Search results
Search for "one-pot synthesis" in Full Text gives 214 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Recent advances and perspectives in ruthenium-catalyzed cyanation reactions
- Thaipparambil Aneeja,
- Cheriya Mukkolakkal Abdulla Afsina,
- Padinjare Veetil Saranya and
- Gopinathan Anilkumar
Beilstein J. Org. Chem. 2022, 18, 37–52, doi:10.3762/bjoc.18.4
- strategy. Proposed mechanistic pathway for the cyanation of the aldimine intermediate. Strecker-type functionalization of N-aryl-substituted tetrahydroisoquinolines under flow conditions. One-pot synthesis of α-aminonitriles using RuCl3 as catalyst. Synthesis of alkyl nitriles using (Ru(TMHD)3) as the

Graphical Abstract

Scheme 1: Starch-immobilized ruthenium trichloride-catalyzed cyanation of tertiary amines.

Scheme 2: Proposed mechanism for the cyanation of tertiary amines using starch-immobilized ruthenium trichlor...

Scheme 3: Cyanation of tertiary amines using heterogeneous Ru/C catalyst.

Scheme 4: Proposed mechanism for cyanation of tertiary amines using a heterogeneous Ru/C catalyst.

Scheme 5: Ruthenium-carbamato complex-catalyzed oxidative cyanation of tertiary amines.

Scheme 6: Cyanation of tertiary amines using immobilized MCM-41-2N-RuCl3 as the catalyst.

Scheme 7: Cyanation of tertiary amines using RuCl3·nH2O as the catalyst and molecular oxygen as oxidant.

Scheme 8: RuCl3-catalyzed cyanation of tertiary amines using NaCN/HCN and H2O2 as oxidant.

Scheme 9: Proposed mechanism for the ruthenium-catalyzed oxidative cyanation using H2O2.

Scheme 10: Proposed mechanism for the ruthenium-catalyzed aerobic oxidative cyanation.

Scheme 11: RuCl3-catalyzed oxidative cyanation of tertiary amines using acetone cyanohydrin as the cyanating a...

Scheme 12: Cyanation of indoles using K4[Fe(CN)6] as cyano source and Ru(III)-exchanged NaY zeolite (RuY) as c...

Scheme 13: Cyanation of arenes and heteroarenes using a ruthenium(II) catalyst and N-cyano-N-phenyl-p-toluenes...

Scheme 14: Proposed mechanism for the cyanation of arenes and heteroarenes using ruthenium(II) as catalyst and...

Scheme 15: Synthesis of N-(2-cyanoaryl)-7-azaindoles.

Figure 1: Structure of the TiO2-immobilized ruthenium polyazine complex.

Scheme 16: Visible-light-induced oxidative cyanation of aza-Baylis–Hillman adducts.

Scheme 17: Synthesis of 1° alkyl nitriles using [Ru(bpy)3](PF6)2 as the photocatalyst.

Scheme 18: Synthesis of 2° and 3° alkyl nitriles using [Ru(bpy)3](PF6)2 as the photocatalyst.

Scheme 19: Photoredox cross coupling reaction.

Scheme 20: Synthesis of α-amino nitriles from amines via a one-pot strategy.

Scheme 21: Proposed mechanistic pathway for the cyanation of the aldimine intermediate.

Scheme 22: Strecker-type functionalization of N-aryl-substituted tetrahydroisoquinolines under flow conditions....

Scheme 23: One-pot synthesis of α-aminonitriles using RuCl3 as catalyst.

Scheme 24: Synthesis of alkyl nitriles using (Ru(TMHD)3) as the catalyst.

Scheme 25: Synthesis of cyanated isoxazolines from alkenyl oximes catalyzed by [RuCl2(p-cymene)]2 in the prese...

Scheme 26: Proposed mechanism for the synthesis of cyanated isoxazolines from alkenyl oximes.

Scheme 27: Oxidative cyanation of differently substituted alcohols.
Total synthesis of the O-antigen repeating unit of Providencia stuartii O49 serotype through linear and one-pot assemblies
- Tanmoy Halder and
- Somnath Yadav
Beilstein J. Org. Chem. 2021, 17, 2915–2921, doi:10.3762/bjoc.17.199
- involving the [1 + (1 + 1 = 2)] assembly, followed by a one-pot synthesis involving [1 + 1 + 1] strategy from the corresponding monosaccharides. The one-pot method provided a higher yield of the protected trisaccharide intermediate (73%) compared to the two step synthesis (66%). The protected trisaccharide
- was then deprotected and N-acetylated to finally afford the desired trisaccharide repeating unit as its α-p-methoxyphenyl glycoside. Keywords: capsular polysaccharide; carbohydrate vaccines; O-antigen; oligosaccharide synthesis; one-pot synthesis; Introduction O-antigens or O-specific
- acceptor. We also demonstrated a follow-up one-pot synthesis involving a [1 + 1 + 1] strategy using the corresponding appropriately protected monosaccharides, providing the opportunity for rapid access to the desired target molecule. Results and Discussion The retrosynthesis of the target trisaccharide 1

Graphical Abstract

Figure 1: Structure of the repeating unit of the lipopolysaccharide of Providencia stuartii O49 serotype.

Scheme 1: Retrosynthetic analysis for the synthesis of the target trisaccharide 1.

Scheme 2: Synthesis of the monosaccharide building blocks 3, 6, and 7.

Scheme 3: Linear synthesis of trisaccharide derivative 2.

Scheme 4: Synthesis of ᴅ-galactose donor 12.

Scheme 5: One-pot synthesis of trisaccharide derivative 2.

Scheme 6: Synthesis of trisaccharide derivative 1.
A two-phase bromination process using tetraalkylammonium hydroxide for the practical synthesis of α-bromolactones from lactones
- Yuki Yamamoto,
- Akihiro Tabuchi,
- Kazumi Hosono,
- Takanori Ochi,
- Kento Yamazaki,
- Shintaro Kodama,
- Akihiro Nomoto and
- Akiya Ogawa
Beilstein J. Org. Chem. 2021, 17, 2906–2914, doi:10.3762/bjoc.17.198
- layer was washed with H2O (10 mL × 2), dried by anhydrous Na2SO4, then filtered. The filtrate was concentrated under reduced pressure. Finally, the residue was purified by silica-gel column chromatography (AcOMe/isohexane) to give pure product 5. One-pot synthesis of a functional lactone 6 using PhS−Na
- 2a. Synthesis of α-functionalized lactones using the two-phase system. Synthesis of unsymmetrically functionalized sulfide 5 via the two-phase system-promoted intramolecular cyclization of 2b. Sequential nucleophilic substitution in the two-phase system. One-pot synthesis of 2-phenylthio-α

Graphical Abstract

Scheme 1: General procedure for α-bromination of δ-valerolactone (1a) and the method described in this work.

Scheme 2: Tetraalkylammonium salt-mediated intramolecular cyclization of 2a.

Scheme 3: Synthesis of α-functionalized lactones using the two-phase system.

Scheme 4: Synthesis of unsymmetrically functionalized sulfide 5 via the two-phase system-promoted intramolecu...

Scheme 5: Sequential nucleophilic substitution in the two-phase system.

Scheme 6: One-pot synthesis of 2-phenylthio-α-valerolactone 6.
Recent advances in the syntheses of anthracene derivatives
- Giovanni S. Baviera and
- Paulo M. Donate
Beilstein J. Org. Chem. 2021, 17, 2028–2050, doi:10.3762/bjoc.17.131
- -workers reported a one-pot synthesis of substituted anthracenes 37 from o-tolualdehyde 34 and aryl iodides 35 via a palladium-catalyzed C–H arylation with a silver oxidant (Scheme 8) [42]. During optimization studies, the authors noted that steric and electronic effects strongly affected the cyclization
- reaction also included anthraquinones 173b and 173c, obtained in good yields (74–94%) [73]. In 2015, in a related approach, Lee et al. disclosed a direct one-pot synthesis of anthraquinones and tetracenediones by using ʟ-proline as organocatalyst and benzoic acid as additive (Scheme 40) [74]. They
- -trimethoxybenzaldehyde also afforded the corresponding para-adducts 189 in different proportions in the reaction mixture. As expected, when the authors used aliphatic aldehydes, they did not detect the corresponding derivatives 188 [79]. Other procedures In 2009, Naeimi and Namdari published a one-pot synthesis of

Graphical Abstract

Figure 1: Examples of anthracene derivatives and their applications.

Scheme 1: Rhodium-catalyzed oxidative coupling reactions of arylboronic acids with internal alkynes.

Scheme 2: Rhodium-catalyzed oxidative benzannulation reactions of 1-adamantoyl-1-naphthylamines with internal...

Scheme 3: Gold/bismuth-catalyzed cyclization of o-alkynyldiarylmethanes.

Scheme 4: [2 + 2 + 2] Cyclotrimerization reactions with alkynes/nitriles in the presence of nickel and cobalt...

Scheme 5: Cobalt-catalyzed [2 + 2 + 2] cyclotrimerization reactions with bis(trimethylsilyl)acetylene (23).

Scheme 6: [2 + 2 + 2] Alkyne-cyclotrimerization reactions catalyzed by a CoCl2·6H2O/Zn reagent.

Scheme 7: Pd(II)-catalyzed sp3 C–H alkenylation of diphenyl carboxylic acids with acrylates.

Scheme 8: Pd(II)-catalyzed sp3 C–H arylation with o-tolualdehydes and aryl iodides.

Scheme 9: Alkylation of arenes with aromatic aldehydes in the presence of acetyl bromide and ZnBr2/SiO2.

Scheme 10: BF3·H2O-catalyzed hydroxyalkylation of arenes with aromatic dialdehyde 44.

Scheme 11: Bi(OTf)3-promoted Friedel–Crafts alkylation of triarylmethanes and aromatic acylals and of arenes a...

Scheme 12: Reduction of anthraquinones by using Zn/pyridine or Zn/NaOH reductive methods.

Scheme 13: Two-step route to novel substituted Indenoanthracenes.

Scheme 14: Synthesis of 1,8-diarylanthracenes through Suzuki–Miyaura coupling reaction in the presence of Pd-P...

Scheme 15: Synthesis of five new substituted anthracenes by using LAH as reducing agent.

Scheme 16: One-pot procedure to synthesize substituted 9,10-dicyanoanthracenes.

Scheme 17: Reduction of bromoanthraquinones with NaBH4 in alkaline medium.

Scheme 18: In(III)-catalyzed reductive-dehydration intramolecular cycloaromatization of 2-benzylic aromatic al...

Scheme 19: Acid-catalyzed cyclization of new O-protected ortho-acetal diarylmethanols.

Scheme 20: Lewis acid-mediated regioselective cyclization of asymmetric diarylmethine dipivalates and diarylme...

Scheme 21: BF3·OEt2/CF3SO3H-mediated cyclodehydration reactions of 2-(arylmethyl)benzaldehydes and 2-(arylmeth...

Scheme 22: Synthesis of 2,3,6,7-anthracenetetracarbonitrile (90) by double Wittig reaction followed by deprote...

Scheme 23: Homo-elongation protocol for the synthesis of substituted acene diesters/dinitriles.

Scheme 24: Synthesis of two new parental BN anthracenes via borylative cyclization.

Scheme 25: Synthesis of substituted anthracenes from a bifunctional organomagnesium alkoxide.

Scheme 26: Palladium-catalyzed tandem C–H activation/bis-cyclization of propargylic carbonates.

Scheme 27: Ruthenium-catalyzed C–H arylation of acetophenone derivatives with arenediboronates.

Scheme 28: Pd-catalyzed intramolecular cyclization of (Z,Z)-p-styrylstilbene derivatives.

Scheme 29: AuCl-catalyzed double cyclization of diiodoethynylterphenyl compounds.

Scheme 30: Iodonium-induced electrophilic cyclization of terphenyl derivatives.

Scheme 31: Oxidative photocyclization of 1,3-distyrylbenzene derivatives.

Scheme 32: Oxidative cyclization of 2,3-diphenylnaphthalenes.

Scheme 33: Suzuki-Miyaura/isomerization/ring closing metathesis strategy to synthesize benz[a]anthracenes.

Scheme 34: Green synthesis of oxa-aza-benzo[a]anthracene and oxa-aza-phenanthrene derivatives.

Scheme 35: Triple benzannulation of substituted naphtalene via a 1,3,6-naphthotriyne synthetic equivalent.

Scheme 36: Zinc iodide-catalyzed Diels–Alder reactions with 1,3-dienes and aroyl propiolates followed by intra...

Scheme 37: H3PO4-promoted intramolecular cyclization of substituted benzoic acids.

Scheme 38: Palladium-catalyzed intermolecular direct acylation of aromatic aldehydes and o-iodoesters.

Scheme 39: Cycloaddition/oxidative aromatization of quinone and β-enamino esters.

Scheme 40: ʟ-Proline-catalyzed [4 + 2] cycloaddition reaction of naphthoquinones and α,β-unsaturated aldehydes....

Scheme 41: Iridium-catalyzed [2 + 2 + 2] cycloaddition of a 1,2-bis(propiolyl)benzene derivative with alkynes.

Scheme 42: Synthesis of several anthraquinone derivatives by using InCl3 and molecular iodine.

Scheme 43: Indium-catalyzed multicomponent reactions employing 2-hydroxy-1,4-naphthoquinone (186), β-naphthol (...

Scheme 44: Synthesis of substituted anthraquinones catalyzed by an AlCl3/MeSO3H system.

Scheme 45: Palladium(II)-catalyzed/visible light-mediated synthesis of anthraquinones.

Scheme 46: [4 + 2] Anionic annulation reaction for the synthesis of substituted anthraquinones.
Progress and challenges in the synthesis of sequence controlled polysaccharides
- Giulio Fittolani,
- Theodore Tyrikos-Ergas,
- Denisa Vargová,
- Manishkumar A. Chaube and
- Martina Delbianco
Beilstein J. Org. Chem. 2021, 17, 1981–2025, doi:10.3762/bjoc.17.129


Graphical Abstract

Figure 1: Overview of the methods available for the synthesis of polysaccharides. For each method, advantages...

Figure 2: Overview of the classes of polysaccharides discussed in this review. Each section deals with polysa...

Scheme 1: Enzymatic and chemical polymerization approaches provide cellulose oligomers with a non-uniform dis...

Scheme 2: AGA of a collection of cellulose analogues obtained using BBs 6–9. Specifically placed modification...

Figure 3: Chemical structure of the different branches G, X, L, F commonly found in XGs. Names are given foll...

Scheme 3: AGA of XG analogues with defined side chains. The AGA cycle includes coupling (TMSOTf), Fmoc deprot...

Figure 4: Synthetic strategies and issues associated to the formation of the β(1–3) linkage.

Scheme 4: Convergent synthesis of β(1–3)-glucans using a regioselective glycosylation strategy.

Scheme 5: DMF-mediated 1,2-cis glycosylation. A) General mechanism and B) examples of α-glucans prepared usin...

Scheme 6: Synergistic glycosylation strategy employing a nucleophilic modulation strategy (TMSI and Ph3PO) in...

Scheme 7: Different approaches to produce xylans. A) Polymerization techniques including ROP, and B) enzymati...

Scheme 8: A) Synthesis of arabinofuranosyl-decorated xylan oligosaccharides using AGA. Representative compoun...

Scheme 9: Chemoenzymatic synthesis of COS utilizing a lysozyme-catalyzed transglycosylation reaction followed...

Scheme 10: Synthesis of COS using an orthogonal glycosylation strategy based on the use of two different LGs.

Scheme 11: Orthogonal N-PGs permitted the synthesis of COS with different PA.

Scheme 12: AGA of well-defined COS with different PA using two orthogonally protected BBs. The AGA cycle inclu...

Scheme 13: A) AGA of β(1–6)-N-acetylglucosamine hexasaccharide and dodecasaccharide. AGA includes cycles of co...

Figure 5: ‘Double-faced’ chemistry exemplified for ᴅ-Man and ʟ-Rha. Constructing β-Man linkages is considerab...

Figure 6: Implementation of a capping step after each glycosylation cycle for the AGA of a 50mer oligomannosi...

Scheme 14: AGA enabled the synthesis of a linear α(1–6)-mannoside 100mer 93 within 188 h and with an average s...

Scheme 15: The 151mer branched polymannoside was synthesized by a [30 + 30 + 30 + 30 + 31] fragment coupling. ...

Figure 7: PG stereocontrol strategy to obtain β-mannosides. A) The mechanism of the β-mannosylation reaction ...

Scheme 16: A) Mechanism of 1,2-cis stereoselective glycosylation using ManA donors. Once the ManA donor is act...

Figure 8: A) The preferred 4H3 conformation of the gulosyl oxocarbenium ion favors the attack of the alcohol ...

Scheme 17: AGA of type I rhamnans up to 16mer using disaccharide BB 115 and CNPiv PG. The AGA cycle includes c...

Figure 9: Key BBs for the synthesis of the O-antigen of Bacteroides vulgatus up to a 128mer (A) and the CPS o...

Figure 10: Examples of type I and type II galactans synthesized to date.

Figure 11: A) The DTBS PG stabilizes the 3H4 conformation of the Gal oxocarbenium ion favoring the attack of t...

Figure 12: Homogalacturonan oligosaccharides synthesized to date. Access to different patterns of methyl-ester...

Figure 13: GlfT2 from Mycobacterium tuberculosis catalyzes the sequential addition of UPD-Galf donor to a grow...

Figure 14: The poor reactivity of acceptor 137 hindered a stepwise synthesis of the linear galactan backbone a...

Scheme 18: AGA of a linear β(1–5) and β(1–6)-linked galactan 20mer. The AGA cycle includes coupling (NIS/TfOH)...

Figure 15: The 92mer arabinogalactan was synthesized using a [31 + 31 + 30] fragment coupling between a 31mer ...

Scheme 19: Synthesis of the branched arabinofuranose fragment using a six component one-pot synthesis. i) TTBP...

Figure 16: A) Chemical structure and SNFG of the representative disaccharide units forming the GAG backbones, ...

Figure 17: Synthetic challenges associated to the H/HS synthesis.

Scheme 20: Degradation of natural heparin and heparosan generated valuable disaccharides 150 and 151 that can ...

Scheme 21: A) The one-step conversion of cyanohydrin 156 to ʟ-iduronamide 157 represent the key step for the s...

Scheme 22: A) Chemoenzymatic synthesis of heparin structures, using different types of UDP activated natural a...

Scheme 23: Synthesis of the longest synthetic CS chain 181 (24mer) using donor 179 and acceptor 180 in an iter...

Scheme 24: AGA of a collection of HA with different lengths. The AGA cycle includes coupling (TfOH) and Lev de...
Asymmetric organocatalyzed synthesis of coumarin derivatives
- Natália M. Moreira,
- Lorena S. R. Martelli and
- Arlene G. Corrêa
Beilstein J. Org. Chem. 2021, 17, 1952–1980, doi:10.3762/bjoc.17.128
- obtained with good to excellent yields and enantiomeric excesses. Besides, the one-pot synthesis of coumarins followed by the Michael addition step was proven to be a good alternative, affording the desired product with excellent yield and ee. The applicability of the methodology was also demonstrated by a
- intermediate and the coumarin through hydrogen bonding, as shown in Scheme 25. An enantioselective one-pot synthesis of spiro[dihydrofurocoumarin/pyrazolone] 83 mediated by quinine and squaramide catalyst 84 was reported by Xu et al. [61]. The work draws attention for the wide range of compounds obtained with

Graphical Abstract

Figure 1: Coumarin-derived commercially available drugs.

Figure 2: Inhibition of acetylcholinesterase by coumarin derivatives.

Scheme 1: Michael addition of 4-hydroxycoumarins 1 to α,β‐unsaturated enones 2.

Scheme 2: Organocatalytic conjugate addition of 4-hydroxycoumarin 1 to α,β-unsaturated aldehydes 2 followed b...

Scheme 3: Synthesis of 3,4-dihydrocoumarin derivatives 10 through decarboxylative and dearomatizative cascade...

Scheme 4: Total synthesis of (+)-smyrindiol (17).

Scheme 5: Michael addition of 4-hydroxycoumarin (1) to enones 2 through a bifunctional modified binaphthyl or...

Scheme 6: Michael addition of ketones 20 to 3-aroylcoumarins 19 using a cinchona alkaloid-derived primary ami...

Scheme 7: Enantioselective reaction of cyclopent-2-enone-derived MBH alcohols 24 with 4-hydroxycoumarins 1.

Scheme 8: Sequential Michael addition/hydroalkoxylation one-pot approach to annulated coumarins 28 and 30.

Scheme 9: Michael addition of 4-hydroxycoumarins 1 to enones 2 using a binaphthyl diamine catalyst 31.

Scheme 10: Asymmetric Michael addition of 4-hydroxycoumarin 1 with α,β-unsaturated ketones 2 catalyzed by a ch...

Scheme 11: Catalytic asymmetric β-C–H functionalization of ketones via enamine oxidation.

Scheme 12: Enantioselective synthesis of polycyclic coumarin derivatives 37 catalyzed by an primary amine-imin...

Scheme 13: Allylic alkylation reaction between 3-cyano-4-methylcoumarins 39 and MBH carbonates 40.

Scheme 14: Enantioselective synthesis of cyclopropa[c]coumarins 45.

Scheme 15: NHC-catalyzed lactonization of 2-bromoenals 46 with 4-hydroxycoumarin (1).

Scheme 16: NHC-catalyzed enantioselective synthesis of dihydrocoumarins 51.

Scheme 17: Domino reaction of enals 2 with hydroxylated malonate 53 catalyzed by NHC 55.

Scheme 18: Oxidative [4 + 2] cycloaddition of enals 57 to coumarins 56 catalyzed by NHC 59.

Scheme 19: Asymmetric [3 + 2] cycloaddition of coumarins 43 to azomethine ylides 60 organocatalyzed by quinidi...

Scheme 20: Synthesis of α-benzylaminocoumarins 64 through Mannich reaction between 4-hydroxycoumarins (1) and ...

Scheme 21: Asymmetric addition of malonic acid half-thioesters 67 to coumarins 66 using the sulphonamide organ...

Scheme 22: Enantioselective 1,4-addition of azadienes 71 to 3-homoacyl coumarins 70.

Scheme 23: Michael addition/intramolecular cyclization of 3-acylcoumarins 43 to 3-halooxindoles 74.

Scheme 24: Enantioselective synthesis of 3,4-dihydrocoumarins 78 catalyzed by squaramide 73.

Scheme 25: Organocatalyzed [4 + 2] cycloaddition between 2,4-dienals 79 and 3-coumarincarboxylates 43.

Scheme 26: Enantioselective one-pot Michael addition/intramolecular cyclization for the synthesis of spiro[dih...

Scheme 27: Michael/hemiketalization addition enantioselective of hydroxycoumarins (1) to: (a) enones 2 and (b)...

Scheme 28: Synthesis of 2,3-dihydrofurocoumarins 89 through Michael addition of 4-hydroxycoumarins 1 to β-nitr...

Scheme 29: Synthesis of pyrano[3,2-c]chromene derivatives 93 via domino reaction between 4-hydroxycoumarins (1...

Scheme 30: Conjugated addition of 4-hydroxycoumarins 1 to nitroolefins 95.

Scheme 31: Michael addition of 4-hydroxycoumarin 1 to α,β-unsaturated ketones 2 promoted by primary amine thio...

Scheme 32: Enantioselective synthesis of functionalized pyranocoumarins 99.

Scheme 33: 3-Homoacylcoumarin 70 as 1,3-dipole for enantioselective concerted [3 + 2] cycloaddition.

Scheme 34: Synthesis of warfarin derivatives 107 through addition of 4-hydroxycoumarins 1 to β,γ-unsaturated α...

Scheme 35: Asymmetric multicatalytic reaction sequence of 2-hydroxycinnamaldehydes 109 with 4-hydroxycoumarins ...

Scheme 36: Mannich asymmetric addition of cyanocoumarins 39 to isatin imines 112 catalyzed by the amide-phosph...

Scheme 37: Enantioselective total synthesis of (+)-scuteflorin A (119).
Development of N-F fluorinating agents and their fluorinations: Historical perspective
- Teruo Umemoto,
- Yuhao Yang and
- Gerald B. Hammond
Beilstein J. Org. Chem. 2021, 17, 1752–1813, doi:10.3762/bjoc.17.123


Graphical Abstract

Scheme 1: Fluorination with N-F amine 1-1.

Scheme 2: Preparation of N-F amine 1-1.

Scheme 3: Reactions of N-F amine 1-1.

Scheme 4: Synthesis of N-F perfluoroimides 2-1 and 2-2.

Scheme 5: Synthesis of 1-fluoro-2-pyridone (3-1).

Scheme 6: Fluorination with 1-fluoro-2-pyridone (3-1).

Figure 1: Synthesis of N-F sulfonamides 4-1a–g.

Scheme 7: Fluorination with N-F reagent 4-1b,c,f.

Scheme 8: Fluorination of alkenyllithiums with N-F 4-1h.

Scheme 9: Synthesis of N-fluoropyridinium triflate (5-4a).

Scheme 10: Synthetic methods for N-F-pyridinium salts.

Figure 2: Synthesis of various N-fluoropyridinium salts. Note: athis yield was the one by the improved method...

Scheme 11: Fluorination power order of N-fluoropyridinium salts.

Scheme 12: Fluorinations with N-F salts 5-4.

Scheme 13: Fluorination of Corey lactone 5-7 with N-F-bis(methoxymethyl) salt 5-4l.

Scheme 14: Fluorination with NFPy.

Scheme 15: Synthesis of the N-F reagent, N-fluoroquinuclidinium fluoride (6-1).

Scheme 16: Fluorinations achieved with N-F fluoride 6-1.

Scheme 17: Synthesis of N-F imides 7-1a–g.

Scheme 18: Fluorination with (CF3SO2)2NF, 7-1a.

Scheme 19: Fluorination reactions of various substrates with 7-1a.

Scheme 20: Synthesis of N-F triflate 8-1.

Scheme 21: Synthesis of chiral N-fluoro sultams 9-1 and 9-2.

Scheme 22: Fluorination with chiral N-fluoro sultams 9-1 and 9-2.

Scheme 23: Synthesis of saccharin-derived N-fluorosultam 10-2.

Scheme 24: Fluorination with N-fluorosultam 10-2.

Scheme 25: Synthesis of N-F reagent 11-2.

Scheme 26: Fluorination with N-F reagent 11-2.

Scheme 27: Synthesis and reaction of N-fluorolactams 12-1.

Scheme 28: Synthesis of NFOBS 13-2.

Scheme 29: Fluorination with NFOBS 13-2.

Scheme 30: Synthesis of NFSI (14-2).

Scheme 31: Fluorination with NFSI 14-2.

Scheme 32: Synthesis of N-fluorosaccharin (15-1) and N-fluorophthalimide (15-2).

Scheme 33: Synthesis of N-F salts 16-3.

Scheme 34: Fluorination with N-F salts 16-3.

Figure 3: Monofluorination with Selectfluor (16-3a).

Figure 4: Difluorination with Selectfluor (16-3a).

Scheme 35: Transfer fluorination of Selectfluor (16-3a).

Scheme 36: Fluorination of substrates with Selectfluor (16-3a).

Scheme 37: Synthesis of chiral N-fluoro-sultam 17-2.

Scheme 38: Asymmetric fluorination with chiral 17-2.

Figure 5: Synthesis of Zwitterionic N-fluoropyridinium salts 18-2a–h.

Scheme 39: Fluorinating power order of zwitterionic N-fluoropyridinium salts.

Scheme 40: Fluorination with zwitterionic 18-2.

Scheme 41: Activation of salt 18-2h with TfOH.

Scheme 42: Synthesis of NFTh, 19-2.

Scheme 43: Fluorination with NFTh, 19-2.

Scheme 44: Synthesis of 3-fluorobenzo-1,2,3-oxathiazin-4-one 2,2-dioxide (20-2).

Scheme 45: Fluorination with 20-2.

Scheme 46: Synthesis of N-F amide 21-3.

Scheme 47: Fluorination with N-F amide 21-2.

Scheme 48: Synthesis of N,N’-difluorodiazoniabicyclo[2.2.2]octane salts 22-1.

Scheme 49: One-pot synthesis of N,N’-difluoro-1,4-diazoniabicyclo[2.2.2]octane bistetrafluoroborate salt (22-1d...

Figure 6: Fluorination of anisole with 22-1a, d, e.

Scheme 50: Fluorination with N,N’-diF bisBF4 22-1d.

Scheme 51: Synthesis of bis-N-F reagents 23-1–5.

Scheme 52: Fluorination with 23-2, 4, 5.

Figure 7: Synthesis of N,N’-difluorobipyridinium salts 24-2.

Figure 8: Controlled fluorination of N,N’-diF 24-2.

Scheme 53: Fluorinating power of N,N’-diF salts 24-2 and N-F salt 5-4a.

Scheme 54: Fluorination reactions with SynfluorTM (24-2b).

Scheme 55: Additional fluorination reactions with SynfluorTM (24-2b).

Scheme 56: Synthesis of N-F 25-1.

Scheme 57: Fluorination of polycyclic aromatics with 25-1.

Scheme 58: Synthesis of 26-1 and dimethyl analog 26-2.

Scheme 59: Fluorination with reagents 26-1, 26-2, 1-1, and 26-3.

Scheme 60: Synthesis of N-F reagent 27-2.

Scheme 61: Synthesis of chiral N-F reagents 27-6.

Scheme 62: Synthesis of chiral N-F 27-7–9.

Scheme 63: Asymmetric fluorination with 27-6.

Scheme 64: Synthesis of chiral N-F reagents 28-3.

Scheme 65: Asymmetric fluorination with 28-3.

Scheme 66: Synthesis of chiral N-F reagents 28-7.

Figure 9: Asymmetric fluorination with 28-7.

Scheme 67: In situ formation of N-fluorinated cinchona alkaloids with SelectfluorTM.

Scheme 68: Asymmetric fluorination with N-F alkaloids formed in situ.

Scheme 69: Synthesis of N-fluorocinchona alkaloids with Selectfluor.

Scheme 70: Asymmetric fluorination with 30-1–4.

Scheme 71: Transfer fluorination from various N-F reagents.

Figure 10: Asymmetric fluorination of silyl enol ethers.

Scheme 72: Synthesis of N-fluoro salt 32-2.

Scheme 73: Reactivity of N-fluorotriazinium salt 32-2.

Scheme 74: Synthesis of bulky N-fluorobenzenesulfonimide NFBSI 33-3.

Scheme 75: Comparison of NFSI and NFBSI.

Scheme 76: Synthesis of p-substituted N-fluorobenzenesulfonimides 34-3.

Figure 11: Asymmetric fluorination with 34-3 and a chiral catalyst 34-4.

Scheme 77: 1,4-Fluoroamination with Selecfluor and a chiral catalyst.

Figure 12: Asymmetric fluoroamination with 35-5a, b.

Scheme 78: Synthesis of Selectfluor analogs 35-5a, b.

Scheme 79: Synthesis of chiral dicationic DABCO-based N-F reagents 36-5.

Scheme 80: Asymmetric fluorocyclization with chiral 36-5b.

Scheme 81: Synthesis of chiral 37-2a,b.

Scheme 82: Asymmetric fluorination with chiral 37-2a,b.

Scheme 83: Asymmetric fluorination with chiral 37-2b.

Scheme 84: Reaction of indene with chiral 37-2a,b.

Scheme 85: Synthesis of Me-NFSI, 38-2.

Scheme 86: Fluorination of active methine compounds with Me-NFSI.

Scheme 87: Fluorination of malonates with Me-NFSI.

Scheme 88: Fluorination of keto esters with Me-NFSI.

Scheme 89: Synthesis of N-F 39-3 derived from the ethylene-bridged Tröger’s base.

Scheme 90: Fluorine transfer from N-F 39-3.

Scheme 91: Fluorination with N-F 39-3.

Scheme 92: Synthesis of SelectfluorCN.

Scheme 93: Bistrifluoromethoxylation of alkenes using SelectfluorCN.

Figure 13: Synthesis of NFAS 41-2.

Scheme 94: Radical fluorination with different N-F reagents.

Scheme 95: Radical fluorination of alkenes with NFAS 41-2.

Scheme 96: Radical fluorination of alkenes with NFAS 41-2f.

Scheme 97: Decarboxylative fluorination with NFAS 41-2a,f.

Scheme 98: Fluorine plus detachment (FPD).

Figure 14: FPD values of representative N-F reagents in CH2Cl2 and CH3CN (in parentheses). Adapted with permis...

Scheme 99: N-F homolytic bond dissociation energy (BDE).

Figure 15: BDE values of representative N-F reagents in CH3CN. Adapted with permission from ref. [127]. Copyright 2...

Figure 16: Quantitative reactivity scale for popular N-F reagents. Adapted with permission from ref. [138], publish...

Scheme 100: SET and SN2 mechanisms.

Scheme 101: Radical clock reactions.

Scheme 102: Reaction of potassium enolate of citronellic ester with N-F reagents, 10-1, NFSI, and 8-1.

Scheme 103: Reaction of compound IV with Selectfluor (OTf) and NFSI.

Scheme 104: Reaction of TEMPO with Selecfluor.
A recent overview on the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles
- Pezhman Shiri,
- Ali Mohammad Amani and
- Thomas Mayer-Gall
Beilstein J. Org. Chem. 2021, 17, 1600–1628, doi:10.3762/bjoc.17.114
- (hetero)arylboronic acid 111 was reported. The use of a catalytic amount of CuCl and MeOLi as base in CH3CN as solvent was recognized as the optimized conditions. This strategy provided a new procedure for the one-pot synthesis of fully decorated 1,2,3-triazole derivatives 112 in good to excellent yield

Graphical Abstract

Figure 1: Some significant triazole derivatives [8,23-27].

Scheme 1: A general comparison between synthetic routes for disubstituted 1,2,3-triazole derivatives and full...

Scheme 2: Synthesis of formyltriazoles 3 from the treatment of α-bromoacroleins 1 with azides 2.

Scheme 3: A probable mechanism for the synthesis of formyltriazoles 5 from the treatment of α-bromoacroleins 1...

Scheme 4: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 8 from the reaction of aryl azides 7 with enamino...

Scheme 5: Proposed mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles from the reaction of a...

Scheme 6: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 11 from the reaction of primary amines 10 with 1,...

Scheme 7: The proposed mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 11 from the reacti...

Scheme 8: Synthesis of fully decorated 1,2,3-triazoles 19 containing a sulfur-based side chain.

Scheme 9: Mechanism for the formation of fully decorated 1,2,3-triazoles 19 containing a sulfur-based side ch...

Scheme 10: Synthesis of fully decorated 1,2,3-triazole compounds 25 through the regioselective addition and cy...

Scheme 11: A reasonable mechanism for the synthesis of fully decorated 1,2,3-triazole compounds 25 through the...

Scheme 12: Synthesis of 1,4,5-trisubstituted glycosyl-containing 1,2,3-triazole derivatives 30 from the reacti...

Scheme 13: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 34 via intramolecular cyclization reaction of ket...

Scheme 14: Synthesis of fully decorated 1,2,3-triazoles 38 from the reaction of aldehydes 35, amines 36, and α...

Scheme 15: A reasonable mechanism for the synthesis of fully decorated 1,2,3-triazoles 38 from the reaction of...

Scheme 16: Synthesis of functionally rich double C- and N-vinylated 1,2,3-triazoles 45 and 47.

Scheme 17: Synthesis of disubstituted 4-chloro-, 4-bromo-, and 4-iodo-1,2,3-triazoles 50.

Scheme 18: a) A general route for SPAAC in polymer chemistry and b) synthesis of a novel pH-sensitive polymeri...

Scheme 19: Synthesis of 5-allenyl-1,2,3-triazoles 60 by the treatment of alkynes 57, azides 58, and propargyli...

Scheme 20: A reasonable mechanism for the synthesis of 5-allenyl-1,2,3-triazoles 60 by the treatment of alkyne...

Scheme 21: Synthesis of 5‐alkynyl-1,2,3-triazoles 69.

Scheme 22: A reasonable mechanism for the synthesis of 5‐alkynyl-1,2,3-triazoles 69.

Scheme 23: Synthesis of sulfur-cycle-fused 1,2,3-triazoles 75 and 77.

Scheme 24: A reasonable mechanism for the synthesis of sulfur-cycle-fused 1,2,3‐triazoles 75 and 77.

Scheme 25: Synthesis of 5-selanyltriazoles 85 from the reaction of ethynylstibanes 82, organic azides 83, and ...

Scheme 26: A mechanism for the synthesis of 5-selanyltriazoles 85 from the reaction of ethynylstibanes 82, org...

Scheme 27: Synthesis of trisubstituted triazoles containing an Sb substituent at position C5 in 93 and 5-unsub...

Scheme 28: Synthesis of asymmetric triazole disulfides 98 from disulfide-containing tert-butyltosyl disulfide 97...

Scheme 29: A mechanism for the synthesis of asymmetric triazole disulfides 98 from disulfide-containing tert-bu...

Scheme 30: Synthesis of triazole-fused sultams 104.

Scheme 31: Synthesis of 1,2,3-triazole-fused tricyclic heterocycles 106.

Scheme 32: A reasonable mechanism for the synthesis of 1,2,3-triazole-fused tricyclic heterocycles 106.

Scheme 33: Synthesis of 5-aryl-substituted 1,2,3-triazole derivatives 112.

Scheme 34: A reasonable mechanism for the synthesis of 5-aryl-substituted 1,2,3-triazole derivatives 112.

Scheme 35: Synthesis of 1,4,5-trisubstituted 1,2,3-triazole-5-carboxamides 119.

Scheme 36: A probable mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazole-5-carboxamides 119.

Scheme 37: Synthesis of fully decorated triazoles 125 via the Pd/C-catalyzed arylation of disubstituted triazo...

Scheme 38: Synthesis of triazolo[1,5-a]indolones 131.

Scheme 39: Synthesis of unsymmetrically substituted triazole-fused enediyne systems 135 and 5-aryl-4-ethynyltr...

Scheme 40: Synthesis of Pd/Cu-BNP 139 and application of 139 in the synthesis of polycyclic triazoles 142.

Scheme 41: A probable mechanism for the synthesis of polycyclic triazoles 142.

Scheme 42: Synthesis of highly functionalized 1,2,3-triazole-fused 5-, 6-, and 7-membered rings 152–154.

Scheme 43: A probable mechanism for the synthesis of highly functionalized 1,2,3-triazole-fused 5-, 6-, and 7-...

Scheme 44: Synthesis of fully functionalized 1,2,3-triazolo-fused chromenes 162, 164, and 166 via the intramol...

Scheme 45: Ru-catalyzed synthesis of fully decorated triazoles 172.

Scheme 46: Synthesis of 4-cyano-1,2,3-triazoles 175.

Scheme 47: Synthesis of functionalized triazoles from the reaction of 1-alkyltriazenes 176 and azides 177 and ...

Scheme 48: Mechanism for the synthesis of functionalized triazoles from the reaction of 1-alkyltriazenes 176 a...
Synthesis of functionalized imidazo[4,5-e]thiazolo[3,2-b]triazines by condensation of imidazo[4,5-e]triazinethiones with DMAD or DEAD and rearrangement to imidazo[4,5-e]thiazolo[2,3-c]triazines
- Alexei N. Izmest’ev,
- Dmitry B. Vinogradov,
- Natalya G. Kolotyrkina,
- Angelina N. Kravchenko and
- Galina A. Gazieva
Beilstein J. Org. Chem. 2021, 17, 1141–1148, doi:10.3762/bjoc.17.87
- imidazo[4,5-e]thiazolo[3,2-b]triazines 4a–n into imidazo[4,5-e]thiazolo[2,3-c]triazines 5a–n. One-pot synthesis of compounds 5a,b,h,i from imidazo[4,5-e]triazines 3a,b. Plausible mechanism of the formation and the rearrangement of compounds 4 into isomers 5. Results for the screening of the reaction

Graphical Abstract

Figure 1: Biologically active compounds having thiazolidin-4-one and thiazolo-1,2,4-triazine units.

Scheme 1: Regioselectivity of the cyclization of 3-thioxo-1,2,4-triazin-5-ones 1 with DMAD (A) and general co...

Scheme 2: Synthesis of imidazo[4,5-e]thiazolo[3,2-b]triazine derivatives 4a–n by the reaction of imidazo[4,5-...

Scheme 3: Reaction of imidazo[4,5-e]thiazolo[3,2-b]triazine 4h with aqueous KOH.

Scheme 4: Rearrangement of imidazo[4,5-e]thiazolo[3,2-b]triazines 4a–n into imidazo[4,5-e]thiazolo[2,3-c]tria...

Scheme 5: One-pot synthesis of compounds 5a,b,h,i from imidazo[4,5-e]triazines 3a,b.

Scheme 6: Plausible mechanism of the formation and the rearrangement of compounds 4 into isomers 5.

Figure 2: 1H NMR spectra of compounds 4h and 5h in DMSO-d6 in the region of 3.5–8.8 ppm.

Figure 3: X-ray crystal structure of compound 5i.
Prins cyclization-mediated stereoselective synthesis of tetrahydropyrans and dihydropyrans: an inspection of twenty years
- Asha Budakoti,
- Pradip Kumar Mondal,
- Prachi Verma and
- Jagadish Khamrai
Beilstein J. Org. Chem. 2021, 17, 932–963, doi:10.3762/bjoc.17.77
- allylisilane 152. Further manipulation of functional groups of 153 leads to the synthesis of (−)-dactyloide (Scheme 36) [71]. The one-pot synthesis of a 2,6-disubstituted THP was reported by Minehan and co-workers and involved treating 3-iodo-2-[(trimethylsilyl)methyl]propene with an aldehyde in the presence
- Alder’s chair-like transition state 227 in which the (Z)-alkene accounts for the trans-stereocontrol at the C3 position and equatorial iodide addition accounts for the cis-stereocontrol at the C4 position, as shown below in Scheme 54. The one-pot synthesis of tetrahydropyran by utilizing the Babier–Prins

Graphical Abstract

Scheme 1: General strategy for the synthesis of THPs.

Scheme 2: Developments towards the Prins cyclization.

Scheme 3: General stereochemical outcome of the Prins cyclization.

Scheme 4: Regioselectivity in the Prins cyclization.

Scheme 5: Mechanism of the oxonia-Cope reaction in the Prins cyclization.

Scheme 6: Cyclization of electron-deficient enantioenriched alcohol 27.

Scheme 7: Partial racemization through 2-oxonia-Cope allyl transfer.

Scheme 8: Partial racemization by reversible 2-oxonia-Cope rearrangement.

Scheme 9: Rychnovsky modification of the Prins cyclization.

Scheme 10: Synthesis of (−)-centrolobine and the C22–C26 unit of phorboxazole A.

Scheme 11: Axially selective Prins cyclization by Rychnovsky et al.

Scheme 12: Mechanism for the axially selectivity Prins cyclization.

Scheme 13: Mukaiyama aldol–Prins cyclization reaction.

Scheme 14: Application of the aldol–Prins reaction.

Scheme 15: Hart and Bennet's acid-promoted Prins cyclization.

Scheme 16: Tetrahydropyran core of polycarvernoside A as well as (−)-clavoslide A and D.

Scheme 17: Scheidt and co-workers’ route to tetrahydropyran-4-one.

Scheme 18: Mechanism for the Lewis acid-catalyzed synthesis of tetrahydropyran-4-one.

Scheme 19: Hoveyda and co-workers’ strategy for 2,6-disubstituted 4-methylenetetrahydropyran.

Scheme 20: Funk and Cossey’s ene-carbamates strategy.

Scheme 21: Yadav and Kumar’s cyclopropane strategy for THP synthesis.

Scheme 22: 2-Arylcylopropylmethanolin in centrolobine synthesis.

Scheme 23: Yadav and co-workers’ strategy for the synthesis of THP.

Scheme 24: Yadav and co-workers’ Prins–Ritter reaction sequence for 4-amidotetrahydropyran.

Scheme 25: Yadav and co-workers’ strategy to prelactones B, C, and V.

Scheme 26: Yadav and co-workers’ strategy for the synthesis of (±)-centrolobine.

Scheme 27: Loh and co-workers’ strategy for the synthesis of zampanolide and dactylolide.

Scheme 28: Loh and Chan’s strategy for THP synthesis.

Scheme 29: Prins cyclization of cyclohexanecarboxaldehyde.

Scheme 30: Prins cyclization of methyl ricinoleate (127) and benzaldehyde (88).

Scheme 31: AlCl3-catalyzed cyclization of homoallylic alcohol 129 and aldehyde 130.

Scheme 32: Martín and co-workers’ stereoselective approach for the synthesis of highly substituted tetrahydrop...

Scheme 33: Ene-IMSC strategy by Marko and Leroy for the synthesis of tetrahydropyran.

Scheme 34: Marko and Leroy’s strategy for the synthesis of tetrahydropyrans 146.

Scheme 35: Sakurai dimerization/macrolactonization reaction for the synthesis of cyanolide A.

Scheme 36: Hoye and Hu’s synthesis of (−)-dactyloide by intramolecular Sakurai cyclization.

Scheme 37: Minehan and co-workers’ strategy for the synthesis of THPs 157.

Scheme 38: Yu and co-workers’ allylic transfer strategy for the construction of tetrahydropyran 161.

Scheme 39: Reactivity enhancement in intramolecular Prins cyclization.

Scheme 40: Floreancig and co-workers’ Prins cyclization strategy to (+)-dactyloide.

Scheme 41: Panek and Huang’s DHP synthesis from crotylsilanes: a general strategy.

Scheme 42: Panek and Huang’s DHP synthesis from syn-crotylsilanes.

Scheme 43: Panek and Huang’s DHP synthesis from anti-crotylsilanes.

Scheme 44: Roush and co-workers’ [4 + 2]-annulation strategy for DHP synthesis [82].

Scheme 45: TMSOTf-promoted annulation reaction.

Scheme 46: Dobb and co-workers’ synthesis of DHP.

Scheme 47: BiBr3-promoted tandem silyl-Prins reaction by Hinkle et al.

Scheme 48: Substrate scope of Hinkle and co-workers’ strategy.

Scheme 49: Cho and co-workers’ strategy for 2,6 disubstituted 3,4-dimethylene-THP.

Scheme 50: Furman and co-workers’ THP synthesis from propargylsilane.

Scheme 51: THP synthesis from silyl enol ethers.

Scheme 52: Rychnovsky and co-workers’ strategy for THP synthesis from hydroxy-substituted silyl enol ethers.

Scheme 53: Li and co-workers’ germinal bissilyl Prins cyclization strategy to (−)-exiguolide.

Scheme 54: Xu and co-workers’ hydroiodination strategy for THP.

Scheme 55: Wang and co-workers’ strategy for tetrahydropyran synthesis.

Scheme 56: FeCl3-catalyzed synthesis of DHP from alkynylsilane alcohol.

Scheme 57: Martín, Padrón, and co-workers’ proposed mechanism of alkynylsilane Prins cyclization for the synth...

Scheme 58: Marko and co-workers’ synthesis of 2,6-anti-configured tetrahydropyran.

Scheme 59: Loh and co-workers’ strategy for 2,6-syn-tetrahydropyrans.

Scheme 60: Loh and co-workers’ strategy for anti-THP synthesis.

Scheme 61: Cha and co-workers’ strategy for trans-2,6-tetrahydropyran.

Scheme 62: Mechanism proposed by Cha et al.

Scheme 63: TiCl4-mediated cyclization to trans-THP.

Scheme 64: Feng and co-workers’ FeCl3-catalyzed Prins cyclization strategy to 4-hydroxy-substituted THP.

Scheme 65: Selectivity profile of the Prins cyclization under participation of an iron ligand.

Scheme 66: Sequential reactions involving Prins cyclization.

Scheme 67: Banerjee and co-workers’ strategy of Prins cyclization from cyclopropane carbaldehydes and propargy...

Scheme 68: Mullen and Gagné's (R)-[(tolBINAP)Pt(NC6F5)2][SbF6]2-catalyzed asymmetric Prins cyclization strateg...

Scheme 69: Yu and co-workers’ DDQ-catalyzed asymmetric Prins cyclization strategy to trisubstituted THPs.

Scheme 70: Lalli and Weghe’s chiral-Brønsted-acid- and achiral-Lewis-acid-promoted asymmetric Prins cyclizatio...

Scheme 71: List and co-workers’ iIDP Brønsted acid-promoted asymmetric Prins cyclization strategy.

Scheme 72: Zhou and co-workers’ strategy for chiral phosphoric acid (CPA)-catalyzed cascade Prins cyclization.

Scheme 73: List and co-workers’ approach for asymmetric Prins cyclization using chiral imidodiphosphoric acid ...
Deoxygenative C2-heteroarylation of quinoline N-oxides: facile access to α-triazolylquinolines
- Geetanjali S. Sontakke,
- Rahul K. Shukla and
- Chandra M. R. Volla
Beilstein J. Org. Chem. 2021, 17, 485–493, doi:10.3762/bjoc.17.42
- isoquinolines was achieved from readily available N-oxides and N-sulfonyl-1,2,3-triazoles. A variety of α-triazolylquinoline derivatives were synthesized with good regioselectivity and in excellent yields under mild reaction conditions. Further, a gram-scale and one-pot synthesis illustrated the efficacy and
- derivatives, we performed a sequential one-pot synthesis by combining a Cu(I)-catalyzed “Click” reaction of phenylacetylene (7) with TsN3 and a metal-free C2-heteroarylation of quinoline N-oxide (1a, Scheme 6b). Remarkably, the yield of the desired product 3a in the one-pot synthesis (80%) was comparable to
- isoquinoline N-oxide. Gram-scale and one-pot synthesis. Proposed mechanism. Optimization of the reaction conditions.a Supporting Information Supporting Information File 220: Experimental details. Funding The activity was generously supported by Science and Engineering Research Board (SERB), India: CRG/2019

Graphical Abstract

Figure 1: Bioactive molecules containing the 2-aminoquinoline motif.

Scheme 1: C2-selective C–N bond formation of N-oxides.

Scheme 2: Substrate scope of N-sulfonyl-1,2,3-triazoles. Reaction conditions: 1a (0.2 mmol), 2 (0.24 mmol) an...

Scheme 3: Substrate scope of quinoline N-oxides. Reaction conditions: 1 (0.2 mmol), 2a (0.24 mmol) and DCE (2...

Scheme 4: Late-stage modification of natural products.

Scheme 5: Substrate scope for the reaction of substituted triazoles with isoquinoline N-oxide.

Scheme 6: Gram-scale and one-pot synthesis.

Scheme 7: Proposed mechanism.
Decarboxylative trifluoromethylthiolation of pyridylacetates
- Ryouta Kawanishi,
- Kosuke Nakada and
- Kazutaka Shibatomi
Beilstein J. Org. Chem. 2021, 17, 229–233, doi:10.3762/bjoc.17.23
- lithium pyridylacetates undergo decarboxylative fluorination upon treatment with an electrophilic fluorination reagent to afford fluoromethylpyridines under catalyst-free conditions. Furthermore, we demonstrated the one-pot synthesis of fluoromethylpyridines from methyl pyridylacetates by saponification
- , entry 11). In the absence of MS 4 Å, the yield of 2a was diminished even when the reaction was carried out in THF (Table 1, entry 12). With the optimized reaction conditions in hand, we examined the one-pot synthesis of 2a from methyl ester 7a. Methyl 2-pyridylacetate 7a were saponified with lithium

Graphical Abstract

Scheme 1: Electrophilic decarboxylative functionalization of 2-pyridylacetates.

Scheme 2: One-pot procedure for the synthesis of 2a.

Scheme 3: Substrate scope. aSaponification was carried out with 2.5 equiv of LiOH, and 2.5 equiv of 6 was use...

Scheme 4: Reaction of α-monosubstituted 2-pyridylacetates.

Scheme 5: Proposed reaction pathway.

Scheme 6: Reaction of 3- and 4-pyridylacetates.
Anion exchange resins in phosphate form as versatile carriers for the reactions catalyzed by nucleoside phosphorylases
- Julia N. Artsemyeva,
- Ekaterina A. Remeeva,
- Tatiana N. Buravskaya,
- Irina D. Konstantinova,
- Roman S. Esipov,
- Anatoly I. Miroshnikov,
- Natalia M. Litvinko and
- Igor A. Mikhailopulo
Beilstein J. Org. Chem. 2020, 16, 2607–2622, doi:10.3762/bjoc.16.212
- cited therein). The classical two-stage version of the enzymatic transglycosylation reaction [16][17][18][19][20], as well as one-pot synthesis, and the more sophisticated option employing two cross-glycosylation transformations for nucleoside synthesis [23][24][25][26][27], seemed less attractive and

Graphical Abstract

Scheme 1: General scheme of the suggested synthesis of nucleosides employing the enzymatic phosphorolysis of ...

Figure 1: Phosphorolysis (5.0 mM K-phosphate buffer, pH 7.0; 23 °C) of Ara-U and thymidine (Thd) catalyzed by...

Scheme 2: Transarabinosylation of O6-methylguanine (OMG) employing Ara-U as a donor of the Ara-1Pi (1:1.5 mol...

Figure 2: Optimized conditions of phosphorolysis of Ara-U: 0.20 mmol of Ara-U in distilled water (30 mL) cont...

Scheme 3: Synthesis of nelarabine with intermediate preparation of crude Ara-1Pi.

Scheme 4: Synthesis of kinetin riboside with intermediate preparation of crude Rib-1Pi.
Palladium nanoparticles supported on chitin-based nanomaterials as heterogeneous catalysts for the Heck coupling reaction
- Tony Jin,
- Malickah Hicks,
- Davis Kurdyla,
- Sabahudin Hrapovic,
- Edmond Lam and
- Audrey Moores
Beilstein J. Org. Chem. 2020, 16, 2477–2483, doi:10.3762/bjoc.16.201

- NPs. A one-pot synthesis method was used to both deposit Pd salts and reduce them into NPs onto the support material. First, PdCl2 was mixed for 15 min with either ChNC or ChsNC in an acidic aqueous medium to form a dark yellow mixture. This step facilitated coordination of Pd salts onto the support

Graphical Abstract

Scheme 1: Pathway for the formation of ChNC and subsequently ChsNCs from bulk chitin.

Figure 1: TEM micrographs of (a) ChNCs and (b) ChsNCs. Both samples were stained and prepared on glow-dischar...

Scheme 2: Catalyst fabrication method for the deposition of Pd NPs onto chitin (PdNP@ChNC) and chitosan (PdNP...

Figure 2: TEM micrographs of (a) PdNP@ChNCs and (b) PdNP@ChsNCs. The samples were placed on glow discharged T...

Figure 3: High-resolution X-ray photoelectron spectroscopy of the Pd 3d region of (a) PdNP@ChNC and (b) PdNP@...
Reactions of 3-aryl-1-(trifluoromethyl)prop-2-yn-1-iminium salts with 1,3-dienes and styrenes
- Thomas Schneider,
- Michael Keim,
- Bianca Seitz and
- Gerhard Maas
Beilstein J. Org. Chem. 2020, 16, 2064–2072, doi:10.3762/bjoc.16.173
- theory), but also represent powerful 1,3-biselectrophiles. Thus, Diels–Alder reactions followed by an intramolecular SE(Ar) reaction of the α-(trifluoromethyl)iminium functional group were achieved as a two-step one-pot synthesis. On the other hand, an electrophilic (Markownikow type) addition of the

Graphical Abstract

Scheme 1: Diels–Alder reaction of propyn-1-iminium salt 1a compared with the reported [29] reaction of 4-phenyl-1...

Scheme 2: Sequential Diels–Alder/intramolecular SE(Ar) reaction of propyn-1-iminium triflates 1a,b. Condition...

Scheme 3: Diels–Alder reaction of 1a and anthracene followed by an intramolecular SE(Ar) reaction.

Figure 1: Solid-state molecular structure of 11 (ORTEP plot).

Scheme 4: Reactions of propyn-1-iminium salt 1a with styrenes.

Figure 2: Solid-state molecular structure of 12c (ORTEP plot).

Figure 3: Solid-state molecular structure of 12d (ORTEP plot). Both the R and the S enantiomer are present in...

Scheme 5: A mechanistic proposal for the reaction of alkyne 1a with styrenes.

Scheme 6: Reaction of alkyne 1a with 1,2-dihydronaphthalene.

Scheme 7: Synthesis and solid-state molecular structure (ORTEP plot) of pentafulvene 19; selected bond distan...

Scheme 8: Proposed mechanistic pathway leading to fulvene 19.
Synthesis of 3(2)-phosphonylated thiazolo[3,2-a]oxopyrimidines
- Ksenia I. Kaskevich,
- Anastasia A. Babushkina,
- Vladislav V. Gurzhiy,
- Dmitrij M. Egorov,
- Nataly I. Svintsitskaya and
- Albina V. Dogadina
Beilstein J. Org. Chem. 2020, 16, 1947–1954, doi:10.3762/bjoc.16.161
- uracil ring, leading to the formation of 7-oxopyrimidines. Structure of ritanserin and setoperone drugs. One-pot synthesis of 5(7)-oxothiazolopyrimidine-6-carbonitriles. Synthesis of thiazolopyrimidine-5-ones through the reaction of 2-aminothiazoles with ethyl acetoacetate. Synthesis of 2-(benzo[d

Graphical Abstract

Figure 1: Structure of ritanserin and setoperone drugs.

Scheme 1: One-pot synthesis of 5(7)-oxothiazolopyrimidine-6-carbonitriles.

Scheme 2: Synthesis of thiazolopyrimidine-5-ones through the reaction of 2-aminothiazoles with ethyl acetoace...

Scheme 3: Synthesis of 2-(benzo[d]thiazol-2-yl)-2-(7-R-5-oxo-5H-thiazolo[3,2-a]pyrimidin-3-yl)acetonitriles.

Scheme 4: Synthesis of 3-acyl-7-methyl-5H-thiazolo[3,2-a]pyrimidin-5-ones.

Scheme 5: Sonogashira coupling reaction of 6-amino-2-thiouracil with propargyl bromide.

Scheme 6: Reactions of 6-substituted 2-thiouracils 1a,b with chloroethynylphosphonates 2a–c.

Scheme 7: Reaction of 5-methyl-2-thiouracil (1c) with chloroethynylphosphonates 2a–c.

Scheme 8: Reaction of 2-thiouracil (1d) with chloroethynylphosphonates 2a–c.

Scheme 9: Reaction of 6-trifluoromethyl-2-thiouracil (1e) with chloroethynylphosphonates 2a–c.

Scheme 10: A plausible mechanism of the reaction between 6-trifluoromethyl-2-thiouracil (1e) and chloroethynyl...
Regiodivergent synthesis of functionalized pyrimidines and imidazoles through phenacyl azides in deep eutectic solvents
- Paola Vitale,
- Luciana Cicco,
- Ilaria Cellamare,
- Filippo M. Perna,
- Antonio Salomone and
- Vito Capriati
Beilstein J. Org. Chem. 2020, 16, 1915–1923, doi:10.3762/bjoc.16.158

- intramolecular nucleophilic attack to the terminal imino group of 9a, provides cyclized adduct 10a, and finally pyrimidine derivative 7a by aromatization/elimination of NH3. To the best of our knowledge, this is the first one-pot synthesis of functionalized pyrimidines using phenacyl azides as the sole starting
- in Supporting Information File 1. One-pot synthesis of 2,5-diarylpyrazines (A) (path a) or 2-aroyl-(4 or 5)-aryl-(1H)-imidazoles (B) (path b), or 2,4-diaroyl-6-arylpyrimidines (C) (path c) in DES from phenacyl azides (rt = room temperature). Transformation of phenacyl bromide (1a) in ChCl/Gly into

Graphical Abstract

Scheme 1: One-pot synthesis of 2,5-diarylpyrazines (A) (path a) or 2-aroyl-(4 or 5)-aryl-(1H)-imidazoles (B) ...

Scheme 2: Transformation of phenacyl bromide (1a) in ChCl/Gly into phenacyl azide (2a) and 2-benzoyl-(4 or 5)...

Scheme 3: Synthesis of 2-aroyl-(4 or 5)-aryl-(1H)-imidazoles 3. Scope of the reaction. Typical conditions: 1 ...

Scheme 4: Proposed mechanism for the formation of 2-aroyl-(4 or 5)-aryl-(1H)-imidazoles 3/3' from α-phenacyl ...

Scheme 5: Proposed mechanism for the formation of 2-benzoyl-(4 or 5)-phenyl-(1H)-imidazoles 3a/3a' and 2,4-di...

Scheme 6: Scope of the synthesis of 2,4-diaroyl-6-arylpyrimidines 7. Typical conditions: 2 (0.3 mmol), Et3N (...
One-pot and metal-free synthesis of 3-arylated-4-nitrophenols via polyfunctionalized cyclohexanones from β-nitrostyrenes
- Haruyasu Asahara,
- Minami Hiraishi and
- Nagatoshi Nishiwaki
Beilstein J. Org. Chem. 2020, 16, 1830–1836, doi:10.3762/bjoc.16.150
- (CH), 129.3 (CH), 134.8 (C), 138.1 (C), 139.8 (C), 142.3 (C), 158.9 (C). One-pot synthesis of 3-arylated-4-nitrophenol 5b To a solution of Danishefsky’s diene (2, 129.2 mg, 0.75 mmol) in toluene (1 mL), nitrostyrene 1b (90.0 mg, 0.50 mmol) was added, and the resulting mixture was heated at 120 °C for
- structure of nitroalkenes 1b and 1d. Synthetic scheme of the 3-arylated-4-nitrophenols 5. Conversion from 3a to 4a and one-pot synthesis of 4a. Deuteration of cyclohexanone 4a. A plausible mechanism for the formation of 5a. Optimization of the reaction conditions for the Diels–Alder reaction.a Aromatization
- of cyclohexanone 4a. One-pot synthesis of 3-arylated-4-nitrophenols 5. Supporting Information Supporting Information File 124: Spectral data for 5b–f, NMR spectra (1H, 13C, and DEPT) for 4a and 5a–f, and crystallographic data for 4a. Acknowledgements The authors appreciate the kind assistance

Graphical Abstract

Scheme 1: Synthetic scheme of the 3-arylated-4-nitrophenols 5.

Figure 1: X-ray crystallography of the major isomer of 4a. The thermal ellipsoids indicate 50% probability.

Scheme 2: Conversion from 3a to 4a and one-pot synthesis of 4a.

Scheme 3: Deuteration of cyclohexanone 4a.

Scheme 4: A plausible mechanism for the formation of 5a.

Figure 2: Resonance structure of nitroalkenes 1b and 1d.
One-pot synthesis of oxazolidinones and five-membered cyclic carbonates from epoxides and chlorosulfonyl isocyanate: theoretical evidence for an asynchronous concerted pathway
- Esra Demir,
- Ozlem Sari,
- Yasin Çetinkaya,
- Ufuk Atmaca,
- Safiye Sağ Erdem and
- Murat Çelik
Beilstein J. Org. Chem. 2020, 16, 1805–1819, doi:10.3762/bjoc.16.148
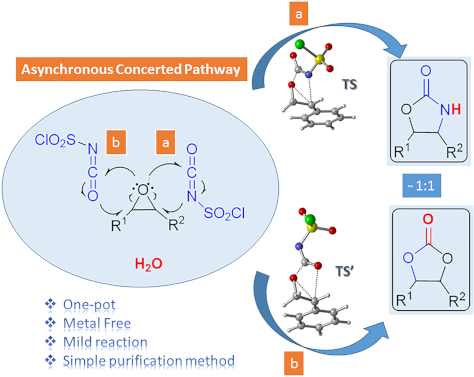
- , dichloromethane, toluene, and n-hexane/dichloromethane. While no reaction was observed in diethyl ether, the best conversion was achieved in dichloromethane. Benzene was not used as a solvent because of having toxic and carcinogenic effects. Herein, we report mild reaction conditions for the one-pot synthesis of

Graphical Abstract

Scheme 1: Oxazolidinone (1), five-membered cyclic carbonate (2) and some important compounds containing an ox...

Scheme 2: Proposed mechanisms by Keshava Murthy and Dhar [41] and De Meijere and co-workers [42].

Figure 1: Possible pathways for the formation of oxazolidinone intermediates 10 and 11. Optimized transition ...

Figure 2: Potential energy profile related to the formation of oxazolidinone intermediates 10 and 11 at the P...

Figure 3: IRC calculated for the formation of (a) 10 and (b) 11 at M06-2X/6-31+G(d,p) level. I-1, I-15, I-35, ...

Figure 4: Optimized geometries for the stationary points for the formation of 10 at PCM(DCM)/M06-2X/6-31+G(d,...

Scheme 3: Proposed mechanisms for the formation of oxazolidinone 9f.

Figure 5: Potential energy profiles for paths 1a (blue), 1b (red), 2 (green) and relative Gibbs free energies...

Figure 6: Optimized geometries for the stationary points of path 1b at PCM(DCM)/M06-2X/6-31+G(d,p)//M06-2X/6-...

Scheme 4: Proposed mechanism for the formation of five-membered cyclic carbonate 8f.

Figure 7: Potential energy profile and relative Gibbs free energies (kcal/mol) in DCM related to the formatio...

Figure 8: Optimized geometries for the stationary points of step 1 for the formation of 16 at PCM(DCM)/M06-2X...

Figure 9: Optimized geometries for the stationary points of step 2 for the formation of 17 at PCM(DCM)/M06-2X...

Figure 10: Optimized geometries for the stationary points of step 3 for the formation of PC8 at PCM(DCM)/M06-2...
Et3N/DMSO-supported one-pot synthesis of highly fluorescent β-carboline-linked benzothiophenones via sulfur insertion and estimation of the photophysical properties
- Dharmender Singh,
- Vipin Kumar and
- Virender Singh
Beilstein J. Org. Chem. 2020, 16, 1740–1753, doi:10.3762/bjoc.16.146
- -tethered benzothiophenone derivatives via incorporation of sulfur. To the best of our knowledge, this is the first report of one-pot synthesis of novel β-carboline-tethered benzothiophenones and evaluation of their light-emitting properties. In this regard, detailed studies are presented and discussed
- found to be slow reacting and produced 2aA in a lower yield (42%). From the perspective of green chemistry, one-pot reactions are preferred as less waste is generated due to the avoidance of work-up, isolation, and purification of intermediates [66]. Accordingly, the feasibility of a one-pot synthesis
- by the results obtained from the one-pot synthesis of β-carboline C-1 substituted benzothiophenone derivatives, we were interested if the scope of this one-pot strategy could be extended for the synthesis of β-carboline C-3-tethered benzothiophenones (Scheme 5). Thus, the Claisen–Schmidt condensation

Graphical Abstract

Figure 1: Representative examples of some commercial drugs and biologically active alkaloids.

Scheme 1: Synthesis of β-carboline-linked 2-nitrochalcones.

Scheme 2: Synthesis of β-carboline-linked benzothiophenone frameworks.

Scheme 3: Comparison of outcome of one-pot vs two-pot approach.

Scheme 4: One-pot synthesis of β-carboline C-1-tethered benzothiophenone derivatives.

Scheme 5: One-pot synthesis of β-carboline C-3-linked benzothiophenone derivatives.

Scheme 6: One-pot synthesis of β-carboline-linked benzothiophene derivative 6C.

Scheme 7: Control experiment in the presence of a radical scavenger.

Figure 2: Proposed reaction mechanism.

Figure 3: Fluorescence spectra of 2aA–nA, 2bB, 2hB, and 6C.

Figure 4: Fluorescence spectra of 4aA–gA, and 4eB.
One-pot synthesis of isosorbide from cellulose or lignocellulosic biomass: a challenge?
- Isaline Bonnin,
- Raphaël Mereau,
- Thierry Tassaing and
- Karine De Oliveira Vigier
Beilstein J. Org. Chem. 2020, 16, 1713–1721, doi:10.3762/bjoc.16.143


Graphical Abstract

Scheme 1: Conversion of cellulose to isosorbide.

Scheme 2: Combination of mineral acids or heteropolyacids and a supported metal catalyst to produce isosorbid...

Scheme 3: Conversion of sorbitol to isosorbide via the formation of sorbitans.

Scheme 4: Conversion of cellulose to isosorbide in the presence of heteropolyacids and metal-supported cataly...

Scheme 5: Summary of the results obtained in one-pot one step processes [21-25].

Scheme 6: Conversion of (ligno)cellulose to isosorbide in the presence of Amberlyt 70 and a Ru/C catalyst [26,27].

Scheme 7: Use of Ru-supported on mesoporous nobium phosphate (mNbPO) for the synthesis of isosorbide from cel...
Microwave-assisted efficient one-pot synthesis of N2-(tetrazol-5-yl)-6-aryl/heteroaryl-5,6-dihydro-1,3,5-triazine-2,4-diamines
- Moustafa Sherief Moustafa,
- Ramadan Ahmed Mekheimer,
- Saleh Mohammed Al-Mousawi,
- Mohamed Abd-Elmonem,
- Hesham El-Zorba,
- Afaf Mohamed Abdel Hameed,
- Tahany Mahmoud Mohamed and
- Kamal Usef Sadek
Beilstein J. Org. Chem. 2020, 16, 1706–1712, doi:10.3762/bjoc.16.142
- Chemistry, Faculty of Science, Minia University, Minia 61519, Egypt Department of Pharmacology, Faculty of Veterinary Medicine, Cairo University, Giza 12211, Egypt 10.3762/bjoc.16.142 Abstract An efficient one-pot synthesis of N2-(tetrazol-5-yl)-6-aryl/heteroaryl-1,3,5-triazine-2,4-diamine derivatives was
- -2,4-diamines; one-pot synthesis; X-ray crystallography; Introduction The family of triazines is of considerable interest in fields related to organic and medicinal chemistry. 2,4-Diaminotriazines are privileged scaffolds exhibiting diverse biological activities such as antibacterial [1], anti-HSV-1
- required temperature control and showed dependence on the amine nucleophile reactivity [27]. Another route involved the reaction of substituted biguanidines with acetic anhydrides, chlorides or carboxylates [11][28][29][30][31]. Liu et al. [32] reported a one-pot synthesis of N2,6-disubstituted-1,3,5

Graphical Abstract

Scheme 1: Previously reported methods for the synthesis of 1,3,5-triazine-2,4-diamine derivatives.

Scheme 2: One-pot synthesis of N2-(tetrazol-5-yl)-6-aryl/heteroaryl-5,6-dihydro-1,3,5-triazine-2,4-diamines 4a...

Figure 1: ORTEP diagram of compound 4i.

Scheme 3: Plausible different routes to account for the formation of products 4.
Pauson–Khand reaction of fluorinated compounds
- Jorge Escorihuela,
- Daniel M. Sedgwick,
- Alberto Llobat,
- Mercedes Medio-Simón,
- Pablo Barrio and
- Santos Fustero
Beilstein J. Org. Chem. 2020, 16, 1662–1682, doi:10.3762/bjoc.16.138

- 21, was unsuccessful and no tricyclic product was formed. Bonnet-Delpon and co-workers reported the one-pot synthesis of several CF3-containing N-tethered amines in good yields (54–86% over 2 steps) [49]. These products were subjected to metathesis reactions in the presence of Grubbs catalyst

Graphical Abstract

Scheme 1: Schematic representation of the Pauson–Khand reaction.

Scheme 2: Substrates included in this review.

Scheme 3: Commonly accepted mechanism for the Pauson–Khand reaction.

Scheme 4: Regioselectivity of the PKR.

Scheme 5: Variability at the acetylenic and olefinic counterpart.

Scheme 6: Pauson–Khand reaction of fluoroolefinic enynes reported by the group of Ishizaki [46].

Scheme 7: PKR of enynes bearing fluorinated groups on the alkynyl moiety, reported by the group of Ishizaki [46]....

Scheme 8: Intramolecular PKR of 1,7-enynes reported by the group of Billard [47].

Scheme 9: Intramolecular PKR of 1,7-enynes reported by the group of Billard [48].

Scheme 10: Intramolecular PKR of 1,7-enynes by the group of Bonnet-Delpon [49]. Reaction conditions: i) Co(CO)8 (1...

Scheme 11: Intramolecular PKR of 1,6-enynes reported by the group of Ichikawa [50].

Scheme 12: Intramolecular Rh(I)-catalyzed PKR reported by the group of Hammond [52].

Scheme 13: Intramolecular PKR of allenynes reported by the group of Osipov [53].

Scheme 14: Intramolecular PKR of 1,7-enynes reported by the group of Osipov [53].

Scheme 15: Intramolecular PKR of fluorine-containing 1,6-enynes reported by the Konno group [54].

Scheme 16: Diastereoselective PKR with enantioenriched fluorinated enynes 34 [55].

Scheme 17: Intramolecular PKR reported by the group of Martinez-Solorio [56].

Scheme 18: Fluorine substitution at the olefinic counterpart.

Scheme 19: Synthesis of fluorinated enynes 37 [59].

Scheme 20: Fluorine-containing substrates in PKR [59].

Scheme 21: Pauson Khand reaction for fluorinated enynes by the Fustero group: scope and limitations [59].

Scheme 22: Synthesis of chloro and bromo analogues [59].

Scheme 23: Dimerization pathway [59].

Scheme 24: Synthesis of fluorine-containing N-tethered 1,7-enynes [61].

Scheme 25: Intramolecular PKR of chiral N-tethered fluorinated 1,7-enynes [61].

Scheme 26: Examples of further modifications to the Pauson−Khand adducts [61].

Scheme 27: Asymmetric synthesis the fluorinated enynes 53.

Scheme 28: Intramolecular PKR of chiral N-tethered 1,7-enynes 53 [64].

Scheme 29: Intramolecular PKR of chiral N-tethered 1,7-enyne bearing a vinyl fluoride [64].

Scheme 30: Catalytic intramolecular PKR of chiral N-tethered 1,7-enynes [64].

Scheme 31: Model fluorinated alkynes used by Riera and Fustero [70].

Scheme 32: PKR with norbornadiene and fluorinated alkynes 58 [71].

Scheme 33: Nucleophilic addition/detrifluoromethylation and retro Diels-Alder reactions [70].

Scheme 34: Tentative mechanism for the nucleophilic addition/retro-aldol reaction sequence.

Scheme 35: Catalytic PKR with norbornadiene [70].

Scheme 36: Scope of the PKR of trifluoromethylalkynes with norbornadiene [72].

Scheme 37: DBU-mediated detrifluoromethylation [72].

Scheme 38: A simple route to enone 67, a common intermediate in the total synthesis of α-cuparenone.

Scheme 39: Effect of the olefin partner in the regioselectivity of the PKR with trifluoromethyl alkynes [79].

Scheme 40: Intermolecular PKR of trifluoromethylalkynes with 2-norbornene reported by the group of Konno [54].

Scheme 41: Intermolecular PKR of diarylalkynes with 2-norbornene reported by the group of Helaja [80].

Scheme 42: Intermolecular PKR reported by León and Fernández [81].

Scheme 43: PKR reported with cyclopropene 73 [82].
Five-component, one-pot synthesis of an electroactive rotaxane comprising a bisferrocene macrocycle
- Natalie Lagesse,
- Luca Pisciottani,
- Maxime Douarre,
- Pascale Godard,
- Brice Kauffmann,
- Vicente Martí-Centelles and
- Nathan D. McClenaghan
Beilstein J. Org. Chem. 2020, 16, 1564–1571, doi:10.3762/bjoc.16.128

Graphical Abstract

Figure 1: (a) Non-functionalized rotaxanes previously described in the literature. (b) The redox-active rotax...

Figure 2: Synthesis of the redox-active rotaxanes 1 and macrocycle 2.

Figure 3: Most stable conformers obtained by Monte Carlo conformational search using model compounds. (a) Mod...

Figure 4: 1H NMR spectrum (300 MHz) of rotaxane 1a (top) and thread 4a (bottom) in CDCl3 (a designation of th...

Figure 5: 1H,1H-ROESY NMR spectrum (600 MHz) of the rotaxane 1a in CDCl3.

Figure 6: Cyclic voltammogram of ferrocene rotaxane 1a (0.67 mM) in CH2Cl2/CH3CN 1:5 (TBAPF6 0.10 M, scan rat...

Figure 7: Single crystal X-ray structures of (a) rotaxane 1a and (b) Leigh’s rotaxane I [4].
Heterogeneous photocatalysis in flow chemical reactors
- Christopher G. Thomson,
- Ai-Lan Lee and
- Filipe Vilela
Beilstein J. Org. Chem. 2020, 16, 1495–1549, doi:10.3762/bjoc.16.125

- the homogeneous photocatalyst. The flow synthesis also prevented the formation of a chlorinated byproduct identified on the batch resins as an intermediate, and purification could be applied to wash out reactants from the first step of the one-pot synthesis, a potentially useful synthetic advantage of

Graphical Abstract

Figure 1: A) Bar chart of the publications per year for the topics “Photocatalysis” (49,662 instances) and “P...

Figure 2: A) Professor Giacomo Ciamician and Dr. Paolo Silber on their roof laboratory at the University of B...

Scheme 1: PRC trifluoromethylation of N-methylpyrrole (1) using hazardous gaseous CF3I safely in a flow react...

Figure 3: A) Unit cells of the three most common crystal structures of TiO2: rutile, brookite, and anatase. R...

Figure 4: Illustration of the key semiconductor photocatalysis events: 1) A photon with a frequency exceeding...

Figure 5: Photocatalytic splitting of water by oxygen vacancies on a TiO2(110) surface. Reprinted with permis...

Figure 6: Proposed adsorption modes of A) benzene, B) chlorobenzene, C) toluene, D) phenol, E) anisole, and F...

Figure 7: Structures of the sulfonate-containing organic dyes RB5 (3) and MX-5B (4) and the adsorption isothe...

Figure 8: Idealised triclinic unit cell of a g-C3N4 type polymer, displaying possible hopping transport scena...

Figure 9: Idealised structure of a perfect g-C3N4 sheet. The central unit highlighted in red represents one t...

Figure 10: Timeline of the key processes of charge transport following the photoexcitation of g-C3N4, leading ...

Scheme 2: Photocatalytic bifunctionalisation of heteroarenes using mpg-C3N4, with the selected examples 5 and ...

Figure 11: A) Structure of four linear conjugated polymer photocatalysts for hydrogen evolution, displaying th...

Figure 12: Graphical representation of the common methods used to immobilise molecular photocatalysts (PC) ont...

Figure 13: Wireless light emitter-supported TiO2 (TiO2@WLE) HPCat spheres powered by resonant inductive coupli...

Figure 14: Graphical representation of zinc–perylene diimide (Zn-PDI) supramolecular assembly photocatalysis v...

Scheme 3: Upconversion of NIR photons to the UV frequency by NaYF4:Yb,Tm nanocrystals sequentially coated wit...

Figure 15: Types of reactors employed in heterogeneous photocatalysis in flow. A) Fixed bed reactors and the s...

Figure 16: Electrochemical potential of common semiconductor, transition metal, and organic dye-based photocat...

Scheme 4: Possible mechanisms of an immobilised molecular photoredox catalyst by oxidative or reductive quenc...

Scheme 5: Scheme of the CMB-C3N4 photocatalytic decarboxylative fluorination of aryloxyacetic acids, with the...

Scheme 6: Scheme of the g-C3N4 photocatalytic desilylative coupling reaction in flow and proposed mechanism [208].

Scheme 7: Proposed mechanism of the radical cyclisation of unsaturated alkyl 2-bromo-1,3-dicarbonyl compounds...

Scheme 8: N-alkylation of benzylamine and schematic of the TiO2-coated microfluidic device [213].

Scheme 9: Proposed mechanism of the Pt@TiO2 photocatalytic deaminitive cyclisation of ʟ-lysine (23) to ʟ-pipe...

Scheme 10: A) Proposed mechanism for the photocatalytic oxidation of phenylboronic acid (24). B) Photos and SE...

Scheme 11: Proposed mechanism for the DA-CMP3 photocatalytic aza-Henry reaction performed in a continuous flow...

Scheme 12: Proposed mechanism for the formation of the cyclic product 32 by TiO2-NC HPCats in a slurry flow re...

Scheme 13: Reaction scheme for the photocatalytic synthesis of homo and hetero disulfides in flow and scope of...

Scheme 14: Reaction scheme for the MoOx/TiO2 HPCat oxidation of cyclohexane (34) to benzene. The graph shows t...

Scheme 15: Proposed mechanism of the TiO2 HPC heteroarene C–H functionalisation via aryl radicals generated fr...

Scheme 16: Scheme of the oxidative coupling of benzylamines with the HOTT-HATN HPCat and selected examples of ...

Scheme 17: Photocatalysis oxidation of benzyl alcohol (40) to benzaldehyde (41) in a microflow reactor coated ...

Figure 17: Mechanisms of Dexter and Forster energy transfer.

Scheme 18: Continuous flow process for the isomerisation of alkenes with an ionic liquid-immobilised photocata...

Scheme 19: Singlet oxygen synthetic step in the total synthesis of canataxpropellane [265].

Scheme 20: Scheme and proposed mechanism of the singlet oxygen photosensitisation by CMP_X HPCats, with the st...

Scheme 21: Structures of CMP HPCat materials applied by Vilela and co-workers for the singlet oxygen photosens...

Scheme 22: Polyvinylchloride resin-supported TDCPP photosensitisers applied for singlet oxygen photosensitisat...

Scheme 23: Structure of the ionically immobilised TPP photosensitiser on amberlyst-15 ion exchange resins (TPP...

Scheme 24: Photosensitised singlet oxygen oxidation of citronellol (46) in scCO2, with automatic phase separat...

Scheme 25: Schematic of PS-Est-BDP-Cl2 being applied for singlet oxygen photosensitisation in flow. A) Pseudo-...

Scheme 26: Reaction scheme of the singlet oxygen oxidation of furoic acid (54) using a 3D-printed microfluidic...

Figure 18: A) Photocatalytic bactericidal mechanism by ROS oxidative cleavage of membrane lipids (R = H, amino...

Figure 19: A) Suggested mechanisms for the aqueous pollutant degradation by TiO2 in a slurry flow reactor [284-287]. B)...

Figure 20: Schematic of the flow system used for the degradation of aqueous oxytetracycline (56) solutions [215]. M...

Scheme 27: Degradation of a salicylic acid (57) solution by a coupled solar photoelectro-Fenton (SPEF) process...

Figure 21: A) Schematic flow diagram using the TiO2-coated NETmix microfluidic device for an efficient mass tr...






