Search results
Search for "furan" in Full Text gives 274 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Nanangenines: drimane sesquiterpenoids as the dominant metabolite cohort of a novel Australian fungus, Aspergillus nanangensis
- Heather J. Lacey,
- Cameron L. M. Gilchrist,
- Andrew Crombie,
- John A. Kalaitzis,
- Daniel Vuong,
- Peter J. Rutledge,
- Peter Turner,
- John I. Pitt,
- Ernest Lacey,
- Yit-Heng Chooi and
- Andrew M. Piggott
Beilstein J. Org. Chem. 2019, 15, 2631–2643, doi:10.3762/bjoc.15.256

- structures of 4 and 9 have one. The cyclohexane rings of all molecules have chair conformations, the cyclohexene rings of all molecules have half-chair conformations, and the furan rings of all molecules have flattened envelope conformations. See Supporting Information File 1 for detailed methods. Genomics

Graphical Abstract

Figure 1: Structures of nanangenines 1–10 isolated from A. nanangensis.

Figure 2: Putative nanangenine biosynthetic gene cluster in A. nanangensis MST-FP2251 and homologs identified...

Figure 3: Putative biosynthetic pathway to the nanangenines.
Combining the Ugi-azide multicomponent reaction and rhodium(III)-catalyzed annulation for the synthesis of tetrazole-isoquinolone/pyridone hybrids
- Gerardo M. Ojeda,
- Prabhat Ranjan,
- Pavel Fedoseev,
- Lisandra Amable,
- Upendra K. Sharma,
- Daniel G. Rivera and
- Erik V. Van der Eycken
Beilstein J. Org. Chem. 2019, 15, 2447–2457, doi:10.3762/bjoc.15.237
- isoquinolones is very wide and may be extended to the preparation of complex compounds having heterocyclic moieties such as pyridone, furan, thiophene and pyrrole, as well as the corresponding benzo-fused derivatives. The developed procedure is simple, reproducible and does not require inert conditions
- rings, such as furan, benzofuran, pyrrole, benzopyrrole, thiophene, benzothiophene and naphthalene were also successfully synthesized (5d–l) in moderate to very good yields. The exceptions were the cases of indolopyridone 5i and thiazolopyridone 5k, the latter one could not be obtained even after many

Graphical Abstract

Figure 1: Bioactive molecules containing a tetrazole, pyridone or isoquinolone ring.

Scheme 1: Approaches for the synthesis of tetrazoles and isoquinolones and their interplay as designed in thi...

Scheme 2: Scope of the Ugi-azide-4CR/deprotection/acylation sequence. Ugi-azide-4CR conducted at the 2.0 mmol...

Scheme 3: Influence of substituents R and R2 on the reaction outcome. For compounds 4k–m the overall yield in...

Scheme 4: Influence of the alkyne and R1 substituent on the reaction outcome.

Scheme 5: Scope of acrylic, heterocyclic and ring-fused N-acylaminomethyl tetrazole substrates.

Scheme 6: Proposed reaction mechanism using substrates 1a and 3a.
N-(1-Phenylethyl)aziridine-2-carboxylate esters in the synthesis of biologically relevant compounds
- Iwona E. Głowacka,
- Aleksandra Trocha,
- Andrzej E. Wróblewski and
- Dorota G. Piotrowska
Beilstein J. Org. Chem. 2019, 15, 1722–1757, doi:10.3762/bjoc.15.168

- lithium enolate of methyl 4-methoxyphenylacetate would give the β-hydroxyester 169 which when treated with Lewis acid experienced the aziridine ring cleavage with simultaneous dihydrofuran-2-one ring closure to produce 170 contaminated with small amounts of the corresponding furan-2(5H)-one 171

Graphical Abstract

Figure 1: Examples of three-carbon chirons.

Figure 2: Structures of derivatives of N-(1-phenylethyl)aziridine-2-carboxylic acid 5–8.

Figure 3: Synthetic equivalency of aziridine aldehydes 6.

Scheme 1: Synthesis of N-(1-phenylethyl)aziridine-2-carboxylates 5. Reagents and conditions: a) TEA, toluene,...

Scheme 2: Absolute configuration at C2 in (2S,1'S)-5a. Reagents and conditions: a) 20% HClO4, 80 °C, 30 h the...

Scheme 3: Major synthetic strategies for a 2-ketoaziridine scaffold [R* = (R)- or (S)-1-phenylethyl; R′ = Alk...

Scheme 4: Synthesis of cyanide (2S,1'S)-13. Reagents and conditions: a) NH3, EtOH/H2O, rt, 72 h; b) Ph3P, CCl4...

Scheme 5: Synthesis of key intermediates (R)-16 and (R)-17 for (R,R)-formoterol (14) and (R)-tamsulosin (15)....

Scheme 6: Synthesis of mitotic kinesin inhibitors (2R/S,1'R)-23. Reagents and conditions: a) H2, Pd(OH)2, EtO...

Scheme 7: Synthesis of (R)-mexiletine ((R)-24). Reagents and conditions: a) TsCl, TEA, DMAP, CH2Cl2, rt, 1 h;...

Scheme 8: Synthesis of (−)-cathinone ((S)-27). Reagents and conditions: a) PhMgBr, ether, 0 °C; b) H2, 10% Pd...

Scheme 9: Synthesis of N-Boc-norpseudoephedrine ((1S,2S)-(+)-29) and N-Boc-norephedrine ((1R,2S)-29). Reagent...

Scheme 10: Synthesis of (−)-ephedrine ((1R,2S)-31). Reagents and conditions: a) TfOMe, MeCN then NaBH3CN, rt; ...

Scheme 11: Synthesis of xestoaminol C ((2S,3R)-35), 3-epi-xestoaminol C ((2S,3S)-35) and N-Boc-spisulosine ((2S...

Scheme 12: Synthesis of ʟ-tryptophanol ((S)-41). Reagents and conditions: a) CDI, MeCN, rt, 1 h then TMSI, MeC...

Scheme 13: Synthesis of ʟ-homophenylalaninol ((S)-42). Reagents and conditions: a) NaH, THF, 0 °C to −78 °C, 1...

Scheme 14: Synthesis of ᴅ-homo(4-octylphenyl)alaninol ((R)-47) and a sphingolipid analogue (R)-48. Reagents an...

Scheme 15: Synthesis of florfenicol ((1R,2S)-49). Reagents and conditions: a) (S)-1-phenylethylamine, TEA, MeO...

Scheme 16: Synthesis of natural tyroscherin ((2S,3R,6E,8R,10R)-55). Reagents and conditions: a) I(CH2)3OTIPS, t...

Scheme 17: Syntheses of (−)-hygrine (S)-61, (−)-hygroline (2S,2'S)-62 and (−)-pseudohygroline (2S,2'R)-62. Rea...

Scheme 18: Synthesis of pyrrolidine (3S,3'R)-68, a fragment of the fluoroquinolone antibiotic PF-00951966. Rea...

Scheme 19: Synthesis of sphingolipid analogues (R)-76. Reagents and conditions: a) BnBr, Mg, THF, reflux, 6 h;...

Scheme 20: Synthesis of ᴅ-threo-PDMP (1R,2R)-81. Reagents and conditions: a) TMSCl, NaI, MeCN, rt, 1 h 50 min,...

Scheme 21: Synthesis of the sphingolipid analogue SG-14 (2S,3S)-84. Reagents and conditions: a) LiAlH4, THF, 0...

Scheme 22: Synthesis of the sphingolipid analogue SG-12 (2S,3R)-88. Reagents and conditions: a) 1-(bromomethyl...

Scheme 23: Synthesis of sphingosine-1-phosphate analogues DS-SG-44 and DS-SG-45 (2S,3R)-89a and (2S,3R)-89a. R...

Scheme 24: Synthesis of N-Boc-safingol ((2S,3S)-95) and N-Boc-ᴅ-erythro-sphinganine ((2S,3R)-95). Reagents and...

Scheme 25: Synthesis of ceramide analogues (2S,3R)-96. Reagents and conditions: a) NaBH4, ZnCl2, MeOH, −78 °C,...

Scheme 26: Synthesis of orthogonally protected serinols, (S)-101 and (R)-102. Reagents and conditions: a) BnBr...

Scheme 27: Synthesis of N-acetyl-3-phenylserinol ((1R,2R)-105). Reagents and conditions: a) AcOH, CH2Cl2, refl...

Scheme 28: Synthesis of (S)-linezolid (S)-107. Reagents and conditions: a) LiAlH4, THF, 0 °C to reflux; b) Boc2...

Scheme 29: Synthesis of (2S,3S,4R)-2-aminooctadecane-1,3,4-triol (ᴅ-ribo-phytosphingosine) (2S,3S,4R)-110. Rea...

Scheme 30: Syntheses of ᴅ-phenylalanine (R)-116. Reagents and conditions: a) AcOH, CH2Cl2, reflux, 4 h; b) MsC...

Scheme 31: Synthesis of N-Boc-ᴅ-3,3-diphenylalanine ((R)-122). Reagents and conditions: a) PhMgBr, THF, −78 °C...

Scheme 32: Synthesis of ethyl N,N’-di-Boc-ʟ-2,3-diaminopropanoate ((S)-125). Reagents and conditions: a) NaN3,...

Scheme 33: Synthesis of the bicyclic amino acid (S)-(+)-127. Reagents and conditions: a) BF3·OEt2, THF, 60 °C,...

Scheme 34: Synthesis of lacosamide, (R)-2-acetamido-N-benzyl-3-methoxypropanamide (R)-130. Reagents and condit...

Scheme 35: Synthesis of N-Boc-norfuranomycin ((2S,2'R)-133). Reagents and conditions: a) H2C=CHCH2I, NaH, THF,...

Scheme 36: Synthesis of MeBmt (2S,3R,4R,6E)-139. Reagents and conditions: a) diisopropyl (S,S)-tartrate (E)-cr...

Scheme 37: Synthesis of (+)-polyoxamic acid (2S,3S,4S)-144. Reagents and conditions: a) AD-mix-α, MeSO2NH2, t-...

Scheme 38: Synthesis of the protected 3-hydroxy-ʟ-glutamic acid (2S,3R)-148. Reagents and conditions: a) LiHMD...

Scheme 39: Synthesis of (+)-isoserine (R)-152. Reagents and conditions: a) AcCl, MeCN, rt, 0.5 h then Na2CO3, ...

Scheme 40: Synthesis of (3R,4S)-N3-Boc-3,4-diaminopentanoic acid (3R,4S)-155. Reagents and conditions: a) Ph3P...

Scheme 41: Synthesis of methyl (2S,3S,4S)-4-(dimethylamino)-2,3-dihydroxy-5-methoxypentanoate (2S,3S,4S)-159. ...

Scheme 42: Syntheses of methyl (3S,4S) 4,5-di-N-Boc-amino-3-hydroxypentanoate ((3S,4S)-164), methyl (3S,4S)-4-N...

Scheme 43: Syntheses of (3R,5S)-5-(aminomethyl)-3-(4-methoxyphenyl)dihydrofuran-2(3H)-one ((3R,5S)-168). Reage...

Scheme 44: Syntheses of a series of imidazolin-2-one dipeptides 175–177 (for R' and R'' see text). Reagents an...

Scheme 45: Syntheses of (2S,3S)-N-Boc-3-hydroxy-2-hydroxymethylpyrrolidine ((2S,3S)-179). Reagents and conditi...

Scheme 46: Syntheses of enantiomers of 1,4-dideoxy-1,4-imino-ʟ- and -ᴅ-lyxitols (2S,3R,4S)-182 and (2R,3S,4R)-...

Scheme 47: Synthesis of 1,4-dideoxy-1,4-imino-ʟ-ribitol (2S,3S,4R)-182. Reagents and conditions: a) AcOH, CH2Cl...

Scheme 48: Syntheses of 1,4-dideoxy-1,4-imino-ᴅ-arabinitol (2R,3R,4R)-182 and 1,4-dideoxy-1,4-imino-ᴅ-xylitol ...

Scheme 49: Syntheses of natural 2,5-imino-2,5,6-trideoxy-ʟ-gulo-heptitol ((2S,3R,4R,5R)-184) and its C4 epimer...

Scheme 50: Syntheses of (−)-dihydropinidine ((2S,6R)-187a) (R = C3H7) and (2S,6R)-isosolenopsins (2S,6R)-187b ...

Scheme 51: Syntheses of (+)-deoxocassine ((2S,3S,6R)-190a, R = C12H25) and (+)-spectaline ((2S,3S,6R)-190b, R ...

Scheme 52: Synthesis of (−)-microgrewiapine A ((2S,3R,6S)-194a) and (+)-microcosamine A ((2S,3R,6S)-194b). Rea...

Scheme 53: Syntheses of ʟ-1-deoxynojirimycin ((2S,3S,4S,5R)-200), ʟ-1-deoxymannojirimycin ((2S,3S,4S,5S)-200) ...

Scheme 54: Syntheses of 1-deoxy-ᴅ-galacto-homonojirimycin (2R,3S,4R,5S)-211. Reagents and conditions: a) MeONH...

Scheme 55: Syntheses of 7a-epi-hyacinthacine A1 (1S,2R,3R,7aS)-220. Reagents and conditions: a) TfOTBDMS, 2,6-...

Scheme 56: Syntheses of 8-deoxyhyacinthacine A1 ((1S,2R,3R,7aR)-221). Reagents and conditions: a) H2, Pd/C, PT...

Scheme 57: Syntheses of (+)-lentiginosine ((1S,2S,8aS)-227). Reagents and conditions: a) (EtO)2P(O)CH2COOEt, L...

Scheme 58: Syntheses of 8-epi-swainsonine (1S,2R,8S,8aR)-231. Reagents and conditions: a) Ph3P=CHCOOMe, MeOH, ...

Scheme 59: Synthesis of a protected vinylpiperidine (2S,3R)-237, a key intermediate in the synthesis of (−)-sw...

Scheme 60: Synthesis of a modified carbapenem 245. Reagents and conditions: a) AcOEt, LiHMDS, THF, −78 °C, 1.5...
Recent advances on the transition-metal-catalyzed synthesis of imidazopyridines: an updated coverage
- Gagandeep Kour Reen,
- Ashok Kumar and
- Pratibha Sharma
Beilstein J. Org. Chem. 2019, 15, 1612–1704, doi:10.3762/bjoc.15.165


Graphical Abstract

Figure 1: Various drugs having IP nucleus.

Figure 2: Participation percentage of various TMs for the syntheses of IPs.

Scheme 1: CuI–NaHSO4·SiO2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 2: Experimental examination of reaction conditions.

Scheme 3: One-pot tandem reaction for the synthesis of 2-haloimidazopyridines.

Scheme 4: Mechanistic scheme for the synthesis of 2-haloimidazopyridine.

Scheme 5: Copper-MOF-catalyzed three-component reaction (3-CR) for imidazo[1,2-a]pyridines.

Scheme 6: Mechanism for copper-MOF-driven synthesis.

Scheme 7: Heterogeneous synthesis via titania-supported CuCl2.

Scheme 8: Mechanism involving oxidative C–H functionalization.

Scheme 9: Heterogeneous synthesis of IPs.

Scheme 10: One-pot regiospecific synthesis of imidazo[1,2-a]pyridines.

Scheme 11: Vinyl azide as an unprecedented substrate for imidazo[1,2-a]pyridines.

Scheme 12: Radical pathway.

Scheme 13: Cu(I)-catalyzed transannulation approach for imidazo[1,5-a]pyridines.

Scheme 14: Plausible radical pathway for the synthesis of imidazo[1,5-a]pyridines.

Scheme 15: A solvent-free domino reaction for imidazo[1,2-a]pyridines.

Scheme 16: Cu-NPs-mediated synthesis of imidazo[1,2-a]pyridines.

Scheme 17: CuI-catalyzed synthesis of isoxazolylimidazo[1,2-a]pyridines.

Scheme 18: Functionalization of 4-bromo derivative via Sonogashira coupling reaction.

Scheme 19: A plausible reaction pathway.

Scheme 20: Cu(I)-catalyzed intramolecular oxidative C–H amidation reaction.

Scheme 21: One-pot synthetic reaction for imidazo[1,2-a]pyridine.

Scheme 22: Plausible reaction mechanism.

Scheme 23: Cu(OAc)2-promoted synthesis of imidazo[1,2-a]pyridines.

Scheme 24: Mechanism for aminomethylation/cycloisomerization of propiolates with imines.

Scheme 25: Three-component synthesis of imidazo[1,2-a]pyridines.

Figure 3: Scope of pyridin-2(1H)-ones and acetophenones.

Scheme 26: CuO NPS-promoted A3 coupling reaction.

Scheme 27: Cu(II)-catalyzed C–N bond formation reaction.

Scheme 28: Mechanism involving Chan–Lam/Ullmann coupling.

Scheme 29: Synthesis of formyl-substituted imidazo[1,2-a]pyridines.

Scheme 30: A tandem sp3 C–H amination reaction.

Scheme 31: Probable mechanistic approach.

Scheme 32: Dual catalytic system for imidazo[1,2-a]pyridines.

Scheme 33: Tentative mechanism.

Scheme 34: CuO/CuAl2O4/ᴅ-glucose-promoted 3-CCR.

Scheme 35: A tandem CuOx/OMS-2-based synthetic strategy.

Figure 4: Biomimetic catalytic oxidation in the presence of electron-transfer mediators (ETMs).

Scheme 36: Control experiment.

Scheme 37: Copper-catalyzed C(sp3)–H aminatin reaction.

Scheme 38: Reaction of secondary amines.

Scheme 39: Probable mechanistic pathway.

Scheme 40: Coupling reaction of α-azidoketones.

Scheme 41: Probable pathway.

Scheme 42: Probable mechanism with free energy calculations.

Scheme 43: MCR for cyanated IP synthesis.

Scheme 44: Substrate scope for the reaction.

Scheme 45: Reaction mechanism.

Scheme 46: Probable mechanistic pathway for Cu/ZnAl2O4-catalyzed reaction.

Scheme 47: Copper-catalyzed double oxidative C–H amination reaction.

Scheme 48: Application towards different coupling reactions.

Scheme 49: Reaction mechanism.

Scheme 50: Condensation–cyclization approach for the synthesis of 1,3-diarylated imidazo[1,5-a]pyridines.

Scheme 51: Optimized reaction conditions.

Scheme 52: One-pot 2-CR.

Scheme 53: One-pot 3-CR without the isolation of chalcone.

Scheme 54: Copper–Pybox-catalyzed cyclization reaction.

Scheme 55: Mechanistic pathway catalyzed by Cu–Pybox complex.

Scheme 56: Cu(II)-promoted C(sp3)-H amination reaction.

Scheme 57: Wider substrate applicability for the reaction.

Scheme 58: Plausible reaction mechanism.

Scheme 59: CuI assisted C–N cross-coupling reaction.

Scheme 60: Probable reaction mechanism involving sp3 C–H amination.

Scheme 61: One-pot MCR-catalyzed by CoFe2O4/CNT-Cu.

Scheme 62: Mechanistic pathway.

Scheme 63: Synthetic scheme for 3-nitroimidazo[1,2-a]pyridines.

Scheme 64: Plausible mechanism for CuBr-catalyzed reaction.

Scheme 65: Regioselective synthesis of halo-substituted imidazo[1,2-a]pyridines.

Scheme 66: Synthesis of 2-phenylimidazo[1,2-a]pyridines.

Scheme 67: Synthesis of diarylated compounds.

Scheme 68: CuBr2-mediated one-pot two-component oxidative coupling reaction.

Scheme 69: Decarboxylative cyclization route to synthesize 1,3-diarylimidazo[1,5-a]pyridines.

Scheme 70: Mechanistic pathway.

Scheme 71: C–H functionalization reaction of enamines to produce diversified heterocycles.

Scheme 72: A plausible mechanism.

Scheme 73: CuI-promoted aerobic oxidative cyclization reaction of ketoxime acetates and pyridines.

Scheme 74: CuI-catalyzed pathway for the formation of imidazo[1,2-a]pyridine.

Scheme 75: Mechanistic pathway.

Scheme 76: Mechanistic rationale for the synthesis of products.

Scheme 77: Copper-catalyzed synthesis of vinyloxy-IP.

Scheme 78: Regioselective product formation with propiolates.

Scheme 79: Proposed mechanism for vinyloxy-IP formation.

Scheme 80: Regioselective synthesis of 3-hetero-substituted imidazo[1,2-a]pyridines with different reaction su...

Scheme 81: Mechanistic pathway.

Scheme 82: CuI-mediated synthesis of 3-formylimidazo[1,2-a]pyridines.

Scheme 83: Radical pathway for 3-formylated IP synthesis.

Scheme 84: Pd-catalyzed urea-cyclization reaction for IPs.

Scheme 85: Pd-catalyzed one-pot-tandem amination and intramolecular amidation reaction.

Figure 5: Scope of aniline nucleophiles.

Scheme 86: Pd–Cu-catalyzed Sonogashira coupling reaction.

Scheme 87: One-pot amide coupling reaction for the synthesis of imidazo[4,5-b]pyridines.

Scheme 88: Urea cyclization reaction for the synthesis of two series of pyridines.

Scheme 89: Amidation reaction for the synthesis of imidazo[4,5-b]pyridines.

Figure 6: Amide scope.

Scheme 90: Pd NPs-catalyzed 3-component reaction for the synthesis of 2,3-diarylated IPs.

Scheme 91: Plausible mechanistic pathway for Pd NPs-catalyzed MCR.

Scheme 92: Synthesis of chromenoannulated imidazo[1,2-a]pyridines.

Scheme 93: Mechanism for the synthesis of chromeno-annulated IPs.

Scheme 94: Zinc oxide NRs-catalyzed synthesis of imidazo[1,2-a]azines/diazines.

Scheme 95: Zinc oxide-catalyzed isocyanide based GBB reaction.

Scheme 96: Reaction pathway for ZnO-catalyzed GBB reaction.

Scheme 97: Mechanistic pathway.

Scheme 98: ZnO NRs-catalyzed MCR for the synthesis of imidazo[1,2-a]azines.

Scheme 99: Ugi type GBB three-component reaction.

Scheme 100: Magnetic NPs-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 101: Regioselective synthesis of 2-alkoxyimidazo[1,2-a]pyridines catalyzed by Fe-SBA-15.

Scheme 102: Plausible mechanistic pathway for the synthesis of 2-alkoxyimidazopyridine.

Scheme 103: Iron-catalyzed synthetic approach.

Scheme 104: Iron-catalyzed aminooxygenation reaction.

Scheme 105: Mechanistic pathway.

Scheme 106: Rh(III)-catalyzed double C–H activation of 2-substituted imidazoles and alkynes.

Scheme 107: Plausible reaction mechanism.

Scheme 108: Rh(III)-catalyzed non-aromatic C(sp2)–H bond activation–functionalization for the synthesis of imid...

Scheme 109: Reactivity and selectivity of different substrates.

Scheme 110: Rh-catalyzed direct C–H alkynylation by Li et al.

Scheme 111: Suggested radical mechanism.

Scheme 112: Scandium(III)triflate-catalyzed one-pot reaction and its mechanism for the synthesis of benzimidazo...

Scheme 113: RuCl3-assisted Ugi-type Groebke–Blackburn condensation reaction.

Scheme 114: C-3 aroylation via Ru-catalyzed two-component reaction.

Scheme 115: Regioselective synthetic mechanism.

Scheme 116: La(III)-catalyzed one-pot GBB reaction.

Scheme 117: Mechanistic approach for the synthesis of imidazo[1,2-a]pyridines.

Scheme 118: Synthesis of imidazo[1,2-a]pyridine using LaMnO3 NPs under neat conditions.

Scheme 119: Mechanistic approach.

Scheme 120: One-pot 3-CR for regioselective synthesis of 2-alkoxy-3-arylimidazo[1,2-a]pyridines.

Scheme 121: Formation of two possible products under optimization of the catalysts.

Scheme 122: Mechanistic strategy for NiFe2O4-catalyzed reaction.

Scheme 123: Two-component reaction for synthesizing imidazodipyridiniums.

Scheme 124: Mechanistic scheme for the synthesis of imidazodipyridiniums.

Scheme 125: CuI-catalyzed arylation of imidazo[1,2-a]pyridines.

Scheme 126: Mechanism for arylation reaction.

Scheme 127: Cupric acetate-catalyzed double carbonylation approach.

Scheme 128: Radical mechanism for double carbonylation of IP.

Scheme 129: C–S bond formation reaction catalyzed by cupric acetate.

Scheme 130: Cupric acetate-catalyzed C-3 formylation approach.

Scheme 131: Control experiments for signifying the role of DMSO and oxygen.

Scheme 132: Mechanism pathway.

Scheme 133: Copper bromide-catalyzed CDC reaction.

Scheme 134: Extension of the substrate scope.

Scheme 135: Plausible radical pathway.

Scheme 136: Transannulation reaction for the synthesis of imidazo[1,5-a]pyridines.

Scheme 137: Plausible reaction pathway for denitrogenative transannulation.

Scheme 138: Cupric acetate-catalyzed C-3 carbonylation reaction.

Scheme 139: Plausible mechanism for regioselective C-3 carbonylation.

Scheme 140: Alkynylation reaction at C-2 of 3H-imidazo[4,5-b]pyridines.

Scheme 141: Two-way mechanism for C-2 alkynylation of 3H-imidazo[4,5-b]pyridines.

Scheme 142: Palladium-catalyzed SCCR approach.

Scheme 143: Palladium-catalyzed Suzuki coupling reaction.

Scheme 144: Reaction mechanism.

Scheme 145: A phosphine free palladium-catalyzed synthesis of C-3 arylated imidazopyridines.

Scheme 146: Palladium-mediated Buchwald–Hartwig cross-coupling reaction.

Figure 7: Structure of the ligands optimized.

Scheme 147: Palladium acetate-catalyzed direct arylation of imidazo[1,2-a]pyridines.

Scheme 148: Palladium acetate-catalyzed mechanistic pathway.

Scheme 149: Palladium acetate-catalyzed regioselective arylation reported by Liu and Zhan.

Scheme 150: Mechanism for selective C-3 arylation of IP.

Scheme 151: Pd(II)-catalyzed alkenylation reaction with styrenes.

Scheme 152: Pd(II)-catalyzed alkenylation reaction with acrylates.

Scheme 153: A two way mechanism.

Scheme 154: Double C–H activation reaction catalyzed by Pd(OAc)2.

Scheme 155: Probable mechanism.

Scheme 156: Palladium-catalyzed decarboxylative coupling.

Scheme 157: Mechanistic cycle for decarboxylative arylation reaction.

Scheme 158: Ligand-free approach for arylation of imidazo[1,2-a]pyridine-3-carboxylic acids.

Scheme 159: Mechanism for ligandless arylation reaction.

Scheme 160: NHC-Pd(II) complex assisted arylation reaction.

Scheme 161: C-3 arylation of imidazo[1,2-a]pyridines with aryl bromides catalyzed by Pd(OAc)2.

Scheme 162: Pd(II)-catalyzed C-3 arylations with aryl tosylates and mesylates.

Scheme 163: CDC reaction for the synthesis of imidazo[1,2-a]pyridines.

Scheme 164: Plausible reaction mechanism for Pd(OAc)2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 165: Pd-catalyzed C–H amination reaction.

Scheme 166: Mechanism for C–H amination reaction.

Scheme 167: One-pot synthesis for 3,6-di- or 2,3,6-tri(hetero)arylimidazo[1,2-a]pyridines.

Scheme 168: C–H/C–H cross-coupling reaction of IPs and azoles catalyzed by Pd(II).

Scheme 169: Mechanistic cycle.

Scheme 170: Rh-catalyzed C–H arylation reaction.

Scheme 171: Mechanistic pathway for C–H arylation of imidazo[1,2-a]pyridine.

Scheme 172: Rh(III)-catalyzed double C–H activation of 2-phenylimidazo[1,2-a]pyridines and alkynes.

Scheme 173: Rh(III)-catalyzed mechanistic pathway.

Scheme 174: Rh(III)-mediated oxidative coupling reaction.

Scheme 175: Reactions showing functionalization of the product obtained by the group of Kotla.

Scheme 176: Mechanism for Rh(III)-catalyzed oxidative coupling reaction.

Scheme 177: Rh(III)-catalyzed C–H activation reaction.

Scheme 178: Mechanistic cycle.

Scheme 179: Annulation reactions of 2-arylimidazo[1,2-a]pyridines and alkynes.

Scheme 180: Two-way reaction mechanism for annulations reaction.

Scheme 181: [RuCl2(p-cymene)]2-catalyzed C–C bond formation reaction.

Scheme 182: Reported reaction mechanism.

Scheme 183: Fe(III) catalyzed C-3 formylation approach.

Scheme 184: SET mechanism-catalyzed by Fe(III).

Scheme 185: Ni(dpp)Cl2-catalyzed KTC coupling.

Scheme 186: Pd-catalyzed SM coupling.

Scheme 187: Vanadium-catalyzed coupling of IP and NMO.

Scheme 188: Mechanistic cycle.

Scheme 189: Selective C3/C5–H bond functionalizations by mono and bimetallic systems.

Scheme 190: rGO-Ni@Pd-catalyzed C–H bond arylation of imidazo[1,2-a]pyridine.

Scheme 191: Mechanistic pathway for heterogeneously catalyzed arylation reaction.

Scheme 192: Zinc triflate-catalyzed coupling reaction of substituted propargyl alcohols.
Enantioselective PCCP Brønsted acid-catalyzed aza-Piancatelli rearrangement
- Gabrielle R. Hammersley,
- Meghan F. Nichol,
- Helena C. Steffens,
- Jose M. Delgado,
- Gesine K. Veits and
- Javier Read de Alaniz
Beilstein J. Org. Chem. 2019, 15, 1569–1574, doi:10.3762/bjoc.15.160
- furan ring required to initiate the cascade sequence. This highlights that a balance between nucleophilicity and hydrogen bond capabilities are required to obtain an efficient and selective reaction. Compared to the rearrangement of furylcarbinol 1a, sterically bulky aryl groups attached to the

Graphical Abstract

Figure 1: Proposed mechanism of the asymmetric aza-Piancatelli reaction.

Scheme 1: Asymmetric aza-Piancatelli rearrangement with a range of substituted anilines. *To simplify the pur...

Scheme 2: Asymmetric aza-Piancatelli rearrangement with a range of substituted furylcarbinols. *To simplify t...
Ugi reaction-derived prolyl peptide catalysts grafted on the renewable polymer polyfurfuryl alcohol for applications in heterogeneous enamine catalysis
- Alexander F. de la Torre,
- Gabriel S. Scatena,
- Oscar Valdés,
- Daniel G. Rivera and
- Márcio W. Paixão
Beilstein J. Org. Chem. 2019, 15, 1210–1216, doi:10.3762/bjoc.15.118
- Abstract The multicomponent synthesis of prolyl pseudo-peptide catalysts using the Ugi reaction with furfurylamines or isocyanides is described. The incorporation of such a polymerizable furan handle enabled the subsequent polymerization of the peptide catalyst with furfuryl alcohol, thus rendering
- synthesis of prolyl pseudo-peptides having a furan ring handle, which could be subsequently incorporated into PFA during the polymerization process. Results and Discussion To this end, a solution-phase multicomponent procedure based on the Ugi four-component reaction (Ugi-4CR) [14], was employed to
- incorporate proline [15] and the furan functionality into pseudo-peptide catalysts. As shown in Scheme 2, Boc-L-proline and acetone were employed as acid and oxo components, respectively, in combination either with furfurylamine and cyclohexyl isocyanide or with (S)-α-methylbenzylamine and furfuryl isocyanide

Graphical Abstract

Scheme 1: Schematic synthesis of polyfurfulyl alcohol (PFA) incorporating a prolyl peptide catalyst. AA: Amin...

Scheme 2: Utilization of the Ugi four-component reaction (Ugi-4CR) for the synthesis of prolyl pseudo-peptide...

Figure 1: Analysis of the continuous-flow catalytic system producing γ-nitroaldehyde 5 with PFA-supported cat...
Switchable selectivity in Pd-catalyzed [3 + 2] annulations of γ-oxy-2-cycloalkenones with 3-oxoglutarates: C–C/C–C vs C–C/O–C bond formation
- Yang Liu,
- Julie Oble and
- Giovanni Poli
Beilstein J. Org. Chem. 2019, 15, 1107–1115, doi:10.3762/bjoc.15.107
- combination may allow direct access to either fused bicyclic cyclopentanic (pentalene-, indene-, or azulene-type) structures via a γ-C-allylation/β-C-1,4 addition process, or annulated furan-based motifs through a γ-C-allylation/β-O-1,4 addition process, both motifs being incorporated into biologically
- or C–C/O–C annulation at will, thus providing an easy access to annulated cyclopentanic structures III or annulated furan-based motifs IV, respectively. Additionally, in the case of C–C/C–C bond forming annulations, the products III can undergo two decarboxylation steps leading to bis-cycloalkanone
- annulated cylopentanic (top) and furan-based (bottom) substructures in natural products. Previously developed bis-nucleophile/bis-electrophile [3 + 2] annulations. Concept: [3 + 2] C–C/C–C vs C–C/O–C bond-forming annulations. C–C/O–C bond forming annulations with dimethyl 3-oxoglutarate (1a). C–C/C–C bond

Graphical Abstract

Scheme 1: Previously developed bis-nucleophile/bis-electrophile [3 + 2] annulations.

Scheme 2: Concept: [3 + 2] C–C/C–C vs C–C/O–C bond-forming annulations.

Figure 1: Examples of annulated cylopentanic (top) and furan-based (bottom) substructures in natural products....

Scheme 3: C–C/O–C bond forming annulations with dimethyl 3-oxoglutarate (1a).

Scheme 4: C–C/C–C bond-forming annulations with dimethyl 3-oxoglutarate (1a).

Scheme 5: C–C/C–O bond-forming annulations with various bis-nucleophiles.

Scheme 6: Decarboxylative rearrangement of 4a into 5a.

Scheme 7: Proposed mechanism for the Pd-catalyzed part of the [3 + 2] annulation reaction.

Scheme 8: Proposed mechanism for the temperature dependent cyclization part of the [3 + 2] annulation.
Efficient synthesis of 4-substituted-ortho-phthalaldehyde analogues: toward the emergence of new building blocks
- Clémence Moitessier,
- Ahmad Rifai,
- Pierre-Edouard Danjou,
- Isabelle Mallard and
- Francine Cazier-Dennin
Beilstein J. Org. Chem. 2019, 15, 721–726, doi:10.3762/bjoc.15.67
- -dihydroisobenzofuran-5-ol intermediate has been tested and oxidation parameters of the dedicated structures were optimized. Finally, a rapid deprotection step was initiated to afford 4-MeO-OPA and 4-HO-OPA with 51% overall yield. Results and Discussion Firstly, 2-((prop-2-ynyloxy)methyl)furan (2) was synthesized
- . Synthetic procedures 2-((Prop-2-ynyloxy)methyl)furan (2). Adapted from reference [17]: Furfuryl alcohol (9.0 mL, 103.8 mmol) was added dropwise to a solution of NaH (5.0 g, 207.5 mmol) in DMF (100 mL) at 0 °C. After 90 minutes, propargyl bromide (12.7 mL, 114.2 mmol) was added dropwise and the resulting

Graphical Abstract

Scheme 1: Synthesis of 4,5-dihydroisobenzofuran-5-ol (3).

Scheme 2: Protection strategy of 4,5-dihydroisobenzofuran-5-ol (3).

Scheme 3: Oxidation of 5-substituted-4,5-dihydroisobenzofuran-5-ol in presence of SeO2 or DDQ.

Scheme 4: Synthesis of 4-hydroxy-ortho-phthalaldehyde (6) through MAOS demethylation of 4-methoxy-ortho-phtha...
Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction
- Vaida Milišiūnaitė,
- Rūta Paulavičiūtė,
- Eglė Arbačiauskienė,
- Vytas Martynaitis,
- Wolfgang Holzer and
- Algirdas Šačkus
Beilstein J. Org. Chem. 2019, 15, 679–684, doi:10.3762/bjoc.15.62
- -c]pyrazoles, where both the fused pyrazole and furan moieties are aromatic, are still quite limited, and their chemistry and functional properties remain largely underexplored. Huang et al. have reported the synthesis and the evaluation of antiplatelet and anti-allergic activities of 2,3,4
- alkoxide [11]. Notably, the 2H-furo[2,3-c]pyrazole ring system is structurally similar to the benzo[b]furan system, which is a privileged motif in natural products and biologically active compounds [13][14]. As a result, numerous strategies have been developed for the construction of benzo[b]furan and its
- alkynylation/cyclization reaction between 2-iodophenol and (triethoxysilyl)alkynes [20]. In recent years silver and gold salts have found application as versatile and mild catalysts to access the benzo[b]furan ring system through intramolecular cyclization of 2-alkynylphenol substrates [21], including

Graphical Abstract

Scheme 1: Preparation of hydroxyalkynyl substrates from 1-phenyl-1H-pyrazol-3-ol (1).

Scheme 2: Cyclization of hydroxyalkynyl substrates to 2,5-disubstituted 2H-furo[2,3-c]pyrazoles.

Figure 1: a) ORTEP diagram of the asymmetric unit consisting of two independent molecules 4d(A) and 4d(B); b)...
Synthesis of 1,2-divinylcyclopropanes by metal-catalyzed cyclopropanation of 1,3-dienes with cyclopropenes as vinyl carbene precursors
- Jesús González,
- Alba de la Fuente,
- María J. González,
- Laura Díez de Tejada,
- Luis A. López and
- Rubén Vicente
Beilstein J. Org. Chem. 2019, 15, 285–290, doi:10.3762/bjoc.15.25
- proceeded in reasonable yields while the diastereoselectivity strongly depends on the structure of the diene. An example of an intramolecular process as well as the use of furan and 1,4-cyclohexadiene as dienes are also reported. Keywords: cyclopropanation; cyclopropenes; dienes; divinylcyclopropanes
- good yield (77%) in a stereoctive manner (Scheme 4). Finally, we were curious to study the reactivity of metal–vinyl carbenes generated from 3,3-disubstituted cyclopropenes with some particular dienes (Scheme 5). Interestingly, we found that the reaction of cyclopropenes 1a,b with furan (6) using ZnCl2
- compounds, which led again to oxy-Cope rearranged or [4 + 3]-cycloaddition products using rhodium catalysts [14][28][29][30], or to a C2-allylation of furan with gold catalysts [31]. Finally, to compare the reactivity of cyclopropenes and vinyldiazo compounds, we probed the reaction of 1a with 1,4

Graphical Abstract

Scheme 1: Typical syntheses of 1,2-divinylcyclopropanes and rationale hypothesis for their syntheses from cyc...

Scheme 2: Synthesis of 1,2-divinylcyclopropane 3a: Optimization studies. aIsolated yield. bDetermined by 1H N...

Scheme 3: Synthesis of 1,2-divinylcyclopropanes 3 from cyclopropenes 1 and unbiased 1,3-dienes 2: Scope. (Yie...

Scheme 4: Rh-catalyzed intramolecular cyclopropanation with dienylcyclopropene 4 (the trans/cis ratio is rela...

Scheme 5: Zn- or Rh-catalyzed reactions of cyclopropenes 1 with furan (6) and 1,4-cyclohexadiene (8) and comp...
Synthesis of nonracemic hydroxyglutamic acids
- Dorota G. Piotrowska,
- Iwona E. Głowacka,
- Andrzej E. Wróblewski and
- Liwia Lubowiecka
Beilstein J. Org. Chem. 2019, 15, 236–255, doi:10.3762/bjoc.15.22

- L-malic acid [62], was carefully reduced and immediately acetylated to (S)-52 which was reacted with furan to produce a 67:33 mixture of readily separable (2S,3S)-53 and (2R,3S)-53, respectively. Steric hindrance of the acetoxy substituent controls the formation of higher amounts of the trans-isomer
- . Ozone efficiently completed the degradation of the furan ring to the carboxyl group which was esterified with diazomethane to give methyl (2S,3S)-3-acetoxypyroglutamate (2S,3S)-54, a cyclized variant of 3-hydroxyglutamic acid. Treatment with a concentrated acid afforded (2S,3S)-2 as the hydrochloride
- (2S,3R)-2 via Sharpless epoxidation. Reagents and conditions: a) TBHP, D-(−)-DIPT, Ti(OiPr)4, MS, CH2Cl2; b) t-BuMe2SiCl, imidazole, DMAP, DMF; c) NaIO4, RuO2, AcOEt/H2O. Synthesis of (2S,3S)-2 from the imide 51. Reagents and conditions: a) NaBH4, MeOH/CH2Cl2; b) Ac2O, pyridinium perchlorate; c) furan

Graphical Abstract

Figure 1: Structure of L-glutamic acid.

Figure 2: 3-Hydroxy- (2), 4-hydroxy- (3) and 3,4-dihydroxyglutamic acids (4).

Figure 3: Enantiomers of 3-hydroxyglutamic acid (2).

Scheme 1: Synthesis of (2S,3R)-2 from (R)-Garner's aldehyde. Reagents and conditions: a) MeOCH=CH–CH(OTMS)=CH2...

Scheme 2: Synthesis of (2S,3R)-2 and (2S,3S)-2 from (R)-Garner’s aldehyde. Reagents and conditions: a) H2C=CH...

Scheme 3: Two-carbon homologation of the protected L-serine. Reagents and conditions: a) Fmoc-succinimide, Na2...

Scheme 4: Synthesis of di-tert-butyl ester of (2R,3S)-2 from L-serine. Reagents and conditions: a) PhSO2Cl, K2...

Scheme 5: Synthesis of (2R,3S)-2 from O-benzyl-L-serine. Reagents and conditions: a) (CF3CH2O)2P(O)CH2COOMe, ...

Scheme 6: Synthesis of (2S,3R)-2 employing a one-pot cis-olefination–conjugate addition sequence. Reagents an...

Scheme 7: Synthesis of the orthogonally protected (2S,3R)-2 from a chiral aziridine. Reagents and conditions:...

Scheme 8: Synthesis of N-Boc-protected (2S,3R)-2 from D-phenylglycine. Reagents and conditions: a) BnMgCl, et...

Scheme 9: Synthesis of (2S,3R)-2 employing ketopinic acid as chiral auxiliary. Reagents and conditions: a) Br2...

Scheme 10: Synthesis of dimethyl ester of (2S,3R)-2 employing (1S)-2-exo-methoxyethoxyapocamphane-1-carboxylic...

Scheme 11: Synthesis of N-Boc-protected dimethyl ester of (2S,3R)-2 from (S)-N-(1-phenylethyl)thioacetamide. R...

Scheme 12: Synthesis of N-Boc-protected dimethyl ester of (2S,3R)-2 via Sharpless epoxidation. Reagents and co...

Scheme 13: Synthesis of (2S,3S)-2 from the imide 51. Reagents and conditions: a) NaBH4, MeOH/CH2Cl2; b) Ac2O, ...

Scheme 14: Synthesis of (2R,3S)-2 and (2S,3S)-2 from the acetolactam 55 (PMB = p-methoxybenzyl). Reagents and ...

Scheme 15: Synthesis of (2S,3R)-2 from D-glucose. Reagents and conditions: a) NaClO2, 30% H2O2, NaH2PO4, MeCN;...

Figure 4: Enantiomers of 3-hydroxyglutamic acid (3).

Scheme 16: Synthesis of (4S)-4-hydroxy-L-glutamic acid [(2S,4S)-3] by electrophilic hydroxylation. Reagents an...

Scheme 17: Synthesis of all stereoisomers of 4-hydroxyglutamic acid (3). Reagents and conditions: a) Br2, PBr5...

Scheme 18: Synthesis of the orthogonally protected 4-hydroxyglutamic acid (2S,4S)-73. Reagents and conditions:...

Scheme 19: Synthesis of (2S,4R)-4-acetyloxyglutamic acid as a component of a dipeptide. Reagents and condition...

Scheme 20: Synthesis of N-Boc-protected dimethyl esters of (2S,4R)- and (2S,4S)-3 from (2S,4R)-4-hydroxyprolin...

Scheme 21: Synthesis of orthogonally protected (2S,4S)-3 from (2S,4R)-4-hydroxyproline. Reagents and condition...

Scheme 22: Synthesis of the protected (4R)-4-hydroxy-L-pyroglutamic acid (2S,4R)-87 by electrophilic hydroxyla...

Figure 5: Enantiomers of 3,4-dihydroxy-L-glutamic acid (4).

Scheme 23: Synthesis of (2S,3S,4R)-4 from the epoxypyrrolidinone 88. Reagents and conditions: a) MeOH, THF, KC...

Scheme 24: Synthesis of (2S,3R,4R)-4 from the orthoester 92. Reagents and conditions: a) OsO4, NMO, acetone/wa...

Scheme 25: Synthesis of (2S,3S,4S)-4 from the aziridinolactone 95. Reagents and conditions: a) BnOH, BF3·OEt2,...

Scheme 26: Synthesis of (2S,3S,4R)-4 and (2R,3S,4R)-4 from cyclic imides 106. Reagents and conditions: a) NaBH4...

Scheme 27: Synthesis of (2R,3R,4R)-4 and (2S,3R,4R)-4 from the cyclic meso-imide 110. Reagents and conditions:...

Scheme 28: Synthesis of (2S,3S,4S)-4 from the protected serinal (R)-23. Reagents and conditions: a) Ph3P=CHCOO...

Scheme 29: Synthesis of (2S,3S,4S)-4 from O-benzyl-N-Boc-D-serine. Reagents and conditions: a) ClCOOiBu, TEA, ...

Scheme 30: Synthesis of (2S,3S,4R)-127 by enantioselective conjugate addition and asymmetric dihydroxylation. ...

Figure 6: Structures of selected compounds containing hydroxyglutamic motives (in blue).
Unnatural α-amino ethyl esters from diethyl malonate or ethyl β-bromo-α-hydroxyiminocarboxylate
- Eloi P. Coutant,
- Vincent Hervin,
- Glwadys Gagnot,
- Candice Ford,
- Racha Baatallah and
- Yves L. Janin
Beilstein J. Org. Chem. 2018, 14, 2853–2860, doi:10.3762/bjoc.14.264
- yields of α-hydroxyimino esters 2t–v reached 49, 59 and 62% from aldehydes 5t–v, respectively. Of note is that we also tried this reducing agent to improve the overall yield of the benzyloxy-bearing oxime 2ab, but to no avail. We next focused on the furan-bearing α-hydroxyimino esters such as compound
- 2ae. First of all, reduction of the alkylidenemalonate 6ae using a palladium-based catalytic hydrogenation also led to the concomitant hydrogenation of the furan ring to give compound 8. As depicted, this actually allowed us to prepare to the 2-tetrahydrofuranyl-bearing α-aminoester 10 in a 35
- % overall yield via α-hydroxyimino ester 9, but it also forced us to use borohydrides to avoid the furan ring hydrogenation. Accordingly, an extensive study of the reduction of compound 6ae with borohydrides was made. Trials included reactions run at 0 °C overnight, the use of wet ethanol or dry THF, the

Graphical Abstract

Scheme 1: Malonate-based retrosynthesis of α-amino esters.

Scheme 2: Some side products and synthesis of α-amino ester 10.

Scheme 3: Syntheses of α-amino esters 22, 24, 26, 28 and 33.

Scheme 4: Syntheses of α-amino esters 38, 41 and 46a,b.

Scheme 5: Syntheses of α-amino esters 53 and 58.
Synthesis of unnatural α-amino esters using ethyl nitroacetate and condensation or cycloaddition reactions
- Glwadys Gagnot,
- Vincent Hervin,
- Eloi P. Coutant,
- Sarah Desmons,
- Racha Baatallah,
- Victor Monnot and
- Yves L. Janin
Beilstein J. Org. Chem. 2018, 14, 2846–2852, doi:10.3762/bjoc.14.263
- improvements were observed with these conditions. Other series of trials were made to improve the overall yields of the furan-bearing α-nitro esters 6n–r. We first tried to avoid the preparation of the acetals 5n or 5o and used the previously reported direct condensation between furfural (3n) and ethyl
- acid in ethanol overnight gave a sufficient amount of the (pure) target phenyl-bearing α-amino ester 10 which had been previously obtained by catalytic hydrogenation using palladium [20]. The same transformation sequences were used starting with compound 6n and provided the furan-bearing α-amino ester

Graphical Abstract

Scheme 1: α-Amino esters from ethyl nitroacetate (4).

Scheme 2: Preparations of α-amino esters 10, 12 and 14.

Scheme 3: Syntheses of α-amino ester 18 and piperazinediones 23a,b.

Scheme 4: Syntheses of α-hydroximino ester 29 and α-amino ester 36.

Scheme 5: Synthesis of α-amino ester 43.
Novel photochemical reactions of carbocyclic diazodiketones without elimination of nitrogen – a suitable way to N-hydrazonation of C–H-bonds
- Liudmila L. Rodina,
- Xenia V. Azarova,
- Jury J. Medvedev,
- Dmitrij V. Semenok and
- Valerij A. Nikolaev
Beilstein J. Org. Chem. 2018, 14, 2250–2258, doi:10.3762/bjoc.14.200
- %. Further irradiation of hydrazones derived from furan-fused tricyclic diazocyclopentanediones culminates in the cycloelimination of furans to yield 2-N-(alkyl)hydrazone of cyclopentene-1,2,3-trione. By contrast, the direct photolysis of carbocyclic diazodiketones gives only Wolff rearrangement products
- unexpected side reaction was observed during irradiation of furan-fused tricyclic diazodiketones 1d,e (Scheme 1). In the case of diazodiketone 1d, hydrazone 3 (47%) was isolated from reaction mixture in addition to the expected hydrazone 2d (35%; total yield 82%, Table 1, entry 11), whereas the same reaction
- light-induced cycloelimination of furan from the initially generated tricyclic hydrazones 2d,e. Hence it was established that the sensitized photoexcitation of diazodiketones 1a–d,f in THF solution proceeds through the insertion of the terminal N-atom of the diazo group in the α-С–Н-bond of THF with the

Graphical Abstract

Figure 1: The structures of carbocyclic diazodiketones 1a–g and C–H-donating tetrahydrofuran used in the proj...

Figure 2: Molecular structure of hydrazone 2b as determined by X-ray analysis data (Olex2 plot with 50% proba...

Scheme 1: Photochemical cycloelimination of furans from hydrazones 2d,e.

Scheme 2: Different pathways of diazodiketones 1 light-induced reactions in the singlet (reaction I) and trip...
Studies towards the synthesis of hyperireflexolide A
- G. Hari Mangeswara Rao
Beilstein J. Org. Chem. 2018, 14, 2106–2111, doi:10.3762/bjoc.14.185
- . Experimental procedures and analytical data Methyl 1,1,5-trimethyl-3,4-dioxohexahydro-1H-cyclopenta[c]furan-5-carboxylate (8): To a solution of the γ-lactone-fused cyclopentanone 6 (16 mg, 0.07 mmol) in dry acetone (0.4 mL) under argon was added anhydrous K2CO3 (9.7 mg, 0.07 mmol) and methyl iodide (12 mg
- . Methyl 1,1,3a,5-tetramethyl-3,4-dioxohexahydro-1H-cyclopenta[c]furan-5-carboxylate (9): To a solution of the γ-lactone-fused cyclopentanone 8 (15 mg, 0.07 mmol) in dry acetone (0.4 mL) under argon was added anhydrous K2CO3 (9.7 mg, 0.07 mmol) and methyl iodide (12 mg, 0.08 mmol) at 0 °C. The reaction was
- -dioxohexahydro-1H-cyclopenta[c]furan-5-carboxylate (4): To a solution of the γ-lactone-fused cyclopentanone 6 (75 mg, 0.331 mmol) in dry acetone (1 mL) under argon was added anhydrous K2CO3 (55 mg, 0.3972 mmol) and allyl bromide (48 mg, 0.3972 mmol) at 0 °C. The reaction was stirred at 0 °C for 21 h. Completion

Graphical Abstract

Figure 1: Hyperireflexolide A.

Scheme 1: Retrosynthetic strategy.

Scheme 2: Hydrolysis of dimethyl ketal 5.

Scheme 3: Alkylation of γ-lactone-fused β-ketoester 6.

Scheme 4: Synthesis of α,β-unsaturated ketone 11.
Synthesis of pyrazolopyrimidinones using a “one-pot” approach under microwave irradiation
- Mark Kelada,
- John M. D. Walsh,
- Robert W. Devine,
- Patrick McArdle and
- John C. Stephens
Beilstein J. Org. Chem. 2018, 14, 1222–1228, doi:10.3762/bjoc.14.104
- 1). The heterocyclic furan and thiophene substituents allowed generation of the desired 5-aminopyrazoles in 75% and 81% yields, respectively (Scheme 1). Alkyl groups appeared to be less well tolerated, where the corresponding alkyl-substituted 5-aminopyrazoles were isolated in lower yields. With a

Graphical Abstract

Figure 1: Bioactive pyrazolo[1,5-a]pyrimidinones.

Scheme 1: Synthesis of 5-aminopyrazoles. Reaction conditions: ketonitrile (2.0 mmol, 1.0 equiv), hydrazine (2...

Scheme 2: One-pot synthesis of pyrazolo[1,5-a]pyrimidinones. aReaction conditions: ketonitrile (0.9 mmol, 1.0...

Figure 2: X-ray crystal structure of pyrazolo[1,5-a]pyrimidinone 3m with ellipsoids at 50% probability.
One hundred years of benzotropone chemistry
- Arif Dastan,
- Haydar Kilic and
- Nurullah Saracoglu
Beilstein J. Org. Chem. 2018, 14, 1120–1180, doi:10.3762/bjoc.14.98

Graphical Abstract

Scheme 1: Tropone (1), tropolone (2) and their resonance structures.

Figure 1: Natural products containing a tropone nucleus.

Figure 2: Possible isomers 11–13 of benzotropone.

Scheme 2: Synthesis of benzotropones 11 and 12.

Scheme 3: Oxidation products of benzotropylium fluoroborate (16).

Scheme 4: Oxidation of 7-bromo-5H-benzo[7]annulene (22).

Scheme 5: Synthesis of 4,5-benzotropone (11) using o-phthalaldehyde (27).

Scheme 6: Synthesis of 4,5-benzotropone (11) starting from oxobenzonorbornadiene 31.

Scheme 7: Acid-catalyzed cleavage of oxo-bridge of 34.

Scheme 8: Synthesis of 4,5-benzotropone (11) from o-xylylene dibromide (38).

Scheme 9: Synthesis of 4,5-benzotropone (11) via the carbene adduct 41.

Scheme 10: Heck coupling strategy for the synthesis of 11.

Scheme 11: Synthesis of benzofulvalenes via carbonyl group of 4,5-benzotropone (11).

Figure 3: Some cycloheptatrienylium cations.

Scheme 12: Synthesis of condensation product 63 and its subsequent oxidative cyclization products.

Figure 4: A novel series of benzo[7]annulenes prepared from 4,5-benzotropone (11).

Scheme 13: Preparation of substituted benzo[7]annulene 72 using the Mukaiyama-Michael reaction.

Figure 5: Possible benzo[7]annulenylidenes 73–75.

Scheme 14: Thermal and photochemical decomposition of 7-diazo-7H-benzo[7]annulene (76) and the trapping of int...

Scheme 15: Synthesis of benzoheptafulvalene 86.

Scheme 16: Synthesis of 7-(diphenylmethylene)-7H-benzo[7]annulene (89).

Scheme 17: Reaction of 4,5-benzotropone (11) with dimethyl diazomethane.

Scheme 18: Synthesis of dihydrobenzomethoxyazocine 103.

Scheme 19: Synthesis and reducibility of benzo-homo-2-methoxyazocines.

Scheme 20: Synthesis of 4,5-benzohomotropones 104 and 115 from 4,5-benzotropones 11 and 113.

Scheme 21: A catalytic deuterogenation of 4,5-benzotropone (11) and synthesis of 5-monosubstituted benzo[7]ann...

Scheme 22: Synthesis of methyl benzo[7]annulenes 131 and 132.

Scheme 23: Ambident reactivity of halobenzo[7]annulenylium cations 133a/b.

Scheme 24: Preparation of benzo[7]annulenylidene–iron complexes 147.

Scheme 25: Synthesis of 1-ethynylbenzotropone (150) and the etheric compound 152 from 4,5-benzotropone (11) wi...

Scheme 26: Thermal decomposition of 4,5-benzotropone (11).

Scheme 27: Reaction of 4,5-benzotropone (11) with 1,2-ethanediol and 1,2-ethanedithiol.

Scheme 28: Conversions of 1-benzosuberone (162) to 2,3-benzotropone (12).

Scheme 29: Synthesis strategies for 2,3-bezotropone (12) using 1-benzosuberones.

Scheme 30: Oxidation-based synthesis of 2,3-benzotropone (12) via 1-benzosuberone (162).

Scheme 31: Synthesis of 2,3-benzotropone (12) from α-tetralone (171) via ring-expansion.

Scheme 32: Preparation of 2,3-benzotropone (12) by using of benzotropolone 174.

Figure 6: Benzoheptafulvenes as condensation products of 2,3-benzotropone (12).

Scheme 33: Conversion of 2,3-benzotropone (12) to tosylhydrazone salt 182 and gem-dichloride 187.

Figure 7: Benzohomoazocines 191–193 and benzoazocines 194–197.

Scheme 34: From 2,3-benzotropone (12) to carbonium ions 198–201.

Scheme 35: Cycloaddition reactions of 2,3-benzotropone (12).

Scheme 36: Reaction of 2,3-benzotropone (12) with various reagents and compounds.

Figure 8: 3,4-Benzotropone (13) and its resonance structure.

Scheme 37: Synthesis of 6,7-benzobicyclo[3.2.0]hepta-3,6-dien-2-one (230).

Figure 9: Photolysis and thermolysis products of 230.

Figure 10: Benzotropolones and their tautomeric structures.

Scheme 38: Synthesis strategies of 4,5-benzotropolone (238).

Scheme 39: Synthesis protocol for 2-hydroxy-4,5-benzotropone (238) using oxazole-benzo[7]annulene 247.

Figure 11: Some quinoxaline and pyrazine derivatives 254–256 prepared from 4,5-benzotropolone (238).

Scheme 40: Nitration product of 4,5-benzotropolone (238) and its isomerization to 1-nitro-naphthoic acid (259)....

Scheme 41: Synthesis protocol for 6-hydroxy-2,3-benzotropone (239) from benzosuberone (162).

Scheme 42: Various reactions via 6-hydroxy-2,3-benzotropone (239).

Scheme 43: Photoreaction of 6-hydroxy-2,3-benzotropone (239).

Scheme 44: Synthesis of 7-hydroxy-2,3-benzotropone (241) from benzosuberone (162).

Scheme 45: Synthesis strategy for 7-hydroxy-2,3-benzotropone (241) from ketone 276.

Scheme 46: Synthesis of 7-hydroxy-2,3-benzotropone (241) from β-naphthoquinone (280).

Scheme 47: Synthesis of 7-hydroxy-2,3-benzotropone (241) from bicyclic endoperoxide 213.

Scheme 48: Synthesis of 7-hydroxy-2,3-benzotropone (241) by ring-closing metathesis.

Figure 12: Various monosubstitution products 289–291 of 7-hydroxy-2,3-benzotropone (241).

Scheme 49: Reaction of 7-hydroxy-2,3-benzotropone (241) with various reagents.

Scheme 50: Synthesis of 4-hydroxy-2,3-benzotropones 174 and 304 from diketones 300/301.

Scheme 51: Catalytic hydrogenation of diketones 300 and 174.

Scheme 52: Synthesis of halo-benzotropones from alkoxy-naphthalenes 306, 307 and 310.

Figure 13: Unexpected byproducts 313–315 during synthesis of chlorobenzotropone 309.

Figure 14: Some halobenzotropones and their cycloadducts.

Scheme 53: Multisep synthesis of 2-chlorobenzotropone 309.

Scheme 54: A multistep synthesis of 2-bromo-benzotropone 26.

Scheme 55: A multistep synthesis of bromo-2,3-benzotropones 311 and 316.

Scheme 56: Oxidation reactions of 8-bromo-5H-benzo[7]annulene (329) with some oxidants.

Scheme 57: Synthesis of 2-bromo-4,5-benzotropone (26).

Scheme 58: Synthesis of 6-chloro-2,3-benzotropone (335) using LiCl and proposed intermediate 336.

Scheme 59: Reaction of 7-bromo-2,3-benzotropone (316) with methylamine.

Scheme 60: Reactions of bromo-2,3-benzotropones 26 and 311 with dimethylamine.

Scheme 61: Reactions of bromobenzotropones 311 and 26 with NaOMe.

Scheme 62: Reactions of bromobenzotropones 26 and 312 with t-BuOK in the presence of DPIBF.

Scheme 63: Cobalt-catalyzed reductive cross-couplings of 7-bromo-2,3-benzotropone (316) with cyclic α-bromo en...

Figure 15: Cycloadduct 357 and its di-π-methane rearrangement product 358.

Scheme 64: Catalytic hydrogenation of 2-chloro-4,5-benzotropone (311).

Scheme 65: Synthesis of dibromo-benzotropones from benzotropones.

Scheme 66: Bromination/dehydrobromination of benzosuberone (162).

Scheme 67: Some transformations of isomeric dibromo-benzotropones 261A/B.

Scheme 68: Transformations of benzotropolone 239B to halobenzotropolones 369–371.

Figure 16: Bromobenzotropolones 372–376 and 290 prepared via bromination/dehydrobromination strategy.

Scheme 69: Synthesis of some halobenzotropolones 289, 377 and 378.

Figure 17: Bromo-chloro-derivatives 379–381 prepared via chlorination.

Scheme 70: Synthesis of 7-iodo-3,4-benzotropolone (382).

Scheme 71: Hydrogenation of bromobenzotropolones 369 and 370.

Scheme 72: Debromination reactions of mono- and dibromides 290 and 375.

Figure 18: Nitratation and oxidation products of some halobenzotropolenes.

Scheme 73: Azo-coupling reactions of some halobenzotropolones 294, 375 and 378.

Figure 19: Four possible isomers of dibenzotropones 396–399.

Figure 20: Resonance structures of tribenzotropone (400).

Scheme 74: Two synthetic pathways for tribenzotropone (400).

Scheme 75: Synthesis of tribenzotropone (400) from dibenzotropone 399.

Scheme 76: Synthesis of tribenzotropone (400) from 9,10-phenanthraquinone (406).

Scheme 77: Synthesis of tribenzotropone (400) from trifluoromethyl-substituted arene 411.

Figure 21: Dibenzosuberone (414).

Figure 22: Reduction products 415 and 416 of tribenzotropone (400).

Figure 23: Structures of tribenzotropone dimethyl ketal 417 and 4-phenylfluorenone (412) and proposed intermed...

Figure 24: Structures of benzylidene- and methylene-9H-tribenzo[a,c,e][7]annulenes 419 and 420 and chiral phos...

Figure 25: Structures of tetracyclic alcohol 422, p-quinone methide 423 and cation 424.

Figure 26: Structures of host molecules 425–427.

Scheme 78: Synthesis of non-helical overcrowded derivatives syn/anti-431.

Figure 27: Hexabenzooctalene 432.

Figure 28: Structures of possible eight isomers 433–440 of naphthotropone.

Scheme 79: Synthesis of naphthotropone 437 starting from 1-phenylcycloheptene (441).

Scheme 80: Synthesis of 10-hydroxy-11H-cyclohepta[a]naphthalen-11-one (448) from diester 445.

Scheme 81: Synthesis of naphthotropone 433.

Scheme 82: Synthesis of naphthotropones 433 and 434 via cycloaddition reaction.

Scheme 83: Synthesis of naphthotropone 434 starting from 452.

Figure 29: Structures of tricarbonyl(tropone)irons 458, and possible cycloadducts 459.

Scheme 84: Synthesis of naphthotropone 436.

Scheme 85: Synthesis of precursor 465 for naphthotropone 435.

Scheme 86: Generation of naphthotropone 435 from 465.

Figure 30: Structures of tropylium cations 469 and 470.

Figure 31: Structures of tropylium ions 471+.BF4−, 472+.BF4−, and 473+.BF4−.

Scheme 87: Synthesis of tropylium ions 471+.BF4− and 479+.ClO4−.

Scheme 88: Synthesis of 1- and 2-methylanthracene (481 and 482) via carbene–carbene rearrangement.

Figure 32: Trapping products 488–490.

Scheme 89: Generation and chemistry of a naphthoannelated cycloheptatrienylidene-cycloheptatetraene intermedia...

Scheme 90: Proposed intermediates and reaction pathways for adduct 498.

Scheme 91: Exited-state intramolecular proton transfer of 505.

Figure 33: Benzoditropones 506 and 507.

Scheme 92: Synthesis of benzoditropone 506e.

Scheme 93: Synthetic approaches for dibenzotropone 507 via tropone (1).

Scheme 94: Formation mechanisms of benzoditropone 507 and 516 via 515.

Scheme 95: Synthesis of benzoditropones 525 and 526 from pyromellitic dianhydride (527).

Figure 34: Possible three benzocyclobutatropones 534–536.

Scheme 96: Synthesis of benzocyclobutatropones 534 and 539.

Scheme 97: Synthesis attempts for benzocyclobutatropone 545.

Scheme 98: Generation and trapping of symmetric benzocyclobutatropone 536.

Scheme 99: Synthesis of chloro-benzocyclobutatropone 552 and proposed mechanism of fluorenone derivatives.

Scheme 100: Synthesis of tropolone analogue 559.

Scheme 101: Synthesis of tropolones 561 and 562.

Figure 35: o/p-Tropoquinone rings (563 and 564) and benzotropoquinones (565–567).

Scheme 102: Synthesis of benzotropoquinone 566.

Scheme 103: Synthesis of benzotropoquinone 567 via a Diels–Alder reaction.

Figure 36: Products 575–577 through 1,2,3-benzotropoquinone hydrate 569.

Scheme 104: Structures 578–582 prepared from tropoquinone 567.

Figure 37: Two possible structures 583 and 584 for dibenzotropoquinone, and precursor compound 585 for 583.

Scheme 105: Synthesis of saddle-shaped ketone 592 using dibenzotropoquinone 584.
An overview of recent advances in duplex DNA recognition by small molecules
- Sayantan Bhaduri,
- Nihar Ranjan and
- Dev P. Arya
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
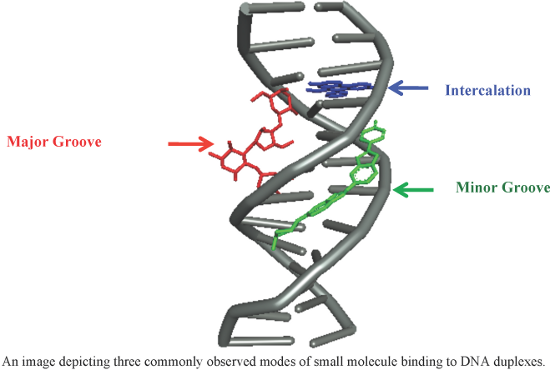
- heterocyclic rings such as furan 62 and 63 [126], thiophene 64 [127] and pyridine 65 (Figure 11) [128]. These conjugates exhibit potent antibacterial and antiprotozoal activity with much reduced toxicity. It was further concluded that π-stacking, H-bonding with the floor of the minor groove along with

Graphical Abstract

Figure 1: A figure showing the hydrogen bonding patterns observed in (a) duplex (b) triplex and (c) quadruple...

Figure 2: (a) Portions of MATα1–MATα2 are shown contacting the minor groove of the DNA substrate. Key arginin...

Figure 3: Chemical structures of naturally occurring and synthetic hybrid minor groove binders.

Figure 4: Synthetic structural analogs of distamycin A by replacing one or more pyrrole rings with other hete...

Figure 5: Pictorial representation of the binding model of pyrrole–imidazole (Py/Im) polyamides based on the ...

Figure 6: Chemical structures of synthetic “hairpin” pyrrole–imidazole (Py/Im) conjugates.

Figure 7: (a) Minor groove complex formation between DNA duplex and 8-ring cyclic Py/Im polyamide (conjugate ...

Figure 8: Telomere-targeting tandem hairpin Py/Im polyamides 23 and 24 capable of recognizing >10 base pairs; ...

Figure 9: Representative examples of recently developed DNA minor groove binders.

Figure 10: Chemical structures of bisbenzamidazoles Hoechst 33258 and 33342 and their synthetic structural ana...

Figure 11: Chemical structures of bisamidines such as diminazene, DAPI, pentamidine and their synthetic struct...

Figure 12: Representative examples of recently developed bisamidine derivatives.

Figure 13: Chemical structures of chromomycin, mithramycin and their synthetic structural analogs 91 and 92.

Figure 14: Chemical structures of well-known naturally occurring DNA binding intercalators.

Figure 15: Naturally occurring indolocarbazole rebeccamycin and its synthetic analogs.

Figure 16: Representative examples of naturally occurring and synthetic derivatives of DNA intercalating agent...

Figure 17: Several recent synthetic varieties of DNA intercalators.

Figure 18: Aminoglycoside (neomycin)–Hoechst 33258/intercalator conjugates.

Figure 19: Chemical structures of triazole linked neomycin dimers and neomycin–bisbenzimidazole conjugates.

Figure 20: Representative examples of naturally occurring and synthetic analogs of DNA binding alkylating agen...

Figure 21: Chemical structures of naturally occurring and synthetic analogs of pyrrolobenzodiazepines.
Hypervalent iodine-mediated Ritter-type amidation of terminal alkenes: The synthesis of isoxazoline and pyrazoline cores
- Sang Won Park,
- Soong-Hyun Kim,
- Jaeyoung Song,
- Ga Young Park,
- Darong Kim,
- Tae-Gyu Nam and
- Ki Bum Hong
Beilstein J. Org. Chem. 2018, 14, 1028–1033, doi:10.3762/bjoc.14.89
- (3a–c, 3f) [31]. However, electron-rich aryl allyl ketone oximes such as 1d, 1e and 1g proved inferior. Also the furan-substituted allyl ketone oxime delivered the desired product 3h albeit in a moderate yield. In addition, various alkyl allyl ketone oximes were investigated. While cyclopropyl allyl

Graphical Abstract

Scheme 1: Hypervalent iodine-mediated heterofunctionalization of terminal alkenes.

Scheme 2: Substrate scope of the Ritter-type oxyamidation: isoxazoline synthesis. All reactions were performe...

Scheme 3: Substrate scope of Ritter-type amido-amidation: pyrazoline synthesis. All reactions were performed ...

Scheme 4: Plausible mechanism of the hypervalent iodine(III)-mediated Ritter-type oxyamidation/amido-amidatio...
Fluorocyclisation via I(I)/I(III) catalysis: a concise route to fluorinated oxazolines
- Felix Scheidt,
- Christian Thiehoff,
- Gülay Yilmaz,
- Stephanie Meyer,
- Constantin G. Daniliuc,
- Gerald Kehr and
- Ryan Gilmour
Beilstein J. Org. Chem. 2018, 14, 1021–1027, doi:10.3762/bjoc.14.88
- explored (to generate 2k and 2l, respectively). Unsurprisingly, whilst the 6-membered ring formed in 42% yield, cyclisation to form the analogous 7-membered ring failed. It was, however, possible to generate heterocyclic species such as the 9-fluorenonyl-substituted oxazoline 2m (48%) and the furan 2n (59

Graphical Abstract

Figure 1: Selected examples of bioactive compounds containing the 2-oxazoline motif.

Figure 2: The catalytic difluorination of alkenes (top) and the proposed fluorocyclisation via the same I(I)/...

Figure 3: Substrate scope. aReaction conducted on 1 mmol scale. bReaction time increased to 40 hours. cReacti...

Scheme 1: Exploring diastereocontrol and the synthesis of the fluorohydrin 3. Yields in parentheses were dete...

Figure 4: X-ray molecular structure of compound 2c. Thermal ellipsoids shown at the 50% propability level. To...
Mannich base-connected syntheses mediated by ortho-quinone methides
- Petra Barta,
- Ferenc Fülöp and
- István Szatmári
Beilstein J. Org. Chem. 2018, 14, 560–575, doi:10.3762/bjoc.14.43
- phenolic Mannich bases, leading to the disappearance of the only aromatic ring. In a recent publication by same research group [72], they elaborated a simple route to 1,2-dihydronaphtho[2,1-b]furan and 2,3-dihydrobenzofurans via base-induced desamination. They also reported the development of a simple

Graphical Abstract

Scheme 1: Formation of amidoalkylnaphthols 4 via o-QM intermediate 3.

Scheme 2: Asymmetric syntheses of triarylmethanes starting from diarylmethylamines.

Scheme 3: Proposed mechanism for the formation of 2,2-dialkyl-3-dialkylamino-2,3-dihydro-1H-naphtho[2,1-b]pyr...

Scheme 4: Cycloadditions of isoflavonoid-derived o-QMs and various dienophiles.

Scheme 5: [4 + 2] Cycloaddition reactions between aminonaphthols and cyclic amines.

Scheme 6: Brønsted acid-catalysed reaction between aza-o-QMs and 2- or 3-substituted indoles.

Scheme 7: Formation of 3-(α,α-diarylmethyl)indoles 52 in different synthetic pathways.

Scheme 8: Alkylation of o-QMs with N-, O- or S-nucleophiles.

Scheme 9: Formation of DNA linkers and o-QM mediated polymers.
Diels–Alder cycloadditions of N-arylpyrroles via aryne intermediates using diaryliodonium salts
- Huangguan Chen,
- Jianwei Han and
- Limin Wang
Beilstein J. Org. Chem. 2018, 14, 354–363, doi:10.3762/bjoc.14.23
- diaryliodonium salts can generate benzynes under severe basic conditions, the resulted benzynes were allowed to undergo cycloaddition reaction with furan or N-arylation of secondary amides and amines. Due to the easy accessibility of the diaryliodonium salts, this kind of benzyne precursor is attracting
- extensive attention [14][15][16]. Also as a five-membered heterocyclic ring, the cycloaddition reaction of N-substituted pyrroles is much less than that of furan [17][18][19][20][21]. As a matter of fact, the Diels–Alder adduct formation of pyrroles with benzyne has been postulated in 1965 as transient

Graphical Abstract

Scheme 1: Arylations of pyrrole derivatives with diaryliodonium salts.

Scheme 2: Formation of N-phenylamine derivatives 4 and 5 via ring opening reactions.

Scheme 3: Preparation of product 6 by hydrogenation.
Recent advances on organic blue thermally activated delayed fluorescence (TADF) emitters for organic light-emitting diodes (OLEDs)
- Thanh-Tuân Bui,
- Fabrice Goubard,
- Malika Ibrahim-Ouali,
- Didier Gigmes and
- Frédéric Dumur
Beilstein J. Org. Chem. 2018, 14, 282–308, doi:10.3762/bjoc.14.18

- 5 hours for T13-based OLEDs. A comparison established with an iridium complex, i.e., tris[1-(2,4-diisopropyldibenzo[b,d]furan-3-yl)-2-phenyl-1H-imidazole]iridium(III) (Ir(dbi)3) evidenced the relevance of the TADF approach, as a device lifetime of only 18 hours was found while operating OLEDs in the

Graphical Abstract

Figure 1: Radiative deactivation pathways existing in fluorescent, phosphorescent and TADF materials.

Figure 2: Boron-containing TADF emitters B1–B10.

Figure 3: Diphenylsulfone-based TADF emitters D1–D7.

Figure 4: Triazine-based TADF emitters T1–T3, T5–T7 and azasiline derivatives T3 and T4.

Figure 5: Triazine-based TADF emitters T8, T9, T11–T14 and carbazole derivative T10.

Figure 6: Triazine-based TADF emitters T15–T19.

Figure 7: Triazine- and pyrimidine-based TADF emitters T20–T26.

Figure 8: Pyrimidine-based TADF emitters T27–T30.

Figure 9: Triazine-based TADF polymers T31–T32.

Figure 10: Phenoxaphosphine oxide and phenoxathiin dioxide-based TADF emitters P1 and P2.

Figure 11: CN-Substituted pyridine and pyrimidine derivatives CN-P1–CN-P8.

Figure 12: CN-Substituted pyridine derivatives CN-P9 and CN-P10.

Figure 13: Phosphine oxide-based TADF blue emitters PO-1–PO-3.

Figure 14: Phosphine oxide-based TADF blue emitters PO-4–PO-9.

Figure 15: Benzonitrile-based emitters BN-1–BN-5.

Figure 16: Benzonitrile-based emitters BN-6–BN-11.

Figure 17: Benzoylpyridine-carbazole hybrid emitters BP-1–BP-6.

Figure 18: Benzoylpyridine-carbazole hybrid emitters BP-7–BP-10.

Figure 19: Triazole-based emitters Trz-1 and Trz-2.

Figure 20: Triarylamine-based emitters TPA-1–TPA-3.

Figure 21: Distribution of the CIE coordinates of ca. 90 blue TADF emitters listed in this review.
One-pot sequential synthesis of tetrasubstituted thiophenes via sulfur ylide-like intermediates
- Jun Ki Kim,
- Hwan Jung Lim,
- Kyung Chae Jeong and
- Seong Jun Park
Beilstein J. Org. Chem. 2018, 14, 243–252, doi:10.3762/bjoc.14.16
- -alkylation compound 7e was obtained instead of the desired furan (Table 1, entry 5). It is possible to consider that the d orbitals of the sulfur atom in a sulfide group could possibly stabilize the adjacent carbanion [73][74]. To expand the scope of substituted N,S-acetals that could provide the desired
- compounds 7f and 7g, the low reactivity resulted from the decreased mesomeric effect of the furan structure: the higher electronegativity of oxygen facilitated the polarized form [71]. Among various arenes, the 4-nitrophenyl substituent 7p only afforded the desired thiophene 8p in a moderated yield (42

Graphical Abstract

Figure 1: The selected examples of sulfur(IV) and sulfur(VI) ylides 1 [1], 2 [5-7], 3 [6,7,9], 4 [11,12], 5 [33,34], 6 [35-38].

Figure 2: Metal-free synthesis of thiophene-based heterocycles (A) [54,55], (B) [56].

Scheme 1: One-pot sequential synthesis of the trisubstituted 5-(pyridine-2-yl)thiophenes 8a. Substrate: amalo...

Figure 3: X-ray crystal structures of 8ad and 8an [68].

Figure 4: The proposed structure of sulfur ylide-like intermediates; resonance contributors (mesomeric struct...

Scheme 2: The substitution reaction with MeOH.
5-Aminopyrazole as precursor in design and synthesis of fused pyrazoloazines
- Ranjana Aggarwal and
- Suresh Kumar
Beilstein J. Org. Chem. 2018, 14, 203–242, doi:10.3762/bjoc.14.15
- -(furan-2-yl)-2-phenylpyrazolo[1,5-a]pyrimidine (168) using a similar synthetic strategy from the reaction of 5-aminopyrazole 16/126 with sodium 3-(furan-2-yl)-3-oxoprop-1-en-1-olate (167, Scheme 47). Ren et al. [108] described the synthesis of 6-aminopyrazolo[1,5-a]pyrimidine derivatives 172 involving

Graphical Abstract

Figure 1: Selected examples of drugs with fused pyrazole rings.

Figure 2: Typical structures of some fused pyrazoloazines from 5-aminopyrazoles.

Scheme 1: Regiospecific synthesis of 4 and 6-trifluoromethyl-1H-pyrazolo[3,4-b]pyridines.

Scheme 2: Synthesis of pyrazolo[3,4-b]pyridine-6-carboxylates.

Scheme 3: Synthesis of 1,4,6-triaryl-1H-pyrazolo[3,4-b]pyridines with ionic liquid .

Scheme 4: Synthesis of coumarin-based isomeric tetracyclic pyrazolo[3,4-b]pyridines.

Scheme 5: Synthesis of 6-substituted pyrazolo[3,4-b]pyridines under Heck conditions.

Scheme 6: Microwave-assisted palladium-catalyzed synthesis of pyrazolo[3,4-b]pyridines.

Scheme 7: Acid-catalyzed synthesis of pyrazolo[3,4-b]pyridines via enaminones.

Scheme 8: Synthesis of pyrazolo[3,4-b]pyridines via aza-Diels–Alder reaction.

Scheme 9: Synthesis of macrocyclane fused pyrazolo[3,4-b]pyridine derivatives.

Scheme 10: Three-component synthesis of 4,7-dihydro-1H-pyrazolo[3,4-b]pyridine derivatives.

Scheme 11: Ultrasonicated synthesis of spiro[indoline-3,4'-pyrazolo[3,4-b]pyridine]-2,6'(1'H)-diones.

Scheme 12: Synthesis of spiro[indoline-3,4'-pyrazolo[3,4-b]pyridine] derivatives under conventional heating co...

Scheme 13: Nanoparticle-catalyzed synthesis of pyrazolo[3,4-b]pyridine-spiroindolinones.

Scheme 14: Microwave-assisted multicomponent synthesis of spiropyrazolo[3,4-b]pyridines.

Scheme 15: Unexpected synthesis of naphthoic acid-substituted pyrazolo[3,4-b]pyridines.

Scheme 16: Multicomponent synthesis of variously substituted pyrazolo[3,4-b]pyridine derivatives.

Scheme 17: Three-component synthesis of 4,7-dihydropyrazolo[3,4-b]pyridines and pyrazolo[3,4-b]pyridines.

Scheme 18: Synthesis of pyrazolo[3,4-b]pyridine-5-spirocycloalkanediones.

Scheme 19: Ultrasound-mediated three-component synthesis of pyrazolo[3,4-b]pyridines.

Scheme 20: Multicomponent synthesis of 4-aryl-3-methyl-1-phenyl-4,6,8,9-tetrahydropyrazolo [3,4-b]thiopyrano[4...

Scheme 21: Synthesis of 2,3-dihydrochromeno[4,3-d]pyrazolo[3,4-b]pyridine-1,6-diones.

Scheme 22: FeCl3-catalyzed synthesis of o-hydroxyphenylpyrazolo[3,4-b]pyridine derivatives.

Scheme 23: Ionic liquid-mediated synthesis of pyrazolo[3,4-b]pyridines.

Scheme 24: Microwave-assisted synthesis of pyrazolo[3,4-b]pyridines.

Scheme 25: Multicomponent synthesis of pyrazolo[3,4-b]pyridine-5-carbonitriles.

Scheme 26: Unusual domino synthesis of 4,7-dihydropyrazolo[3,4-b]pyridine-5-nitriles.

Scheme 27: Synthesis of 4,5,6,7-tetrahydro-4H-pyrazolo[3,4-b]pyridines under conventional heating and ultrasou...

Scheme 28: L-Proline-catalyzed synthesis of of pyrazolo[3,4-b]pyridine.

Scheme 29: Microwave-assisted synthesis of 5-aminoarylpyrazolo[3,4-b]pyridines.

Scheme 30: Microwave-assisted multi-component synthesis of pyrazolo[3,4-e]indolizines.

Scheme 31: Synthesis of fluoropropynyl and fluoroalkyl substituted pyrazolo[1,5-a]pyrimidine.

Scheme 32: Acid-catalyzed synthesis of pyrazolo[1,5-a]pyrimidine derivatives.

Scheme 33: Chemoselective and regiospecific synthesis of 2-(3-methylpyrazol-1’-yl)-5-methylpyrazolo[1,5-a]pyri...

Scheme 34: Regioselective synthesis of 7-trifluoromethylpyrazolo[1,5-a]pyrimidines.

Scheme 35: Microwave-assisted synthesis of 7-trifluoromethylpyrazolo[1,5-a]pyrimidine carboxylates.

Scheme 36: Microwave and ultrasound-assisted synthesis of 7-trifluoromethylpyrazolo[1,5-a]pyrimidines.

Scheme 37: Base-catalyzed unprecedented synthesis of pyrazolo[1,5-a]pyrimidines via C–C bond cleavage.

Scheme 38: Synthesis of aminobenzothiazole/piperazine linked pyrazolo[1,5-a]pyrimidines.

Scheme 39: Synthesis of aminoalkylpyrazolo[1,5-a]pyrimidine-7-amines.

Scheme 40: Synthesis of pyrazolo[1,5-a]pyrimidines from condensation of 5-aminopyrazole 126 and ethyl acetoace...

Scheme 41: Synthesis of 7-aminopyrazolo[1,5-a]pyrimidines.

Scheme 42: Unexpected synthesis of 7-aminopyrazolo[1,5-a]pyrimidines under solvent free and solvent-mediated c...

Scheme 43: Synthesis of N-(4-aminophenyl)-7-aryloxypyrazolo[1,5-a]pyrimidin-5-amines.

Scheme 44: Base-catalyzed synthesis of 5,7-diarylpyrazolo[1,5-a]pyrimidines.

Scheme 45: Synthesis of 6,7-dihydropyrazolo[1,5-a]pyrimidines in PEG-400.

Scheme 46: Synthesis of 7-heteroarylpyrazolo[1,5-a]pyrimidine-3-carboxamides.

Scheme 47: Synthesis of 7-heteroarylpyrazolo[1,5-a]pyrimidine derivatives under conventional heating and micro...

Scheme 48: Synthesis of N-aroylpyrazolo[1,5-a]pyrimidine-5-amines.

Scheme 49: Regioselective synthesis of ethyl pyrazolo[1,5-a]pyrimidine-7-carboxylate.

Scheme 50: Sodium methoxide-catalyzed synthesis of 3-cyano-6,7-diarylpyrazolo[1,5-a]pyrimidines.

Scheme 51: Synthesis of various pyrazolo[3,4-d]pyrimidine derivatives.

Scheme 52: Synthesis of hydrazinopyrazolo[3,4-d]pyrimidine derivatives.

Scheme 53: Synthesis of N-arylidinepyrazolo[3,4-d]pyrimidin-5-amines.

Scheme 54: Synthesis of pyrazolo[3,4-d]pyrimidinyl-4-amines.

Scheme 55: Iodine-catalyzed synthesis of pyrazolo[3,4-d]pyrimidinones.

Scheme 56: Synthesis of ethyl 6-amino-2H-pyrazolo[3,4-d]pyrimidine-4-carboxylate.

Scheme 57: Synthesis of 4-substituted-(3,6-dihydropyran-4-yl)-1H-pyrazolo[3,4-d]pyrimidines.

Scheme 58: Synthesis of 1-(2,4-dichlorophenyl)pyrazolo[3,4-d]pyrimidin-4-yl carboxamides.

Scheme 59: Synthesis of 5-(1,3,4-thidiazol-2-yl)pyrazolo[3,4-d]pyrimidine.

Scheme 60: One pot POCl3-catalyzed synthesis of 1-arylpyrazolo[3,4-d]pyrimidin-4-ones.

Scheme 61: Synthesis of 4-amino-N1,C3-dialkylpyrazolo[3,4-d]pyrimidines under Suzuki conditions.

Scheme 62: Microwave-assisted synthesis of pyrazolo[3,4-b]pyrazines.

Scheme 63: Synthesis and derivatization of pyrazolo[3,4-b]pyrazine-5-carbonitriles.

Scheme 64: Synthesis of 2-thioxo-pyrazolo[1,5-a][1,3,5]triazin-4-ones.

Scheme 65: Synthesis of 2,3-dihydropyrazolo[1,5-a][1,3,5]triazin-4(1H)-one.

Scheme 66: Synthesis of pyrazolo[1,5-a][1,3,5]triazine-8-carboxylic acid ethyl ester.

Scheme 67: Microwave-assisted synthesis of 4,7-dihetarylpyrazolo[1,5-a][1,3,5]triazines.

Scheme 68: Alternative synthetic route to 4,7-diheteroarylpyrazolo[1,5-a][1,3,5]triazines.

Scheme 69: Synthesis of 4-aryl-2-ethylthio-7-methylpyrazolo[1,5-a][1,3,5]triazines.

Scheme 70: Microwave-assisted synthesis of 4-aminopyrazolo[1,5-a][1,3,5]triazine.

Scheme 71: Synthesis of pyrazolo[3,4-d][1,2,3]triazines from pyrazol-5-yl diazonium salts.

Scheme 72: Synthesis of 2,5-dihydropyrazolo[3,4-e][1,2,4]triazines.

Scheme 73: Synthesis of pyrazolo[5,1-c][1,2,4]triazines via diazopyrazolylenaminones.

Scheme 74: Synthesis of pyrazolo[5,1-c][1,2,4]triazines in presence of sodium acetate.

Scheme 75: Synthesis of various 7-diazopyrazolo[5,1-c][1,2,4]triazine derivatives.

Scheme 76: One pot synthesis of pyrazolo[5,1-c][1,2,4]triazines.

Scheme 77: Synthesis of 4-amino-3,7,8-trinitropyrazolo-[5,1-c][1,2,4]triazines.

Scheme 78: Synthesis of tricyclic pyrazolo[5,1-c][1,2,4]triazines by azocoupling reaction.






