goyal and
- Raj K. Bansal
Beilstein J. Org. Chem. 2023, 19, 1408–1442, doi:10.3762/bjoc.19.102
- . Two complexes, 61 and 65 (see Scheme 21), were found equally efficient. The products were obtained in 95 to 99% yield and the range of functionalities tolerated included nitro, nitrile, ether, carbonyl, alcohol, and amine [35]. Gautier and co-workers studied the effect of the addition of aromatic N

Graphical Abstract

Scheme 1: In situ generation of imidazolylidene carbene.

Scheme 2: Hg(II) complex of NHC.

Scheme 3: Isolable and bottlable carbene reported by Arduengo [3].

Scheme 4: First air-stable carbene synthesized by Arduengo in 1992 [5].

Figure 1: General structure of an NHC.

Figure 2: Stabilization of an NHC by donation of the lone pair electrons into the vacant p-orbital (LUMO) at ...

Figure 3: Abnormal NHC reported by Bertrand [8,9].

Figure 4: Cu(d) orbital to σ*C-N(NHC) interactions in NHC–CuX complexes computed at the B3LYP/def2-SVP level ...

Figure 5: Molecular orbital contributions to the NHC–metal bond.

Scheme 5: Synthesis of NHC–Cu(I) complexes by deprotonation of NHC precursors with a base.

Scheme 6: Synthesis of [NHC–CuX] complexes.

Scheme 7: Synthesis of [(ICy)CuX] and [(It-Bu)CuX] complexes.

Scheme 8: Synthesis of iodido-bridged copper–NHC complexes by deprotonation of benzimidazolium salts reported...

Scheme 9: Synthesis of copper complexes by deprotonation of triazolium salts.

Scheme 10: Synthesis of thiazolylidene–Cu(I) complex by deprotonation with KOt-Bu.

Scheme 11: Preparation of NHC–Cu(I) complexes.

Scheme 12: Synthesis of methylmalonic acid-derived anionic [(26a,b)CuCl]Li(THF)2 and zwitterionic (28) heterol...

Scheme 13: Synthesis of diaminocarbene and diamidocarbene (DAC)–Cu(I) complexes.

Scheme 14: Synthesis of the cationic (NHC)2Cu(I) complex 39 from benzimidazolium salts 38 with tetrakis(aceton...

Scheme 15: Synthesis of NHC and ADC (acyclic diamino carbenes) Cu(I) hexamethyldisilazide complexes reported b...

Scheme 16: Synthesis of NHC–copper(I) complexes using an acetylacetonate-functionalized imidazolium zwitterion...

Scheme 17: Synthesis of NHC–Cu(I) complexes through deprotonation of azolium salts with Cu2O.

Scheme 18: Synthesis of NHC–CuBr complex through deprotonation with Cu2O reported by Kolychev [31].

Scheme 19: Synthesis of chiral NHC–CuBr complexes from phenoxyimine-imidazolium salts reported by Douthwaite a...

Scheme 20: Preparation of linear neutral NHC–CuCl complexes through the use of Cu2O. For abbreviations, please...

Scheme 21: Synthesis of abnormal-NHC–copper(I) complexes by Bertrand, Cazin and co-workers [35].

Scheme 22: Microwave-assisted synthesis of thiazolylidene/benzothiazolylidene–CuBr complexes by Bansal and co-...

Scheme 23: Synthesis of NHC–CuX complexes through transmetallation.

Scheme 24: Preparation of six- or seven-membered NHC–Cu(I) complexes through transmetalation from Ag(I) comple...

Scheme 25: Synthesis of 1,2,3-triazolylidene–CuCl complexes through transmetallation of Ag(I) complexes genera...

Scheme 26: Synthesis of NHC–copper complexes having both Cu(I) and Cu(II) units through transmetalation report...

Scheme 27: Synthesis of new [(IPr(CH2)3Si(OiPr)3)CuX] complexes and anchoring on MCM-41.

Scheme 28: Synthesis of bis(trimethylsilyl)phosphide–Cu(I)–NHC complexes through ligand displacement.

Scheme 29: Synthesis of silyl- and stannyl [(NHC)Cu−ER3] complexes.

Scheme 30: Synthesis of amido-, phenolato-, thiophenolato–Cu(NHC) complexes.

Scheme 31: Synthesis of first isolable NHC–Cu–difluoromethyl complexes reported by Sanford et al. [44].

Scheme 32: Synthesis of NHC–Cu(I)–bifluoride complexes reported by Riant, Leyssens and co-workers [45].

Scheme 33: Conjugate addition of Et2Zn to enones catalyzed by an NHC–Cu(I) complex reported by Woodward in 200...

Scheme 34: Hydrosilylation of a carbonyl group.

Scheme 35: NHC–Cu(I)-catalyzed hydrosilylation of ketones reported by Nolan et al. [48,49].

Scheme 36: Application of chiral NHC–CuCl complex 104 for the enantioselective hydrosilylation of ketones.

Scheme 37: Hydrosilylation reactions catalyzed by NHC–Cu(Ot-Bu) complexes.

Scheme 38: NHC–CuCl catalyzed carbonylative silylation of alkyl halides.

Scheme 39: Nucleophilic conjugate addition to an activated C=C bond.

Figure 6: Molecular electrostatic potential maps (MESP) of two NHC–CuX complexes computed at the B3LYP/def2-S...

Scheme 40: Conjugate addition of Grignard reagents to 3-alkyl-substituted cyclohexenones catalyzed by a chiral...

Scheme 41: NHC–copper complex-catalyzed conjugate addition of Grignard reagent to 3-substituted hexenone repor...

Scheme 42: Conjugate addition or organoaluminum reagents to β-substituted cyclic enones.

Scheme 43: Conjugate addition of boronates to acyclic α,β-unsaturated carboxylic esters, ketones, and thioeste...

Scheme 44: NHC–Cu(I)-catalyzed hydroboration of an allene reported by Hoveyda [63].

Scheme 45: Conjugate addition of Et2Zn to cyclohexenone catalyzed by NHC–Cu(I) complex derived from benzimidaz...

Scheme 46: Asymmetric conjugate addition of diethylzinc to 3-nonen-2-one catalyzed by NHC–Cu complexes derived...

Scheme 47: General scheme of a [3 + 2] cycloaddition reaction.

Scheme 48: [3 + 2] Cycloaddition of azides with alkynes catalyzed by NHC–Cu(I) complexes reported by Diez-Gonz...

Scheme 49: Application of NHC–CuCl/N-donor combination to catalyze the [3 + 2] cycloaddition of benzyl azide w...

Scheme 50: [3 + 2] Cycloaddition of azides with acetylenes catalyzed by bis(NHC)–Cu complex 131 and mixed NHC–...

Figure 7: NHC–CuCl complex 133 as catalyst for the [3 + 2] cycloaddition of alkynes with azides at room tempe...

Scheme 51: [3 + 2] Cycloaddition of a bulky azide with an alkynylpyridine using [(NHC)Cu(μ-I)2Cu(NHC)] copper ...

Scheme 52: [3 + 2] Cycloaddition of benzyl azide with phenylacetylene under homogeneous and heterogeneous cata...

Scheme 53: [3 + 2] Cycloaddition of benzyl azide with acetylenes catalyzed by bisthiazolylidene dicopper(I) co...

Figure 8: Copper (I)–NHC linear coordination polymer 137 and its conversion into tetranuclear (138) and dinuc...

Scheme 54: An A3 reaction.

Scheme 55: Synthesis of SiO2-immobilized NHC–Cu(I) catalyst 141 and its application in the A3-coupling reactio...

Scheme 56: Preparation of dual-purpose Ru@SiO2–[(NHC)CuCl] catalyst system 142 developed by Bordet, Leitner an...

Scheme 57: Application of the catalyst system Ru@SiO2–[Cu(NHC)] 142 to the one-pot tandem A3 reaction and hydr...

Scheme 58: A3 reaction of phenylacetylene with secondary amines and aldehydes catalyzed by benzothiazolylidene...

Figure 9: Kohn–Sham HOMOs of phenylacetylene and NHC–Cu(I)–phenylacetylene complex computed at the B3LYP/def2...

Figure 10: Energies of the FMOs of phenylacetylene, iminium ion, and NHC–Cu(I)–phenylacetylene complex compute...

Scheme 59: NHC–Cu(I) catalyzed diboration of ketones 147 by reacting with bis(pinacolato)diboron (148) reporte...

Scheme 60: Protoboration of terminal allenes catalyzed by NHC–Cu(I) complexes reported by Hoveyda and co-worke...

Scheme 61: NHC–CuCl-catalyzed borylation of α-alkoxyallenes to give 2-boryl-1,3-butadienes.

Scheme 62: Regioselective hydroborylation of propargylic alcohols and ethers catalyzed by NHC–CuCl complexes 1...

Scheme 63: NHC–CuOt-Bu-catalyzed semihydrogenation and hydroborylation of alkynes.

Scheme 64: Enantioselective NHC–Cu(I)-catalyzed hydroborations of 1,1-disubstituted aryl olefins reported by H...

Scheme 65: Enantioselective NHC–Cu(I)-catalyzed hydroboration of exocyclic 1,1-disubstituted alkenes reported ...

Scheme 66: Markovnikov-selective NHC–CuOH-catalyzed hydroboration of alkenes and alkynes reported by Jones et ...

Scheme 67: Dehydrogenative borylation and silylation of styrenes catalyzed by NHC–CuOt-Bu complexes developed ...

Scheme 68: N–H/C(sp2)–H carboxylation catalyzed by NHC–CuOH complexes.

Scheme 69: C–H Carboxylation of benzoxazole and benzothiazole derivatives with CO2 using a 1,2,3-triazol-5-yli...

Scheme 70: Use of Cu(I) complex derived from diethylene glycol-functionalized imidazo[1,5,a] pyridin-3-ylidene...

Scheme 71: Allylation and alkenylation of polyfluoroarenes and heteroarenes catalyzed by NHC–Cu(I) complexes r...

Scheme 72: Enantioselective C(sp2)–H allylation of (benz)oxazoles and benzothiazoles with γ,γ-disubstituted pr...

Scheme 73: C(sp2)–H arylation of arenes catalyzed by dual NHC–Cu/NHC–Pd catalytic system.

Scheme 74: C(sp2)–H Amidation of (hetero)arenes with N-chlorocarbamates/N-chloro-N-sodiocarbamates catalyzed b...

Scheme 75: NHC–CuI catalyzed thiolation of benzothiazoles and benzoxazoles.
Functional characterisation of twelve terpene synthases from actinobacteria
- Anuj K. Chhalodia,
- Houchao Xu,
- Georges B. Tabekoueng,
- Binbin Gu,
- Kizerbo A. Taizoumbe,
- Lukas Lauterbach and
- Jeroen S. Dickschat
Beilstein J. Org. Chem. 2023, 19, 1386–1398, doi:10.3762/bjoc.19.100
- , Supporting Information File 1). The closest characterised terpene synthase with an amino acid sequence identity of 25% is the (1(10)E,4E,6S,7R)-germacradien-6-ol synthase from Streptomyces pratensis [33]. The recombinant enzyme efficiently converted FPP into one sesquiterpene alcohol whose electron

Graphical Abstract

Figure 1: Terpenes produced by characterised terpene synthases.

Figure 2: Phylogenetic tree constructed from the amino acid sequences of 4018 terpene synthase homologs. Blue...

Figure 3: A) Total ion chromatogram of the products obtained from FPP with KkdCS, B) EI mass spectrum of 10, ...

Figure 4: Total ion chromatograms of the products obtained from FPP A) with SlADS and B) with SsADS, C) EI ma...

Figure 5: Total ion chromatograms of the products obtained with NbEIZS A) from FPP and B) from GPP, and C) st...

Figure 6: Total ion chromatogram of the products obtained with SfES. Peak numbers refer to compound numbers i...

Scheme 1: A) Cope rearrangement of 24 and 26. B) Cyclisation mechanism from FPP to 23, identifying compound 26...

Figure 7: Total ion chromatograms of the products obtained with A) SsHS, B) ScHS, C) SfHS, and D) SmGAS. Peak...

Figure 8: A) Total ion chromatogram of the products obtained from GGPP with KkAS, B) EI mass spectrum of allo...
Synthesis of ether lipids: natural compounds and analogues
- Marco Antônio G. B. Gomes,
- Alicia Bauduin,
- Chloé Le Roux,
- Romain Fouinneteau,
- Wilfried Berthe,
- Mathieu Berchel,
- Hélène Couthon and
- Paul-Alain Jaffrès
Beilstein J. Org. Chem. 2023, 19, 1299–1369, doi:10.3762/bjoc.19.96
- alkenyl compounds, the vinyl ether function is characterized by a (Z)-configuration as shown in Figure 1. In addition, an acyl group is present on the secondary alcohol of the glycerol. This acyl group is constituted by a saturated or unsaturated lipid chain or, in the case of platelet-activating factor
- of the secondary alcohol produced 2.3. The primary alcohol was deprotected in acidic conditions to produce 2.4. The installation of the phosphocholine polar head group was achieved in two steps starting with the reaction of 2.4 with bromoethyl dichlorophosphate (2.5) to produce the phosphate
- derivative 2.6. The treatment of 2.6 with trimethylamine produced an ammonium salt. A treatment with silver carbonate was applied to remove any traces of bromide salts. Then, the secondary alcohol was deprotected by hydrogenolysis to produce 2.7 (lyso-PAF). Finally, the acetylation of the secondary alcohol

Graphical Abstract

Figure 1: Chemical structure of some natural ether lipids (ELs).

Figure 2: Synthesis of lyso-PAF and PAF from 1-O-alkylglycerol [64].

Figure 3: Synthesis of lyso-PAF from 1,3-benzylideneglycerol 3.1 [69].

Figure 4: A) Synthesis of the two enantiomers of octadecylglycerol (4.6 and 4.10) from ᴅ-mannitol (4.1); B) s...

Figure 5: Four-step synthesis of PAF 5.6 from (S)-glycidol [73].

Figure 6: Synthesis of 1-O-alkylglycerol A) from solketal, B) from ᴅ- or ʟ-tartaric acid and the intermediate ...

Figure 7: Synthesis of EL building blocks starting from substituted glycidol 7.1a–c [82].

Figure 8: Synthesis of PAF 8.5 by using phosphoramidite 8.2 [86].

Figure 9: Synthesis of oleyl-PAF 9.7 from ʟ-serine [88].

Figure 10: Synthesis of racemic analogues of lyso-PAF 10.8 and PAF 10.9 featuring a phenyl group between the g...

Figure 11: Synthesis of racemic deoxy-lyso-PAF 11.7 and deoxy-PAF 11.8 [91].

Figure 12: Synthesis of racemic thio-PAF 12.8 [93].

Figure 13: Racemic synthesis of 13.6 to illustrate the modification of the glycerol backbone by adding a methy...

Figure 14: Racemic synthesis of 14.5 as an illustration of the introduction of methyl substituents on the glyc...

Figure 15: Synthesis of functionalized sn-2-acyl chains of PC-EL; A) Steglich esterification or acylation reac...

Figure 16: Synthesis of racemic mc-PAF (16.3), a carbamate analogue of PAF [102].

Figure 17: A) Synthesis of (R)-17.2 and (S)-17.6 starting from (S)-solketal (17.1); B) synthesis of N3-PAF (17...

Figure 18: Modification of the phosphocholine polar head to produce PAF analogues [81].

Figure 19: Racemic PAF analogues 19.3 and 19.5 characterized by the absence of the phosphate group [107].

Figure 20: Synthesis of PIP3-PAF (20.7) [108].

Figure 21: Large-scale synthesis of C18-edelfosine (21.8) [116].

Figure 22: Synthesis of C16-edelfosine (22.10) starting from isopropylidene-ʟ-glyceric acid methyl ester (22.1...

Figure 23: Phosphocholine moiety installation by the use of chlorophosphite 23.2 as key reagent [119].

Figure 24: Synthesis of rac-1-alkyl-2-O-methylglycerol (AMG) [120].

Figure 25: Synthesis of stereocontrolled 1-alkyl-2-O-methyl glycerol 25.9 (AMG) from dimethyl ᴅ-tartrate [81].

Figure 26: A) Racemic synthesis of thioether 26.4 [129,130], B) structure of sulfone analogue 26.5 [129].

Figure 27: Stereocontrolled synthesis of C18-edelfosine thioether analogue 27.8 [118].

Figure 28: Synthesis of thioether 28.4 that include a thiophosphate function [134].

Figure 29: Synthesis of ammonium thioether 29.4 and 29.6 [135].

Figure 30: Synthesis of the N-methylamino analogue of edelfosine 30.6 (BN52211) [138].

Figure 31: Synthesis of 1-desoxy analogues of edelfosine; A) with a saturated alkyl chain; B) synthesis of the...

Figure 32: Stereocontrolled synthesis of edelfosine analogue (S)-32.8 featuring a C18:1 lipid chain [142].

Figure 33: Synthesis of edelfosine analogues with modulation of the lipid chain; A) illustration with the synt...

Figure 34: Synthesis of phospholipid featuring a carbamate function to link the lipid chain to the glycerol un...

Figure 35: Synthesis of sesquiterpene conjugates of phospho glycero ether lipids [148].

Figure 36: Racemic synthesis of methyl-substituted glycerol analogues 36.7 and 36.10: A) synthesis of diether ...

Figure 37: Racemic synthesis of ilmofosine (37.6) [155,156].

Figure 38: A) Stereoselective synthesis of 38.5 via a stereoselective hydroboration reaction; B) synthesis of ...

Figure 39: Racemic synthesis of SRI62-834 (39.6) featuring a spiro-tetrahydrofurane heterocycle in position 2 ...

Figure 40: Racemic synthesis of edelfosine analogue 40.5 featuring an imidazole moiety in sn-2 position [160].

Figure 41: Racemic synthesis of fluorine-functionalized EL: A) Synthesis of 41.6 and B) synthesis of 41.8 [161-163].

Figure 42: A) Synthesis of the β-keto-ester 42.6 that also features a decyl linker between the phosphate and t...

Figure 43: Synthesis of phosphonate-based ether lipids; A) edelfosine phosphonate analogue 43.7 and B) thioeth...

Figure 44: Enantioselective synthesis of phosphonates 44.3 and 44.4 [171].

Figure 45: Racemic synthesis of phosphinate-based ether lipid 45.10 [172].

Figure 46: Racemic synthesis of edelfosine arsonium analogue 46.5 [173].

Figure 47: Synthesis of edelfosine dimethylammonium analogue 47.2 [118].

Figure 48: Synthesis of rac-C18-edelfosine methylammonium analogue 48.4 [176].

Figure 49: A) Synthesis of edelfosine N-methylpyrrolidinium analogue 49.2 or N-methylmorpholinium analogue 49.3...

Figure 50: A) Synthesis of edelfosine’s analogue 50.4 with a PE polar group; B) illustration of a pyridinium d...

Figure 51: A) Synthesis of 51.4 featuring a thiazolium cationic moiety; B) synthesis of thiazolium-based EL 51...

Figure 52: Synthesis of cationic ether lipids 52.3, 52.4 and 52.6 [135,183].

Figure 53: Synthesis of cationic carbamate ether lipid 53.5 [184].

Figure 54: Synthesis of cationic sulfonamide 54.5 [185].

Figure 55: Chemical structure of ONO-6240 (55.1) and SRI-63-119 (55.2).

Figure 56: Synthesis of non-ionic ether lipids 56.2–56.9 [188].

Figure 57: Synthesis of ether lipid conjugated to foscarnet 57.6 [189].

Figure 58: A) Synthesis of ether lipid conjugated to arabinofuranosylcytosine; B) synthesis of AZT conjugated ...

Figure 59: Synthesis of quercetin conjugate to edelfosine [191].

Figure 60: Synthesis of 60.8 (Glc-PAF) [194].

Figure 61: A) Synthesis of amino ether lipid 61.7 functionalized with a rhamnose unit and its amide analogue 6...

Figure 62: A) Synthesis of glucose ether lipid 62.4; B) structure of ether lipid 62.5 possessing a maltose uni...

Figure 63: A) Synthesis of glucuronic methyl ester 63.8; B) structure of cellobiose 63.9 and maltose 63.10 ana...

Figure 64: A) Synthesis of maltosyl glycerolipid 64.7; B) structure of lactose analogue 64.8 prepared followin...

Figure 65: A) Asymmetric synthesis of the aglycone moiety starting from allyl 4-methoxyphenyl ether; B) glycos...

Figure 66: A) Synthesis of ohmline possessing a lactose moiety. B) Structure of other glyco glycero lipids pre...

Figure 67: A) Synthesis of lactose-glycerol ether lipid 67.5; B) analogues possessing a maltose (67.6) or meli...

Figure 68: Synthesis of digalactosyl EL 68.6, A) by using trityl, benzyl and acetyl protecting groups, B) by u...

Figure 69: A) Synthesis of α-ohmline; B) structure of disaccharide ether lipids prepared by using similar meth...

Figure 70: Synthesis of lactose ether lipid 70.3 and its analogue 70.6 featuring a carbamate function as linke...

Figure 71: Synthesis of rhamnopyranoside diether 71.4 [196].

Figure 72: Synthesis of 1-O-hexadecyl-2-O-methyl-3-S-(α-ᴅ-1'-thioglucopyranosyl)-sn-glycerol (72.5) [225].

Figure 73: A) Preparation of lipid intermediate 73.4; B) synthesis of 2-desoxy-C-glycoside 73.10 [226].

Figure 74: Synthesis of galactose-pyridinium salt 74.3 [228].

Figure 75: Synthesis of myo-inositol derivative Ino-C2-PAF (75.10) [230].

Figure 76: A) Synthesis of myo-inositol phosphate building block 76.7; B) synthesis of myo-inositolphosphate d...

Figure 77: A) Synthesis of phosphatidyl-3-desoxy-inositol 77.4; B) synthesis of phosphono-3-desoxyinositol 77.9...

Figure 78: A) Structure of diether phosphatidyl-myo-inositol-3,4-diphosphate 78.1; B) synthesis of phosphatidy...

Figure 79: A) Synthesis of diether-phosphatidyl derivative 79.4 featuring a hydroxymethyl group in place of a ...

Figure 80: Synthesis of Glc-amine-PAF [78].

Figure 81: Synthesis of glucosamine ether lipid 81.4 and its analogues functionalized in position 3 of the ami...

Figure 82: Synthesis of fully deprotected aminoglucoside ether lipid 82.5 [246].

Figure 83: Synthesis of C-aminoglycoside 83.12 using Ramberg–Bäcklund rearrangement as a key step [250].

Figure 84: A) List of the most important glyco lipids and amino glyco lipids included in the study of Arthur a...

Figure 85: Synthesis of mannosamine ether lipid 85.6 [254].

Figure 86: A) Synthesis of glucosamine ether lipids with a non-natural ʟ-glucosamine moiety; B) synthesis of e...

Figure 87: A) Structure of the most efficient anticancer agents 87.1–87.4 featuring a diamino glyco ether lipi...

Figure 88: A) Synthesis of diamino glyco ether lipid 87.4; B) synthesis of bis-glycosylated ether lipid 88.10 [256]....

Figure 89: Synthesis of triamino ether lipid 89.4 [260].

Figure 90: Synthesis of chlorambucil conjugate 90.7 [261].

Figure 91: Three main methods for the preparation of glycerol ether lipid 91.3; A) from solketal and via a tri...

Figure 92: Four different methods for the installation of the phosphocholine polar head group; A) method using...

Figure 93: Illustration of two methods for the installation of saccharides or aminosaccharides; A) O-glycosyla...
Non-noble metal-catalyzed cross-dehydrogenation coupling (CDC) involving ether α-C(sp3)–H to construct C–C bonds
- Hui Yu and
- Feng Xu
Beilstein J. Org. Chem. 2023, 19, 1259–1288, doi:10.3762/bjoc.19.94
- oxidative alkylation of cyclic benzyl ethers with malonates or ketones. Oxygen is used as a terminal oxidant at atmospheric pressure. The key intermediate of this oxidative coupling reaction is benzyl alcohol intermediate C (Scheme 4) [52]. The generation of N–O radicals from NHPI in the presence of oxygen
- coupling process. Initially, ether 64 interacts with tert-butoxyl radicals via hydrogen atom transfer reaction to generate radical A with release of tert-butyl alcohol. Subsequently, the radical A adds to the C=C bond of α-oxo ketene dithioacetal 107 to form radical B, which further reacts with Fe(III) to

Graphical Abstract

Scheme 1: Research progress of coupling reactions and active compounds containing α-C(sp3)-functionalized eth...

Scheme 2: Transition-metal-catalyzed CDC pathways.

Scheme 3: CDC of active methylene compounds in the α-C(sp3) position of ethers.

Scheme 4: InCl3/Cu(OTf)2/NHPI co-catalyzed CDC reaction.

Scheme 5: CDC of cyclic benzyl ethers with aldehydes.

Scheme 6: Cu-catalyzed CDC of (a) unactivated C(sp3)–H ethers with simple ketones and (b) double C(sp3)−H fun...

Scheme 7: Cu-catalyzed CDC of C(sp3)–H/C(sp3)–H bonds.

Scheme 8: Cu-catalyzed synthesis of chiral 2-substituted tetrahydropyrans.

Scheme 9: CDC of thiazole with cyclic ethers.

Scheme 10: Cu(I)-catalyzed oxidative alkenylation of simple ethers.

Scheme 11: Cross-dehydrogenation coupling of isochroman C(sp3)–H bonds with anisole C(sp2)–H bonds.

Scheme 12: Pd(OAc)2/Cu(OTf)2-catalyzed arylation of α-C(sp3)–H bonds of ethers.

Scheme 13: Cu-catalyzed C(sp3)–H/C(sp2)–H activation strategies to construct C(sp3)–C(sp2) bonds.

Scheme 14: Cu(I)-catalyzed C(sp2)–H alkylation.

Scheme 15: Cu-catalyzed C(sp3)–H/C(sp)–H activation to construct C(sp3)–C(sp) bonds (H2BIP: 2,6-bis(benzimidaz...

Scheme 16: Fe-catalyzed CDC reaction pathways.

Scheme 17: Fe2(CO)9-catalyzed functionalization of C–H bonds.

Scheme 18: Ligand-promoted Fe-catalyzed CDC reaction of N-methylaniline with ethers.

Scheme 19: Fe-catalyzed CDC of C(sp3)–H/C(sp3)–H bonds.

Scheme 20: Fe-catalyzed hydroalkylation of α,β-unsaturated ketones with ethers.

Scheme 21: Solvent-free Fe(NO3)3-catalyzed CDC of C(sp3)–H/C(sp2)–H bonds.

Scheme 22: Alkylation of disulfide compounds to afford tetrasubstituted alkenes.

Scheme 23: Fe-catalyzed formation of 1,1-bis-indolylmethane derivatives.

Scheme 24: Alkylation of coumarins and flavonoids.

Scheme 25: Direct CDC α-arylation of azoles with ethers.

Scheme 26: CDC of terminal alkynes with C(sp3)–H bonds adjacent to oxygen, sulfur or nitrogen atoms.

Scheme 27: Alkylation of terminal alkynes.

Scheme 28: Co-catalyzed functionalization of glycine esters.

Scheme 29: Co-catalyzed construction of C(sp2)–C(sp3) bonds.

Scheme 30: Co-catalyzed CDC of imidazo[1,2-a]pyridines with isochroman.

Scheme 31: Co-catalyzed C–H alkylation of (benz)oxazoles with ethers.

Scheme 32: Cobalt-catalyzed CDC between unactivated C(sp2)–H and C(sp3)–H bonds.

Scheme 33: MnO2-catalyzed CDC of the inactive C(sp3)-H.

Scheme 34: Oxidative cross-coupling of ethers with enamides.

Scheme 35: Ni(II)-catalyzed CDC of indoles with 1,4-dioxane.

Scheme 36: Chemo- and regioselective ortho- or para-alkylation of pyridines.

Scheme 37: Asymmetric CDC of 3,6-dihydro-2H-pyrans with aldehydes.

Scheme 38: CDC of heterocyclic aromatics with ethers.

Scheme 39: Indium-catalyzed alkylation of DHPs with 1,3-dicarbonyl compounds.

Scheme 40: Rare earth-metal-catalyzed CDC reaction.

Scheme 41: Visible-light-driven CDC of cycloalkanes with benzazoles.

Scheme 42: Photoinduced alkylation of quinoline with cyclic ethers.

Scheme 43: Photocatalyzed CDC reactions between α-C(sp3)–H bonds of ethers and C(sp2)–H bonds of aromatics.
Metal catalyst-free N-allylation/alkylation of imidazole and benzimidazole with Morita–Baylis–Hillman (MBH) alcohols and acetates
- Olfa Mhasni,
- Jalloul Bouajila and
- Farhat Rezgui
Beilstein J. Org. Chem. 2023, 19, 1251–1258, doi:10.3762/bjoc.19.93
- direct amination of MBH alcohols 4a–d, with a poor leaving group (OH). Under the previous conditions (2 equiv of imidazole, toluene, reflux), the conversion of alcohol 4a [28] into the corresponding imidazole 6a was very slow and the starting materials were almost recovered. However, the continuous
- removal of water formed from the direct amination of alcohol 4a by azeotropic distillation shifted the position of the equilibrium in direction to the formation of the allylated imidazole 6a which was obtained in good 88% yield (Table 1, entry 6). The protocol was also successfully extended to the
- reaction of the primary five-membered alcohol 4b [29] with imidazole (2a) as well as to that of alcohols 4a,b with benzimidazole (2b), leading to the SN2-type products 6b and 7b,c, respectively, in 55–80% yields (Table 1, entries 7–9). In addition, we have shown that the direct amination of the available

Graphical Abstract

Figure 1: Medicines containing an imidazole nucleus.

Scheme 1: Synthesis of N-substituted imidazole derivatives from MBH adducts.

Scheme 2: Proposed mechanism for the allylation of imidazole with alcohol 4a.
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
- Grace A. Lowe
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
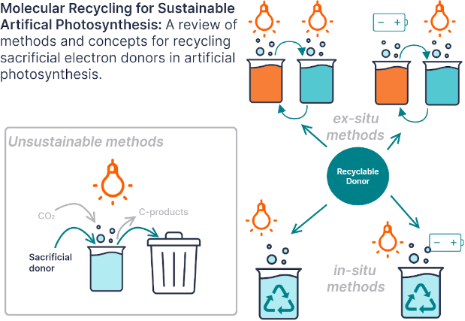
- to the core TEMPO or ferrocene charge-carrying moiety. This both increases the solubility of the species and the oxidation potential in aqueous media. TEMPOL uses an alcohol group to the same effect. Redox couples for RFB catholytes are optimized for increasing the oxidation which decreases their

Graphical Abstract

Figure 1: Diagram comparing the two reaction pathways for sacrificial electron donors (SD) in photocatalyzed ...

Figure 2: Diagram showing water-splitting systems developed by Girault, Scanlon, and co-workers that employ i...

Figure 3: Diagram illustrating the transfer of electrons in a photocatalytic particulate suspensions Z-scheme...

Figure 4: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...

Figure 5: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...
New one-pot synthesis of 4-arylpyrazolo[3,4-b]pyridin-6-ones based on 5-aminopyrazoles and azlactones
- Vladislav Yu. Shuvalov,
- Ekaterina Yu. Vlasova,
- Tatyana Yu. Zheleznova and
- Alexander S. Fisyuk
Beilstein J. Org. Chem. 2023, 19, 1155–1160, doi:10.3762/bjoc.19.83
- ). Their diluted alcohol solutions luminesce with a quantum yield of 0.09–0.23. Pyrazolo[3,4-b]pyridinones 4a–i, 9a, and 10a are characterized by an abnormally high Stokes shift (107–152 nm, 1.07–1.49 eV, Table 2). Such luminophores, which are colorless in daylight but become colored when irradiated with

Graphical Abstract

Figure 1: Biologically active 4-arylpyrazolo[3,4-b]pyridin-6-ones.

Scheme 1: Methods for the synthesis of 4-arylpyrazolo[3,4-b]pyridin-6-ones.

Scheme 2: One-pot synthesis of 4-arylpyrazolo[3,4-b]pyridin-6-ones 4a–i, 9a, and 10a.

Figure 2: Normalized absorption and fluorescence spectra of solutions of compounds 4a–i, 9a, and 10a in EtOH.
Photoredox catalysis harvesting multiple photon or electrochemical energies
- Mattia Lepori,
- Simon Schmid and
- Joshua P. Barham
Beilstein J. Org. Chem. 2023, 19, 1055–1145, doi:10.3762/bjoc.19.81

Graphical Abstract

Figure 1: Oxidative and reductive activations of organic compounds harvesting photoredox catalysis.

Figure 2: General catalytic cycles of radical ion conPET (left) and radical ion e-PRC (right).

Figure 3: “Beginner’s guide”: comparison between advantages, capacities, and prospectives of conPET and PEC.

Figure 4: A) conPET reductive dehalogenation of aryl halides with PDI. B) Reductive C–H arylation with pyrrol...

Figure 5: A) Chromoselective mono- and disubstitution or polybrominated pyrimidines with pyrroles. B) Sequent...

Figure 6: A) Synthesis of pyrrolo[1,2-a]quinolines. B) Synthesis of ullazines.

Figure 7: A) Reductive phosphorylation of aryl halides via conPET. B) Selected examples from the substrate sc...

Figure 8: A) Reductive dehalogenation of aryl halides via conPET and selected examples from the substrate sco...

Figure 9: A) Reductive C–H arylation of aryl halides via conPET (top) and selected examples from the substrat...

Figure 10: A) Reductive hydrodehalogenation of aryl halides with Mes-Acr-BF4. B) Selected examples from the su...

Figure 11: A) Reductive hydrodechlorination of aryl chlorides with 4-DPAIPN. B) Proposed formation of CO2•−. C...

Figure 12: A) Reductive conPET borylation with 3CzEPAIPN (top) and selected examples from the substrate scope ...

Figure 13: Scale-up of conPET phosphorylation with 3CzEPAIPN.

Figure 14: A) Borylation of 1d. B) Characteristics and structure of PC1 with green and red parts showing the l...

Figure 15: A) Reductive C–H arylation scope with polysulfide conPET (top) and selected examples from the subst...

Figure 16: Scale-up of A) C–H arylation and B) dehaloborylation with polysulfide photocatalysis in continuous-...

Figure 17: A) Formation of [Ir1]0 and [Ir2]0 upon PET between [Ir1]+ and Et3N. B) Mechanism of multi-photon ta...

Figure 18: A) Reductive hydrodehalogenation of aryl halides via multi-photon tandem photocatalysis. B) Selecte...

Figure 19: A) Carbonylative amidation of aryl halides in continuous flow. B) Selected examples from the substr...

Figure 20: A) General scheme for reductive (RQ) and oxidative quenching (OQ) protocols using [FeIII(btz)3](PF6)...

Figure 21: A) Carbonylative amidation of alkyl iodides with [IrIII(ppy)2(dtbbpy)]PF6. B) Selected examples fro...

Figure 22: A) Carboxylative C–N bond cleavage in cyclic amines. B) Selected examples from the substrate scope....

Figure 23: A) Formal reduction of alkenes to alkanes via transfer hydrogenation. B) Selected examples from the...

Figure 24: A) Birch-type reduction of benzenes with PMP-BPI. B) Selected examples from the substrate scope (sc...

Figure 25: Proposed mechanism of the OH− mediated conPET Birch-type reduction of benzene via generation of sol...

Figure 26: Reductive detosylation of N-tosylated amides with Mes-Acr-BF4. B) Selected examples from the substr...

Figure 27: A) Reductive detosylation of N-tosyl amides by dual PRC. B) Selected examples from the substrate sc...

Figure 28: A) Mechanism of the dual PRC based on PET between [Cu(dap)2]+ and DCA. B) Mechanism of the dual PRC...

Figure 29: A) N–O bond cleavage in Weinreb amides with anthracene. B) N–O bond cleavage in Weinreb amides rely...

Figure 30: A) Pentafluorosulfanylation and fluoride elimination. B) Mechanism of the pentafluorosulfanylation ...

Figure 31: A) α-Alkoxypentafluorosulfanylation (top) and selected examples from the substrate scope (bottom). ...

Figure 32: A) Oxidative amination of arenes with azoles catalyzed by N-Ph PTZ. B) Selected examples from the s...

Figure 33: A) C(sp3)–H bond activation by HAT via chloride oxidation by *N-Ph PTZ•+. B) Proposed mechanism for...

Figure 34: A) Recycling e-PRC C–H azolation of electron-rich arenes with pyrazoles using Mes-Acr+ as a photoca...

Figure 35: A) Radical ion e-PRC direct oxidation of unactivated arenes using TAC+ as an electro-activated phot...

Figure 36: A) Radical ion e-PRC direct oxidation of unactivated arenes using TPA as an electro-activated photo...

Figure 37: Proposed mechanism (top) and mode of preassembly (bottom).

Figure 38: A) Possible preassemblies of reactive (left) vs unreactive (right) arenes. B) Calculated spin densi...

Figure 39: A) Recycling e-PRC C(sp2 )–H acetoxylation of arenes using DDQ as a photocatalyst. B) Proposed cata...

Figure 40: Gram scale hydroxylation of benzene in a recirculated flow setup.

Figure 41: A) Radical ion e-PRC vicinal diamination of alkylarenes using TAC+ as an electro-activated photocat...

Figure 42: A) Sequential oxygenation of multiple adjacent C–H bonds under radical ion e-PRC using TAC+ as an e...

Figure 43: A) Enantioselective recycling e-PRC cyanation of benzylic C–H bonds using ADQS as photocatalyst. B)...

Figure 44: Proposed tandem mechanism by Xu and co-workers.

Figure 45: A) Enantioselective recycling e-PRC decarboxylative cyanation using Cu(acac)2, Ce(OTf)3 and a box l...

Figure 46: A) Enantioselective recycling e-PRC benzylic cyanation using Cu(MeCN)4BF4, box ligand and anthraqui...

Figure 47: A) Radical ion e-PRC acetoxyhydroxylation of aryl olefins using TAC+ as an electro-activated photoc...

Figure 48: Selected examples from the substrate scope.

Figure 49: Photoelectrochemical acetoxyhydroxylation in a recirculated flow setup.

Figure 50: A) Radical ion e-PRC aminooxygenation of aryl olefins using TAC+ as an electro-activated photocatal...

Figure 51: A) Recycling e-PRC C–H alkylation of heteroarenes with organic trifluoroborates using Mes-Acr+ as p...

Figure 52: A) Recycling e-PRC decarboxylative C–H alkylation of heteroarenes using CeCl3·7H2O as catalyst. B) ...

Figure 53: A) Recycling e-PRC decarboxylative C–H alkylation of heteroarenes using Fe(NH4)2(SO4)2·6H2O as cata...

Figure 54: A) Recycling e-PRC C–H alkylation of heteroarenes with alkyl oxalates and 4CzIPN as photocatalyst. ...

Figure 55: A) Recycling e-PRC decarboxylative C–H carbamoylation of heteroarenes using 4CzIPN as photocatalyst...

Figure 56: A) Photoelectrochemical HAT-mediated hydrocarbon activation via the chlorine radical. B) Proposed m...

Figure 57: A) Selected examples from the substrate scope. B) Gram and decagram scale semi-continuous flow PEC ...

Figure 58: A) Photoelectrochemical HAT-mediated dehydrogenative coupling of benzothiazoles with aliphatic C–H ...

Figure 59: A) Photoelectrochemical HAT activation of ethers using electro-activated TAC+ as photocatalyst. B) ...

Figure 60: Selected examples from the substrate scope.

Figure 61: A) Photoelectrochemical HAT-mediated synthesis of alkylated benzimidazo-fused isoquinolinones using...

Figure 62: A) Decoupled photoelectrochemical cerium-catalyzed oxydichlorination of alkynes using CeCl3 as cata...

Figure 63: Proposed decoupled photoelectrochemical mechanism.

Figure 64: A) Decoupled photoelectrochemical ring-opening bromination of tertiary cycloalkanols using MgBr2 as...

Figure 65: A) Recycling e-PRC ring-opening functionalization of cycloalkanols using CeCl3 as catalyst. B) Prop...

Figure 66: Selected examples from the substrate scope of the PEC ring-opening functionalization.

Figure 67: A) Radical ion e-PRC reduction of chloro- and bromoarenes using DCA as catalyst and various accepto...

Figure 68: A) Screening of different phthalimide derivatives as catalyst for the e-PRC reduction of aryl halid...

Figure 69: Screening of different organic catalysts for the e-PRC reduction of trialkylanilium salts.

Figure 70: A) e-PRC reduction of phosphonated phenols and anilinium salts. B) Selected examples from the subst...

Figure 71: A) ConPET and e-PRC reduction of 4-bromobenzonitrile using a naphthalene diimide (NDI) precatalyst ...

Figure 72: A) Radical ion e-PRC reduction of phosphinated aliphatic alcohols with n-BuO-NpMI as catalyst. B) C...

Figure 73: Selected examples from the substrate scope.

Figure 74: A) Recycling e-PRC reductive dimerization of benzylic chlorides using a [Cu2] catalyst. B) Proposed...

Figure 75: A) Decoupled photoelectrochemical C–H alkylation of heteroarenes through deamination of Katritzky s...

Figure 76: Proposed mechanism by Chen and co-workers.
CO2 complexation with cyclodextrins
- Cecilie Høgfeldt Jessen,
- Jesper Bendix,
- Theis Brock Nannestad,
- Heloisa Bordallo,
- Martin Jæger Pedersen,
- Christian Marcus Pedersen and
- Mikael Bols
Beilstein J. Org. Chem. 2023, 19, 1021–1027, doi:10.3762/bjoc.19.78
- solved in Olex2 using SHELXT and refined using SHELXL (Table 3) [19]. There is some disorder in one water molecule, which was modelled over two positions and some disorder in at least two of the primary alcohol groups, which was modelled. The latter disorder is not uncommon in α-cyclodextrin structures

Graphical Abstract

Figure 1: Structure of cyclodextrins 1–6 studied in this work.

Figure 2: X-ray crystal structure of CO2 bound to α-CD.

Figure 3: TGA curve (blue) and dTGA curve (red) for CO2-1 crystals. Two lumps are seen with the former predom...

Figure 4: Cell used to measure vis spectra under pressure (left), structure of 7 (middle) and spectrum of 7 (...

Figure 5: Binding of CO2 to 1 as a function of pressure.
Linker, loading, and reaction scale influence automated glycan assembly
- Marlene C. S. Dal Colle,
- Manuel G. Ricardo,
- Nives Hribernik,
- José Danglad-Flores,
- Peter H. Seeberger and
- Martina Delbianco
Beilstein J. Org. Chem. 2023, 19, 1015–1020, doi:10.3762/bjoc.19.77
- ] and L2 [3] are based on the o-nitrobenzyl scaffold [23][24] and expose a hydroxy group that serves as glycosyl acceptor in the first AGA cycle (Figure 1B). While L1 displays a flexible aliphatic chain terminating with a primary alcohol, L2 carries a secondary benzylic alcohol. Upon irradiation with UV

Graphical Abstract

Figure 1: Schematic representation of the AGA process (A). Variables that can affect the AGA outcome investig...

Figure 2: AGA of model glycan sequences analyzed in this study: α-1,6-linked dimannosides 1, 2 (A), branched ...

Figure 3: Representative HPLC traces for the crude compounds 1 (A), 3 (B), and 5a (C) after cleavage from the...
Five new sesquiterpenoids from agarwood of Aquilaria sinensis
- Hong Zhou,
- Xu-Yang Li,
- Hong-Bin Fang,
- He-Zhong Jiang and
- Yong-Xian Cheng
Beilstein J. Org. Chem. 2023, 19, 998–1007, doi:10.3762/bjoc.19.75
- ) and H-6/C-4 indicates the presence of a carboxyl group at C-4. Finally, the HMBC correlations of H-13/C-7 (δC 43.9), C-11 (δC 154.7), and C-12 (δC 65.0) demonstrate that C-7 is connected with acryl alcohol. Thus, the planar structure of 1 was assigned (Figure 1). However, the ROSEY data cannot provide
- mg) was separated by PTLC (petroleum ether/isopropyl alcohol 10:1) to afford eight fractions (Fr.6.7.9.5.14.1–Fr.6.7.9.5.14.8). Fr.6.7.9.5.14.3 (25.4 mg) was further purified by semi-preparative HPLC on YMC-PACK-ODS-A (aqueous MeCN, 65%) to yield 1 (12.7 mg, tR = 28.1 min, flow rate: 3 mL/min). Fr

Graphical Abstract

Figure 1: Structures of compounds 1–10.

Figure 2: Key 1H,1H-COSY and HMBC correlations for 1–5.

Figure 3: Key ROESY correlations for 2–5.

Figure 4: The calculated and experimental ECD spectra of 1–5.
Aromatic C–H bond functionalization through organocatalyzed asymmetric intermolecular aza-Friedel–Crafts reaction: a recent update
- Anup Biswas
Beilstein J. Org. Chem. 2023, 19, 956–981, doi:10.3762/bjoc.19.72
- after recrystallization. Subsequent ozonolysis of the terminal alkene functionality with a follow-up reduction furnished primary alcohol 134 which was transformed into the azide 135. Reduction of the azide 135 was accompanied by debenzylation, was followed by tosylation of the primary amine and exchange
- provided ketone 138 which was reacted with an α-keto ester in an intramolecular 5-endo-trig-cyclization process to afford 139. Treatment of compound 139 with sodium borohydride afforded secondary alcohol 140 which after conversion of the tosyl group into a methyl group gave the final product 141 (Scheme 35

Graphical Abstract

Scheme 1: First organocatalyzed asymmetric aza-Friedel–Crafts reaction.

Scheme 2: Aza-Friedel–Crafts reaction between indoles and cyclic ketimines.

Scheme 3: Aza-Friedel–Crafts reaction utilizing trifluoromethyldihydrobenzoazepinoindoles as electrophiles.

Scheme 4: Aza-Friedel–Crafts reaction utilizing cyclic N-sulfimines as electrophiles.

Scheme 5: Aza-Friedel–Crafts reaction involving N-unprotected imino ester as electrophile.

Scheme 6: Aza-Friedel–Crafts and lactonization cascade.

Scheme 7: One-pot oxidation and aza-Friedel–Crafts reaction.

Scheme 8: C1 and C2-symmetric phosphoric acids as catalysts.

Scheme 9: Aza-Friedel–Crafts reaction using Nps-iminophosphonates as electrophiles.

Scheme 10: Aza-Friedel–Crafts reaction between indole and α-iminophosphonate.

Scheme 11: [2.2]-Paracyclophane-derived chiral phosphoric acids as catalyst.

Scheme 12: Aza-Friedel–Crafts reaction through ring opening of sulfamidates.

Scheme 13: Isoquinoline-1,3(2H,4H)-dione scaffolds as electrophiles.

Scheme 14: Functionalization of the carbocyclic ring of substituted indoles.

Scheme 15: Aza-Friedel–Crafts reaction between unprotected imines and aza-heterocycles.

Scheme 16: Anilines and α-naphthols as potential nucleophiles.

Scheme 17: Solvent-controlled regioselective aza-Friedel–Crafts reaction.

Scheme 18: Generating central and axial chirality via aza-Friedel–Crafts reaction.

Scheme 19: Reaction between indoles and racemic 2,3-dihydroisoxazol-3-ol derivatives.

Scheme 20: Exploiting 5-aminoisoxazoles as nucleophiles.

Scheme 21: Reaction between unsubstituted indoles and 3-alkynylated 3-hydroxy-1-oxoisoindolines.

Scheme 22: Synthesis of unnatural amino acids bearing an aza-quaternary stereocenter.

Scheme 23: Atroposelective aza-Friedel–Crafts reaction.

Scheme 24: Coupling of 5-aminopyrazole and 3H-indol-3-ones.

Scheme 25: Pyrophosphoric acid-catalyzed aza-Friedel–Crafts reaction on phenols.

Scheme 26: Squaramide-assisted aza-Friedel–Crafts reaction.

Scheme 27: Thiourea-catalyzed aza-Friedel–Crafts reaction.

Scheme 28: Squaramide-catalyzed reaction between β-naphthols and benzothiazolimines.

Scheme 29: Thiourea-catalyzed reaction between β-naphthol and isatin-derived ketamine.

Scheme 30: Quinine-derived molecule as catalyst.

Scheme 31: Cinchona alkaloid as catalyst.

Scheme 32: aza-Friedel–Crafts reaction by phase transfer catalyst.

Scheme 33: Disulfonamide-catalyzed reaction.

Scheme 34: Heterogenous thiourea-catalyzed aza-Friedel–Crafts reaction.

Scheme 35: Total synthesis of (+)-gracilamine.

Scheme 36: Total synthesis of (−)-fumimycin.
Photoredox catalysis enabling decarboxylative radical cyclization of γ,γ-dimethylallyltryptophan (DMAT) derivatives: formal synthesis of 6,7-secoagroclavine
- Alessio Regni,
- Francesca Bartoccini and
- Giovanni Piersanti
Beilstein J. Org. Chem. 2023, 19, 918–927, doi:10.3762/bjoc.19.70
- ]. Although not yet completely clarified, some previous studies on the detailed mode of closure of the C ring in ergot alkaloids from DMAT have been shown to involve, before decarboxylative cyclization, oxidation on the C4-prenyl chain to give the stable rearranged allyl alcohol and/or the relatively unstable
- interconvertible to each other by hydration or dehydration, i.e., a plausible precursor of the allylic alcohol would be the diene, and vice versa [90]. Since both 8 and 10 are easily obtainable from 2 by Mozoroki–Heck coupling with commercially available 2-methyl-3-buten-2-ol, ester hydrolysis (LiOH in THF/H2O
- ), and, finally for 10, dehydration of the tertiary alcohol (mesylation and elimination) (Scheme 3), we decided to test their roles in the photoredox-catalyzed decarboxylative cyclization. With 8 and 10 in hand with the C4-prenyl side-chain already oxidized/functionalized, we recognized that this

Graphical Abstract

Figure 1: (a) Transformations of DMAT to different classes of ergot alkaloids. (b) and (c) Strategies for the...

Scheme 1: Synthesis of compound 5.

Scheme 2: Photoredox-catalyzed radical decarboxylative cyclization of 5.

Figure 2: Proposed reaction mechanism for photoredox-catalyzed radical decarboxylative cyclization.

Scheme 3: Synthesis of tryptophan derivatives 8 and 10.

Figure 3: Proposed reaction mechanism for photoredox-catalyzed radical decarboxylative cyclization.

Scheme 4: Methylation of 11 and the formal total synthesis of (±)-6,7-secoagroclavine.
Intermediates and shunt products of massiliachelin biosynthesis in Massilia sp. NR 4-1
- Till Steinmetz,
- Blaise Kimbadi Lombe and
- Markus Nett
Beilstein J. Org. Chem. 2023, 19, 909–917, doi:10.3762/bjoc.19.69
- carbon atoms C-13 and C-14 of the thiazole moiety, the loss of one degree of unsaturation and a loss of 2 Da in mass, it can be deduced that compound 5 possesses an alcohol function. Compound 6 (0.4 mg) was obtained as a brown oil. It possesses a molecular ion at m/z 291.1165 [M + H]+, which suggests a

Graphical Abstract

Figure 1: Selected siderophores from β-proteobacteria.

Figure 2: Chemical structures of compounds 1–6 isolated in this study and of the structurally related siderop...

Figure 3: 1H,1H-COSY and selected 1H,13C-HMBC correlations in 1.

Figure 4: Proposed origin of the isolated compounds 1–6 as well as massiliachelin (7). Domain notation of the...
Cyclodextrins as building blocks for new materials
- Miriana Kfoury and
- Sophie Fourmentin
Beilstein J. Org. Chem. 2023, 19, 889–891, doi:10.3762/bjoc.19.66

- reactivity of their alcohol functions. This allows regioselective chemical modification at either the primary or secondary rim [13]. As a result, these molecular hosts can be specifically linked either covalently or noncovalently to a wide variety of ligands. CDs are a significant part of almost all areas of
Non-peptide compounds from Kronopolites svenhedini (Verhoeff) and their antitumor and iNOS inhibitory activities
- Yuan-Nan Yuan,
- Jin-Qiang Li,
- Hong-Bin Fang,
- Shao-Jun Xing,
- Yong-Ming Yan and
- Yong-Xian Cheng
Beilstein J. Org. Chem. 2023, 19, 789–799, doi:10.3762/bjoc.19.59
- activity have revealed the presence of antimicrobial peptides [5], defensive alkaloids [6], and defensive long chain alcohol acetates [7]. Historically, in China, numerous records documented the utilization of animals like arthropods for medicinal purposes. Millipedes hereby represent a traditional Chinese

Graphical Abstract

Figure 1: Structures of compounds 1–9.

Figure 2: Key 1H–1H COSY, HMBC, and ROESY correlations of compounds 1–4, 7, and 8.

Figure 3: Calculated and experimental ECD spectra of compounds 1 and 4.

Figure 4: Biological evaluation. Compounds 3‒5 inhibited the expression of LPS-induced pro-inflammatory expre...
Honeycomb reactor: a promising device for streamlining aerobic oxidation under continuous-flow conditions
- Masahiro Hosoya,
- Yusuke Saito and
- Yousuke Horiuchi
Beilstein J. Org. Chem. 2023, 19, 752–763, doi:10.3762/bjoc.19.55

- ozonolysis reactions [33]. Herein, we describe the feasibility of the honeycomb reactor for aerobic oxidation. Results and Discussion Reaction screening for aerobic oxidation under batch conditions To select the representative aerobic oxidation, the reaction conditions to oxidize 4-methoxybenzyl alcohol (1a
- using the standard tube reactor and the honeycomb reactor (Scheme 4 and Table 5). With benzyl alcohol and benzyl alcohols bearing various substituents such as OMe, Me, Br, CF3 and COOMe at the 4-position and OMe at the 2- or 3-position, the reaction was uniformly accelerated using the honeycomb reactor
- aerobic oxidation using the honeycomb reactor. Flow setup for substrate scope and additional screening. Reaction screening for aerobic oxidation of 4-methoxybenzyl alcohol (1a). Reaction optimization for aerobic oxidation of 4-methoxybenzyl alcohol (1a). Evaluation of the reaction rate using various flow

Graphical Abstract

Figure 1: Honeycomb reactor. (a) Photograph. (b) Schematic diagram.

Scheme 1: Proposed catalytic cycle for aerobic oxidation using Fe(NO3)3/TEMPO.

Figure 2: Time course of the heat of reaction for aerobic oxidation.

Scheme 2: Flow setup for aerobic oxidation using various flow reactors.

Figure 3: Photographs of the various reactors. (a) Standard tube reactor. (b) Tube reactor with a static mixe...

Scheme 3: Flow setup for high-throughput aerobic oxidation using the honeycomb reactor.

Scheme 4: Flow setup for substrate scope and additional screening.
Photocatalytic sequential C–H functionalization expediting acetoxymalonylation of imidazo heterocycles
- Deepak Singh,
- Shyamal Pramanik and
- Soumitra Maity
Beilstein J. Org. Chem. 2023, 19, 666–673, doi:10.3762/bjoc.19.48
- chemodivergent formation of C-3 ethoxycarbonylmethylated and hydroxyalkylated IPs under visible light using water or alcohol as the source of the oxygenated group under degassed conditions [22]. However, all these photochemical methods require the usage of a substantial amount of base, the preactivation with a

Graphical Abstract

Scheme 1: Strategies of C-3 functionalizations of IPs and present work.

Scheme 2: Substrate scope. Conditions: unless otherwise noted, all reactions were carried out with 1 (0.2 mmo...

Scheme 3: Mechanistic investigations.

Scheme 4: Plausible reaction mechanism.
Nucleophile-induced ring contraction in pyrrolo[2,1-c][1,4]benzothiazines: access to pyrrolo[2,1-b][1,3]benzothiazoles
- Ekaterina A. Lystsova,
- Maksim V. Dmitriev,
- Andrey N. Maslivets and
- Ekaterina E. Khramtsova
Beilstein J. Org. Chem. 2023, 19, 646–657, doi:10.3762/bjoc.19.46
- yields of the corresponding products 3 and the general course of the reaction. However, the structure of the alcohols 2a–c had an effect on the studied reaction. Reactions with isopropyl alcohol 2b required longer reaction times (UPLC–UV–MS monitoring). This phenomenon could be due to the steric factors
- brought in by a bulky isopropyl substituent in alcohol 2b. In addition, in all studied cases we observed that the reaction of APBTTs 1 with alcohols 2 always afforded labile side-products 5 (Scheme 9). Compounds 5 were formed when the nucleophile 2 attacked on the position C3a of the substrates 1. Such a
- , benzylamine, and arylamines 11, while alkylamines are unsuitable for it. Notable, the use of bulky nucleophiles (tert-butyl alcohol (16a), benzyl alcohol (16b), benzhydrol (16c), 2-aminobenzothiazole (16d), HCl) makes it possible to obtain pyrrolobenzothiazoles 17 from compounds 1, but their formation

Graphical Abstract

Figure 1: Biologically active PBTAs.

Scheme 1: Approaches to PBTAs via annulation of benzothiazoles.

Scheme 2: Approaches to PBTAs via annulation of o-aminothiophenols.

Scheme 3: Approach to PBTAs via radical substitution reaction in 1-(2-bromophenyl)-5-(butylsulfanyl)pyrrolidi...

Scheme 4: Approach to PBTAs via intramolecular cyclizations of 1-(2-thiophenyl)pyrroles.

Scheme 5: A new approach to PBTAs via nucleophile-induced ring contraction in pyrrolo[2,1-c][1,4]benzothiazin...

Figure 2: Electrophilic centers in FPDs.

Scheme 6: Reaction of APBTT 1a with methanol (2a).

Scheme 7: Derivatization of PBTA 3aa.

Scheme 8: Reaction of APBTTs 1a–h with alcohols 2a–c. Isolated yields are given; reaction scale: a mixture of ...

Scheme 9: Side-reaction of APBTTs 1 with alcohols 2.

Scheme 10: Transformations of compounds 5 in solutions.

Scheme 11: Reaction of APBTT 1a with benzylamine.

Scheme 12: Derivatization of PBTA 7a.

Scheme 13: Reaction of APBTTs 1a–h and benzylamine. Isolated yields are given; reaction scale: a mixture of 1 ...

Scheme 14: Reaction of APBTT 1a with an excess of benzylamine.

Scheme 15: Reaction of APBTT 1a with morpholine.

Scheme 16: Reaction of APBTT 1a with aniline (11a).

Scheme 17: Derivatization of PBTA 12aa.

Scheme 18: Reaction of APBTTs 1a–h and arylamines 11a–d. Isolated yields are given; reaction scale: a mixture ...

Scheme 19: Side-reaction of APBTT 1a with arylamine 11b.

Scheme 20: Reaction of APBTT 1a with compounds 16a–d.

Scheme 21: Formation of compounds 17 as an undesired process during the synthesis of APBTTs 1.
Enolates ambushed – asymmetric tandem conjugate addition and subsequent enolate trapping with conventional and less traditional electrophiles
- Péter Kisszékelyi and
- Radovan Šebesta
Beilstein J. Org. Chem. 2023, 19, 593–634, doi:10.3762/bjoc.19.44
- derivatization is possible through the oxidation of the silyl motif to alcohol or the dehydration of the aldol adduct. Other tandem conjugate addition/enolate-trapping reactions In 2016, Nishiyama and co-workers have studied a three-component coupling reaction of alkynes, enones, and aldehydes via direct
- cyclization (Scheme 51A) [93]. The reaction starts with the coordination of the Rh catalyst to the propargyl alcohol 198. In the presence of a base, the rhodium–alkynyl reagent is generated with the concomitant extrusion of benzophenone. Finally, the alkynylation of the enone is followed by the cyclization

Graphical Abstract

Scheme 1: General scheme depicting tandem reactions based on an asymmetric conjugate addition followed by an ...

Scheme 2: Cu-catalyzed tandem conjugate addition of R2Zn/aldol reaction with chiral acetals.

Scheme 3: Cu-catalyzed asymmetric desymmetrization of cyclopentene-1,3-diones using a tandem conjugate additi...

Scheme 4: Stereocontrolled assembly of dialkylzincs, cyclic enones, and sulfinylimines utilizing a Cu-catalyz...

Scheme 5: Cu-catalyzed tandem conjugate addition/Mannich reaction (A). Access to chiral isoindolinones and tr...

Scheme 6: Cu-catalyzed tandem conjugate addition/nitro-Mannich reaction (A) with syn–anti or syn–syn selectiv...

Figure 1: Various chiral ligands utilized for the tandem conjugate addition/Michael reaction sequences.

Scheme 7: Cu-catalyzed tandem conjugate addition/Michael reaction: side-product formation with chalcone (A) a...

Scheme 8: Zn enolate trapping using allyl iodides (A), Stork–Jung vinylsilane reagents (B), and allyl bromide...

Scheme 9: Cu-catalyzed tandem conjugate addition/acylation through Li R2Zn enolate (A). A four-component coup...

Scheme 10: Selected examples for the Cu-catalyzed tandem conjugate addition/trifluoromethylthiolation sequence....

Scheme 11: Zn enolates trapped by vinyloxiranes: synthesis of allylic alcohols.

Scheme 12: Stereoselective cyclopropanation of Mg enolates formed by ACA of Grignard reagents to chlorocrotona...

Scheme 13: Domino aldol reactions of Mg enolates formed from coumarin and chromone.

Scheme 14: Oxidative coupling of ACA-produced Mg enolates.

Scheme 15: Tandem ACA of Grignard reagents to enones and Mannich reaction.

Scheme 16: Diastereodivergent Mannich reaction of Mg enolates with differently N-protected imines.

Scheme 17: Tandem Grignard–ACA–Mannich using Taddol-based phosphine-phosphite ligands.

Scheme 18: Tandem reaction of Mg enolates with aminomethylating reagents.

Scheme 19: Tandem reaction composed of Grignard ACA to alkynyl enones.

Scheme 20: Rh/Cu-catalyzed tandem reaction of diazo enoates leading to cyclobutanes.

Scheme 21: Tandem Grignard-ACA of cyclopentenones and alkylation of enolates.

Scheme 22: Tandem ACA of Grignard reagents followed by enolate trapping reaction with onium compounds.

Scheme 23: Mg enolates generated from unsaturated lactones in reaction with activated alkenes.

Scheme 24: Lewis acid mediated ACA to amides and SN2 cyclization of a Br-appended enolate.

Scheme 25: Trapping reactions of aza-enolates with Michael acceptors.

Scheme 26: Si enolates generated by TMSOTf-mediated ACA of Grignard reagents and enolate trapping reaction wit...

Scheme 27: Trapping reactions of enolates generated from alkenyl heterocycles (A) and carboxylic acids (B) wit...

Scheme 28: Reactions of heterocyclic Mg enolates with onium compounds.

Scheme 29: Synthetic transformations of cycloheptatrienyl and benzodithiolyl substituents.

Scheme 30: Aminomethylation of Al enolates generated by ACA of trialkylaluminum reagents.

Scheme 31: Trapping reactions of enolates with activated alkenes.

Scheme 32: Alkynylation of racemic aluminum or magnesium enolates.

Scheme 33: Trapping reactions of Zr enolates generated by Cu-ACA of organozirconium reagents.

Scheme 34: Chloromethylation of Zr enolates using the Vilsmeier–Haack reagent.

Scheme 35: Tandem conjugate borylation with subsequent protonation or enolate trapping by an electrophile.

Scheme 36: Tandem conjugate borylation/aldol reaction of cyclohexenones.

Scheme 37: Selected examples for the tandem asymmetric borylation/intramolecular aldol reaction; synthesis of ...

Scheme 38: Cu-catalyzed tandem methylborylation of α,β-unsaturated phosphine oxide in the presence of (R,Sp)-J...

Scheme 39: Cu-catalyzed tandem transannular conjugated borylation/aldol cyclization of macrocycles containing ...

Scheme 40: Stereoselective tandem conjugate borylation/Mannich cyclization: selected examples (A) and a multi-...

Scheme 41: Some examples of Cu-catalyzed asymmetric tandem borylation/aldol cyclization (A). Application to di...

Scheme 42: Atropisomeric P,N-ligands used in tandem conjugate borylation/aldol cyclization sequence.

Scheme 43: Selected examples for the enantioselective Cu-catalyzed borylation/intramolecular Michael addition ...

Scheme 44: Selected examples for the preparation of enantioenriched spiroindanes using a Cu-catalyzed tandem c...

Scheme 45: Enantioselective conjugate borylation of cyclobutene-1-carboxylic acid diphenylmethyl ester 175 wit...

Scheme 46: Cu-catalyzed enantioselective tandem conjugate silylation of α,β-unsaturated ketones with subsequen...

Scheme 47: Cu-catalyzed enantioselective tandem conjugate silylation of α,β-unsaturated ketones with subsequen...

Scheme 48: Cu-catalyzed tandem conjugate silylation/aldol condensation. The diastereoselectivity is controlled...

Scheme 49: Chiral Ru-catalyzed three-component coupling reaction.

Scheme 50: Rh-Phebox complex-catalyzed reductive cyclization and subsequent reaction with Michael acceptors th...

Scheme 51: Rh-catalyzed tandem asymmetric conjugate alkynylation/aldol reaction (A) and subsequent spiro-cycli...

Scheme 52: Rh-bod complex-catalyzed tandem asymmetric conjugate arylation/intramolecular aldol addition (A). S...

Scheme 53: Co-catalyzed C–H-bond activation/asymmetric conjugate addition/aldol reaction.

Scheme 54: (Diisopinocampheyl)borane-promoted 1,4-hydroboration of α,β-unsaturated morpholine carboxamides and...

Figure 2: Some examples of total syntheses that have been recently reviewed.

Scheme 55: Stereoselective synthesis of antimalarial prodrug (+)-artemisinin utilizing a tandem conjugate addi...

Scheme 56: Amphilectane and serrulatane diterpenoids: preparation of chiral starting material via asymmetric t...

Scheme 57: Various asymmetric syntheses of pleuromutilin and related compounds based on a tandem conjugate add...

Scheme 58: Total synthesis of glaucocalyxin A utilizing a tandem conjugate addition/acylation reaction sequenc...

Scheme 59: Installation of the exocyclic double bond using a tandem conjugate addition/aminomethylation sequen...

Scheme 60: Synthesis of the taxol core using a tandem conjugate addition/enolate trapping sequence with Vilsme...

Scheme 61: Synthesis of the tricyclic core of 12-epi-JBIR-23/24 utilizing a Rh-catalyzed asymmetric conjugate ...

Scheme 62: Total synthesis of (−)-peyssonoside A utilizing a Cu-catalyzed enantioselective tandem conjugate ad...
Transition-metal-catalyzed domino reactions of strained bicyclic alkenes
- Austin Pounder,
- Eric Neufeld,
- Peter Myler and
- William Tam
Beilstein J. Org. Chem. 2023, 19, 487–540, doi:10.3762/bjoc.19.38
- allylic alcohol isomerization in 137 resulting in the aldehyde 138. This aldehyde, in close proximity to the tertiary alcohol, leads to the production of the hemiacetal 139 which can finally undergo an oxidation producing the final bicyclo[2.2.2]lactone product 136. In 2011, the Radhakrishnan laboratory

Graphical Abstract

Figure 1: Ring-strain energies of homobicyclic and heterobicyclic alkenes in kcal mol−1. a) [2.2.1]-Bicyclic ...

Figure 2: a) Exo and endo face descriptions of bicyclic alkenes. b) Reactivity comparisons for different β-at...

Scheme 1: Ni-catalyzed ring-opening/cyclization cascade of heterobicyclic alkenes 1 with alkyl propiolates 2 ...

Scheme 2: Ni-catalyzed ring-opening/cyclization cascade of heterobicyclic alkenes 8 with β-iodo-(Z)-propenoat...

Scheme 3: Ni-catalyzed two- and three-component difunctionalizations of norbornene derivatives 15 with alkyne...

Scheme 4: Ni-catalyzed intermolecular three-component difunctionalization of oxabicyclic alkenes 1 with alkyn...

Scheme 5: Ni-catalyzed intermolecular three-component carboacylation of norbornene derivatives 15.

Scheme 6: Photoredox/Ni dual-catalyzed coupling of 4-alkyl-1,4-dihydropyridines 31 with heterobicyclic alkene...

Scheme 7: Photoredox/Ni dual-catalyzed coupling of α-amino radicals with heterobicyclic alkenes 30.

Scheme 8: Cu-catalyzed rearrangement/allylic alkylation of 2,3-diazabicyclo[2.2.1]heptenes 47 with Grignard r...

Scheme 9: Cu-catalyzed aminoboration of bicyclic alkenes 1 with bis(pinacolato)diboron (B2pin2) (53) and O-be...

Scheme 10: Cu-catalyzed borylalkynylation of oxabenzonorbornadiene (30b) with B2pin2 (53) and bromoalkynes 62.

Scheme 11: Cu-catalyzed borylacylation of bicyclic alkenes 1.

Scheme 12: Cu-catalyzed diastereoselective 1,2-difunctionalization of oxabenzonorbornadienes 30 for the synthe...

Scheme 13: Fe-catalyzed carbozincation of heterobicyclic alkenes 1 with arylzinc reagents 74.

Scheme 14: Co-catalyzed addition of arylzinc reagents of norbornene derivatives 15.

Scheme 15: Co-catalyzed ring-opening/dehydration of oxabicyclic alkenes 30 via C–H activation of arenes.

Scheme 16: Co-catalyzed [3 + 2] annulation/ring-opening/dehydration domino reaction of oxabicyclic alkenes 1 w...

Scheme 17: Co-catalyzed enantioselective carboamination of bicyclic alkenes 1 via C–H functionalization.

Scheme 18: Ru-catalyzed cyclization of oxabenzonorbornene derivatives with propargylic alcohols for the synthe...

Scheme 19: Ru-catalyzed coupling of oxabenzonorbornene derivatives 30 with propargylic alcohols and ethers 106...

Scheme 20: Ru-catalyzed ring-opening/dehydration of oxabicyclic alkenes via the C–H activation of anilides.

Scheme 21: Ru-catalyzed of azabenzonorbornadiene derivatives with arylamides.

Scheme 22: Rh-catalyzed cyclization of bicyclic alkenes with arylboronate esters 118.

Scheme 23: Rh-catalyzed cyclization of bicyclic alkenes with dienyl- and heteroaromatic boronate esters.

Scheme 24: Rh-catalyzed domino lactonization of doubly bridgehead-substituted oxabicyclic alkenes with seconda...

Scheme 25: Rh-catalyzed domino carboannulation of diazabicyclic alkenes with 2-cyanophenylboronic acid and 2-f...

Scheme 26: Rh-catalyzed synthesis of oxazolidinone scaffolds 147 through a domino ARO/cyclization of oxabicycl...

Scheme 27: Rh-catalyzed oxidative coupling of salicylaldehyde derivatives 151 with diazabicyclic alkenes 130a.

Scheme 28: Rh-catalyzed reaction of O-acetyl ketoximes with bicyclic alkenes for the synthesis of isoquinoline...

Scheme 29: Rh-catalyzed domino coupling reaction of 2-phenylpyridines 165 with oxa- and azabicyclic alkenes 30....

Scheme 30: Rh-catalyzed domino dehydrative naphthylation of oxabenzonorbornadienes 30 with N-sulfonyl 2-aminob...

Scheme 31: Rh-catalyzed domino dehydrative naphthylation of oxabenzonorbornadienes 30 with arylphosphine deriv...

Scheme 32: Rh-catalyzed domino ring-opening coupling reaction of azaspirotricyclic alkenes using arylboronic a...

Scheme 33: Tandem Rh(III)/Sc(III)-catalyzed domino reaction of oxabenzonorbornadienes 30 with alkynols 184 dir...

Scheme 34: Rh-catalyzed asymmetric domino cyclization and addition reaction of 1,6-enynes 194 and oxa/azabenzo...

Scheme 35: Rh/Zn-catalyzed domino ARO/cyclization of oxabenzonorbornadienes 30 with phosphorus ylides 201.

Scheme 36: Rh-catalyzed domino ring opening/lactonization of oxabenzonorbornadienes 30 with 2-nitrobenzenesulf...

Scheme 37: Rh-catalyzed domino C–C/C–N bond formation of azabenzonorbornadienes 30 with aryl-2H-indazoles 210.

Scheme 38: Rh/Pd-catalyzed domino synthesis of indole derivatives with 2-(phenylethynyl)anilines 212 and oxabe...

Scheme 39: Rh-catalyzed domino carborhodation of heterobicyclic alkenes 30 with B2pin2 (53).

Scheme 40: Rh-catalyzed three-component 1,2-carboamidation reaction of bicyclic alkenes 30 with aromatic and h...

Scheme 41: Pd-catalyzed diarylation and dialkenylation reactions of norbornene derivatives.

Scheme 42: Three-component Pd-catalyzed arylalkynylation reactions of bicyclic alkenes.

Scheme 43: Three-component Pd-catalyzed arylalkynylation reactions of norbornene and DFT mechanistic study.

Scheme 44: Pd-catalyzed three-component coupling N-tosylhydrazones 236, aryl halides 66, and norbornene (15a).

Scheme 45: Pd-catalyzed arylboration and allylboration of bicyclic alkenes.

Scheme 46: Pd-catalyzed, three-component annulation of aryl iodides 66, alkenyl bromides 241, and bicyclic alk...

Scheme 47: Pd-catalyzed double insertion/annulation reaction for synthesizing tetrasubstituted olefins.

Scheme 48: Pd-catalyzed aminocyclopropanation of bicyclic alkenes 1 with 5-iodopent-4-enylamine derivatives 249...

Scheme 49: Pd-catalyzed, three-component coupling of alkynyl bromides 62 and norbornene derivatives 15 with el...

Scheme 50: Pd-catalyzed intramolecular cyclization/ring-opening reaction of heterobicyclic alkenes 30 with 2-i...

Scheme 51: Pd-catalyzed dimer- and trimerization of oxabenzonorbornadiene derivatives 30 with anhydrides 268.

Scheme 52: Pd-catalyzed Catellani-type annulation and retro-Diels–Alder of norbornadiene 15b yielding fused xa...

Scheme 53: Pd-catalyzed hydroarylation and heteroannulation of urea-derived bicyclic alkenes 158 and aryl iodi...

Scheme 54: Access to fused 8-membered sulfoximine heterocycles 284/285 via Pd-catalyzed Catellani annulation c...

Scheme 55: Pd-catalyzed 2,2-bifunctionalization of bicyclic alkenes 1 generating spirobicyclic xanthone deriva...

Scheme 56: Pd-catalyzed Catellani-type annulation and retro-Diels–Alder of norbornadiene (15b) producing subst...

Scheme 57: Pd-catalyzed [2 + 2 + 1] annulation furnishing bicyclic-fused indanes 281 and 283.

Scheme 58: Pd-catalyzed ring-opening/ring-closing cascade of diazabicyclic alkenes 130a.

Scheme 59: Pd-NHC-catalyzed cyclopentannulation of diazabicyclic alkenes 130a.

Scheme 60: Pd-catalyzed annulation cascade generating diazabicyclic-fused indanones 292 and indanols 294.

Scheme 61: Pd-catalyzed skeletal rearrangement of spirotricyclic alkenes 176 towards large polycyclic benzofur...

Scheme 62: Pd-catalyzed oxidative annulation of aromatic enamides 298 and diazabicyclic alkenes 130a.

Scheme 63: Accessing 3,4,5-trisubstituted cyclopentenes 300, 301, 302 via the Pd-catalyzed domino reaction of ...

Scheme 64: Palladacycle-catalyzed ring-expansion/cyclization domino reactions of terminal alkynes and bicyclic...

Scheme 65: Pd-catalyzed carboesterification of norbornene (15a) with alkynes, furnishing α-methylene γ-lactone...






