Search results
Search for "organocatalysts" in Full Text gives 156 result(s) in Beilstein Journal of Organic Chemistry.
Asymmetric organocatalyzed synthesis of coumarin derivatives
- Natália M. Moreira,
- Lorena S. R. Martelli and
- Arlene G. Corrêa
Beilstein J. Org. Chem. 2021, 17, 1952–1980, doi:10.3762/bjoc.17.128
- stereogenic centers. Review A plethora of highly effective small‐molecule organocatalysts have enriched the field of organic synthesis [27], including chiral proline derivatives, N‐heterocyclic carbenes, chiral thioureas and Brønsted acids as well as phase‐transfer catalysts (PTC), such as the quaternary
- . Catalysis via covalent bonding Organocatalysts made from chiral secondary amines have been widely used in the last years. According to Jørgensen, in general, the carbonyl functionalization employing amine catalysts can be separated in four different types [29]. When aldehydes are employed, both
- product 17 was obtained in 15 steps with 6% overall yield. Although chiral secondary amines have proved to be particularly useful catalysts, primary amines as organocatalysts in asymmetric synthesis have also played a significant role [36]. For instance, Kim et al. described the enantioselective Michael

Graphical Abstract

Figure 1: Coumarin-derived commercially available drugs.

Figure 2: Inhibition of acetylcholinesterase by coumarin derivatives.

Scheme 1: Michael addition of 4-hydroxycoumarins 1 to α,β‐unsaturated enones 2.

Scheme 2: Organocatalytic conjugate addition of 4-hydroxycoumarin 1 to α,β-unsaturated aldehydes 2 followed b...

Scheme 3: Synthesis of 3,4-dihydrocoumarin derivatives 10 through decarboxylative and dearomatizative cascade...

Scheme 4: Total synthesis of (+)-smyrindiol (17).

Scheme 5: Michael addition of 4-hydroxycoumarin (1) to enones 2 through a bifunctional modified binaphthyl or...

Scheme 6: Michael addition of ketones 20 to 3-aroylcoumarins 19 using a cinchona alkaloid-derived primary ami...

Scheme 7: Enantioselective reaction of cyclopent-2-enone-derived MBH alcohols 24 with 4-hydroxycoumarins 1.

Scheme 8: Sequential Michael addition/hydroalkoxylation one-pot approach to annulated coumarins 28 and 30.

Scheme 9: Michael addition of 4-hydroxycoumarins 1 to enones 2 using a binaphthyl diamine catalyst 31.

Scheme 10: Asymmetric Michael addition of 4-hydroxycoumarin 1 with α,β-unsaturated ketones 2 catalyzed by a ch...

Scheme 11: Catalytic asymmetric β-C–H functionalization of ketones via enamine oxidation.

Scheme 12: Enantioselective synthesis of polycyclic coumarin derivatives 37 catalyzed by an primary amine-imin...

Scheme 13: Allylic alkylation reaction between 3-cyano-4-methylcoumarins 39 and MBH carbonates 40.

Scheme 14: Enantioselective synthesis of cyclopropa[c]coumarins 45.

Scheme 15: NHC-catalyzed lactonization of 2-bromoenals 46 with 4-hydroxycoumarin (1).

Scheme 16: NHC-catalyzed enantioselective synthesis of dihydrocoumarins 51.

Scheme 17: Domino reaction of enals 2 with hydroxylated malonate 53 catalyzed by NHC 55.

Scheme 18: Oxidative [4 + 2] cycloaddition of enals 57 to coumarins 56 catalyzed by NHC 59.

Scheme 19: Asymmetric [3 + 2] cycloaddition of coumarins 43 to azomethine ylides 60 organocatalyzed by quinidi...

Scheme 20: Synthesis of α-benzylaminocoumarins 64 through Mannich reaction between 4-hydroxycoumarins (1) and ...

Scheme 21: Asymmetric addition of malonic acid half-thioesters 67 to coumarins 66 using the sulphonamide organ...

Scheme 22: Enantioselective 1,4-addition of azadienes 71 to 3-homoacyl coumarins 70.

Scheme 23: Michael addition/intramolecular cyclization of 3-acylcoumarins 43 to 3-halooxindoles 74.

Scheme 24: Enantioselective synthesis of 3,4-dihydrocoumarins 78 catalyzed by squaramide 73.

Scheme 25: Organocatalyzed [4 + 2] cycloaddition between 2,4-dienals 79 and 3-coumarincarboxylates 43.

Scheme 26: Enantioselective one-pot Michael addition/intramolecular cyclization for the synthesis of spiro[dih...

Scheme 27: Michael/hemiketalization addition enantioselective of hydroxycoumarins (1) to: (a) enones 2 and (b)...

Scheme 28: Synthesis of 2,3-dihydrofurocoumarins 89 through Michael addition of 4-hydroxycoumarins 1 to β-nitr...

Scheme 29: Synthesis of pyrano[3,2-c]chromene derivatives 93 via domino reaction between 4-hydroxycoumarins (1...

Scheme 30: Conjugated addition of 4-hydroxycoumarins 1 to nitroolefins 95.

Scheme 31: Michael addition of 4-hydroxycoumarin 1 to α,β-unsaturated ketones 2 promoted by primary amine thio...

Scheme 32: Enantioselective synthesis of functionalized pyranocoumarins 99.

Scheme 33: 3-Homoacylcoumarin 70 as 1,3-dipole for enantioselective concerted [3 + 2] cycloaddition.

Scheme 34: Synthesis of warfarin derivatives 107 through addition of 4-hydroxycoumarins 1 to β,γ-unsaturated α...

Scheme 35: Asymmetric multicatalytic reaction sequence of 2-hydroxycinnamaldehydes 109 with 4-hydroxycoumarins ...

Scheme 36: Mannich asymmetric addition of cyanocoumarins 39 to isatin imines 112 catalyzed by the amide-phosph...

Scheme 37: Enantioselective total synthesis of (+)-scuteflorin A (119).
Development of N-F fluorinating agents and their fluorinations: Historical perspective
- Teruo Umemoto,
- Yuhao Yang and
- Gerald B. Hammond
Beilstein J. Org. Chem. 2021, 17, 1752–1813, doi:10.3762/bjoc.17.123


Graphical Abstract

Scheme 1: Fluorination with N-F amine 1-1.

Scheme 2: Preparation of N-F amine 1-1.

Scheme 3: Reactions of N-F amine 1-1.

Scheme 4: Synthesis of N-F perfluoroimides 2-1 and 2-2.

Scheme 5: Synthesis of 1-fluoro-2-pyridone (3-1).

Scheme 6: Fluorination with 1-fluoro-2-pyridone (3-1).

Figure 1: Synthesis of N-F sulfonamides 4-1a–g.

Scheme 7: Fluorination with N-F reagent 4-1b,c,f.

Scheme 8: Fluorination of alkenyllithiums with N-F 4-1h.

Scheme 9: Synthesis of N-fluoropyridinium triflate (5-4a).

Scheme 10: Synthetic methods for N-F-pyridinium salts.

Figure 2: Synthesis of various N-fluoropyridinium salts. Note: athis yield was the one by the improved method...

Scheme 11: Fluorination power order of N-fluoropyridinium salts.

Scheme 12: Fluorinations with N-F salts 5-4.

Scheme 13: Fluorination of Corey lactone 5-7 with N-F-bis(methoxymethyl) salt 5-4l.

Scheme 14: Fluorination with NFPy.

Scheme 15: Synthesis of the N-F reagent, N-fluoroquinuclidinium fluoride (6-1).

Scheme 16: Fluorinations achieved with N-F fluoride 6-1.

Scheme 17: Synthesis of N-F imides 7-1a–g.

Scheme 18: Fluorination with (CF3SO2)2NF, 7-1a.

Scheme 19: Fluorination reactions of various substrates with 7-1a.

Scheme 20: Synthesis of N-F triflate 8-1.

Scheme 21: Synthesis of chiral N-fluoro sultams 9-1 and 9-2.

Scheme 22: Fluorination with chiral N-fluoro sultams 9-1 and 9-2.

Scheme 23: Synthesis of saccharin-derived N-fluorosultam 10-2.

Scheme 24: Fluorination with N-fluorosultam 10-2.

Scheme 25: Synthesis of N-F reagent 11-2.

Scheme 26: Fluorination with N-F reagent 11-2.

Scheme 27: Synthesis and reaction of N-fluorolactams 12-1.

Scheme 28: Synthesis of NFOBS 13-2.

Scheme 29: Fluorination with NFOBS 13-2.

Scheme 30: Synthesis of NFSI (14-2).

Scheme 31: Fluorination with NFSI 14-2.

Scheme 32: Synthesis of N-fluorosaccharin (15-1) and N-fluorophthalimide (15-2).

Scheme 33: Synthesis of N-F salts 16-3.

Scheme 34: Fluorination with N-F salts 16-3.

Figure 3: Monofluorination with Selectfluor (16-3a).

Figure 4: Difluorination with Selectfluor (16-3a).

Scheme 35: Transfer fluorination of Selectfluor (16-3a).

Scheme 36: Fluorination of substrates with Selectfluor (16-3a).

Scheme 37: Synthesis of chiral N-fluoro-sultam 17-2.

Scheme 38: Asymmetric fluorination with chiral 17-2.

Figure 5: Synthesis of Zwitterionic N-fluoropyridinium salts 18-2a–h.

Scheme 39: Fluorinating power order of zwitterionic N-fluoropyridinium salts.

Scheme 40: Fluorination with zwitterionic 18-2.

Scheme 41: Activation of salt 18-2h with TfOH.

Scheme 42: Synthesis of NFTh, 19-2.

Scheme 43: Fluorination with NFTh, 19-2.

Scheme 44: Synthesis of 3-fluorobenzo-1,2,3-oxathiazin-4-one 2,2-dioxide (20-2).

Scheme 45: Fluorination with 20-2.

Scheme 46: Synthesis of N-F amide 21-3.

Scheme 47: Fluorination with N-F amide 21-2.

Scheme 48: Synthesis of N,N’-difluorodiazoniabicyclo[2.2.2]octane salts 22-1.

Scheme 49: One-pot synthesis of N,N’-difluoro-1,4-diazoniabicyclo[2.2.2]octane bistetrafluoroborate salt (22-1d...

Figure 6: Fluorination of anisole with 22-1a, d, e.

Scheme 50: Fluorination with N,N’-diF bisBF4 22-1d.

Scheme 51: Synthesis of bis-N-F reagents 23-1–5.

Scheme 52: Fluorination with 23-2, 4, 5.

Figure 7: Synthesis of N,N’-difluorobipyridinium salts 24-2.

Figure 8: Controlled fluorination of N,N’-diF 24-2.

Scheme 53: Fluorinating power of N,N’-diF salts 24-2 and N-F salt 5-4a.

Scheme 54: Fluorination reactions with SynfluorTM (24-2b).

Scheme 55: Additional fluorination reactions with SynfluorTM (24-2b).

Scheme 56: Synthesis of N-F 25-1.

Scheme 57: Fluorination of polycyclic aromatics with 25-1.

Scheme 58: Synthesis of 26-1 and dimethyl analog 26-2.

Scheme 59: Fluorination with reagents 26-1, 26-2, 1-1, and 26-3.

Scheme 60: Synthesis of N-F reagent 27-2.

Scheme 61: Synthesis of chiral N-F reagents 27-6.

Scheme 62: Synthesis of chiral N-F 27-7–9.

Scheme 63: Asymmetric fluorination with 27-6.

Scheme 64: Synthesis of chiral N-F reagents 28-3.

Scheme 65: Asymmetric fluorination with 28-3.

Scheme 66: Synthesis of chiral N-F reagents 28-7.

Figure 9: Asymmetric fluorination with 28-7.

Scheme 67: In situ formation of N-fluorinated cinchona alkaloids with SelectfluorTM.

Scheme 68: Asymmetric fluorination with N-F alkaloids formed in situ.

Scheme 69: Synthesis of N-fluorocinchona alkaloids with Selectfluor.

Scheme 70: Asymmetric fluorination with 30-1–4.

Scheme 71: Transfer fluorination from various N-F reagents.

Figure 10: Asymmetric fluorination of silyl enol ethers.

Scheme 72: Synthesis of N-fluoro salt 32-2.

Scheme 73: Reactivity of N-fluorotriazinium salt 32-2.

Scheme 74: Synthesis of bulky N-fluorobenzenesulfonimide NFBSI 33-3.

Scheme 75: Comparison of NFSI and NFBSI.

Scheme 76: Synthesis of p-substituted N-fluorobenzenesulfonimides 34-3.

Figure 11: Asymmetric fluorination with 34-3 and a chiral catalyst 34-4.

Scheme 77: 1,4-Fluoroamination with Selecfluor and a chiral catalyst.

Figure 12: Asymmetric fluoroamination with 35-5a, b.

Scheme 78: Synthesis of Selectfluor analogs 35-5a, b.

Scheme 79: Synthesis of chiral dicationic DABCO-based N-F reagents 36-5.

Scheme 80: Asymmetric fluorocyclization with chiral 36-5b.

Scheme 81: Synthesis of chiral 37-2a,b.

Scheme 82: Asymmetric fluorination with chiral 37-2a,b.

Scheme 83: Asymmetric fluorination with chiral 37-2b.

Scheme 84: Reaction of indene with chiral 37-2a,b.

Scheme 85: Synthesis of Me-NFSI, 38-2.

Scheme 86: Fluorination of active methine compounds with Me-NFSI.

Scheme 87: Fluorination of malonates with Me-NFSI.

Scheme 88: Fluorination of keto esters with Me-NFSI.

Scheme 89: Synthesis of N-F 39-3 derived from the ethylene-bridged Tröger’s base.

Scheme 90: Fluorine transfer from N-F 39-3.

Scheme 91: Fluorination with N-F 39-3.

Scheme 92: Synthesis of SelectfluorCN.

Scheme 93: Bistrifluoromethoxylation of alkenes using SelectfluorCN.

Figure 13: Synthesis of NFAS 41-2.

Scheme 94: Radical fluorination with different N-F reagents.

Scheme 95: Radical fluorination of alkenes with NFAS 41-2.

Scheme 96: Radical fluorination of alkenes with NFAS 41-2f.

Scheme 97: Decarboxylative fluorination with NFAS 41-2a,f.

Scheme 98: Fluorine plus detachment (FPD).

Figure 14: FPD values of representative N-F reagents in CH2Cl2 and CH3CN (in parentheses). Adapted with permis...

Scheme 99: N-F homolytic bond dissociation energy (BDE).

Figure 15: BDE values of representative N-F reagents in CH3CN. Adapted with permission from ref. [127]. Copyright 2...

Figure 16: Quantitative reactivity scale for popular N-F reagents. Adapted with permission from ref. [138], publish...

Scheme 100: SET and SN2 mechanisms.

Scheme 101: Radical clock reactions.

Scheme 102: Reaction of potassium enolate of citronellic ester with N-F reagents, 10-1, NFSI, and 8-1.

Scheme 103: Reaction of compound IV with Selectfluor (OTf) and NFSI.

Scheme 104: Reaction of TEMPO with Selecfluor.
A comprehensive review of flow chemistry techniques tailored to the flavours and fragrances industries
- Guido Gambacorta,
- James S. Sharley and
- Ian R. Baxendale
Beilstein J. Org. Chem. 2021, 17, 1181–1312, doi:10.3762/bjoc.17.90

Graphical Abstract

Figure 1: Representative shares of the global F&F market (2018) segmented on their applications [1].

Figure 2: General structure of an international fragrance company [2].

Figure 3: The Michael Edwards fragrance wheel.

Figure 4: Examples of oriental (1–3), woody (4–7), fresh (8–10), and floral (11 and 12) notes.

Figure 5: A basic depiction of batch vs flow.

Scheme 1: Examples of reactions for which flow processing outperforms batch.

Scheme 2: Some industrially important aldol-based transformations.

Scheme 3: Biphasic continuous aldol reactions of acetone and various aldehydes.

Scheme 4: Aldol synthesis of 43 in flow using LiHMDS as the base.

Scheme 5: A semi-continuous synthesis of doravirine (49) involving a key aldol reaction.

Scheme 6: Enantioselective aldol reaction using 5-(pyrrolidin-2-yl)tetrazole (51) as catalyst in a microreact...

Scheme 7: Gröger's example of asymmetric aldol reaction in aqueous media.

Figure 6: Immobilised reagent column reactor types.

Scheme 8: Photoinduced thiol–ene coupling preparation of silica-supported 5-(pyrrolidin-2-yl)tetrazole 63 and...

Scheme 9: Continuous-flow approach for enantioselective aldol reactions using the supported catalyst 67.

Scheme 10: Ötvös’ employment of a solid-supported peptide aldol catalyst in flow.

Scheme 11: The use of proline tetrazole packed in a column for aldol reaction between cyclohexanone (65) and 2...

Scheme 12: Schematic diagram of an aminosilane-grafted Si-Zr-Ti/PAI-HF reactor for continuous-flow aldol and n...

Scheme 13: Continuous-flow condensation for the synthesis of the intermediate 76 to nabumetone (77) and Microi...

Scheme 14: Synthesis of ψ-Ionone (80) in continuous-flow via aldol condensation between citral (79) and aceton...

Scheme 15: Synthesis of β-methyl-ionones (83) from citral (79) in flow. The steps are separately described, an...

Scheme 16: Continuous-flow synthesis of 85 from 84 described by Gavriilidis et al.

Scheme 17: Continuous-flow scCO2 apparatus for the synthesis of 2-methylpentanal (87) and the self-condensed u...

Scheme 18: Chen’s two-step flow synthesis of coumarin (90).

Scheme 19: Pechmann condensation for the synthesis of 7-hydroxyxcoumarin (93) in flow. The setup extended to c...

Scheme 20: Synthesis of the dihydrojasmonate 35 exploiting nitro derivative proposed by Ballini et al.

Scheme 21: Silica-supported amines as heterogeneous catalyst for nitroaldol condensation in flow.

Scheme 22: Flow apparatus for the nitroaldol condensation of p-hydroxybenzaldehyde (102) to nitrostyrene 103 a...

Scheme 23: Nitroaldol reaction of 64 to 105 employing a quaternary ammonium functionalised PANF.

Scheme 24: Enantioselective nitroaldol condensation for the synthesis of 108 under flow conditions.

Scheme 25: Enatioselective synthesis of 1,2-aminoalcohol 110 via a copper-catalysed nitroaldol condensation.

Scheme 26: Examples of Knoevenagel condensations applied for fragrance components.

Scheme 27: Flow apparatus for Knoevenagel condensation described in 1989 by Venturello et al.

Scheme 28: Knoevenagel reaction using a coated multichannel membrane microreactor.

Scheme 29: Continuous-flow apparatus for Knoevenagel condensation employing sugar cane bagasse as support deve...

Scheme 30: Knoevenagel reaction for the synthesis of 131–135 in flow using an amine-functionalised silica gel. ...

Scheme 31: Continuous-flow synthesis of compound 137, a key intermediate for the synthesis of pregabalin (138)...

Scheme 32: Continuous solvent-free apparatus applied for the synthesis of compounds 140–143 using a TSE. Throu...

Scheme 33: Lewis et al. developed a spinning disc reactor for Darzens condensation of 144 and a ketone to furn...

Scheme 34: Some key industrial applications of conjugate additions in the F&F industry.

Scheme 35: Continuous-flow synthesis of 4-(2-hydroxyethyl)thiomorpholine 1,1-dioxide (156) via double conjugat...

Scheme 36: Continuous-flow system for Michael addition using CsF on alumina as the catalyst.

Scheme 37: Calcium chloride-catalysed asymmetric Michael addition using an immobilised chiral ligand.

Scheme 38: Continuous multistep synthesis for the preparation of (R)-rolipram (173). Si-NH2: primary amine-fun...

Scheme 39: Continuous-flow Michael addition using ion exchange resin Amberlyst® A26.

Scheme 40: Preparation of the heterogeneous catalyst 181 developed by Paixão et al. exploiting Ugi multicompon...

Scheme 41: Continuous-flow system developed by the Paixão’s group for the preparation of Michael asymmetric ad...

Scheme 42: Continuous-flow synthesis of nitroaldols catalysed by supported catalyst 184 developed by Wennemers...

Scheme 43: Heterogenous polystyrene-supported catalysts developed by Pericàs and co-workers.

Scheme 44: PANF-supported pyrrolidine catalyst for the conjugate addition of cyclohexanone (65) and trans-β-ni...

Scheme 45: Synthesis of (−)-paroxetine precursor 195 developed by Ötvös, Pericàs, and Kappe.

Scheme 46: Continuous-flow approach for the 5-step synthesis of (−)-oseltamivir (201) as devised by Hayashi an...

Scheme 47: Continuous-flow enzyme-catalysed Michael addition.

Scheme 48: Continuous-flow copper-catalysed 1,4 conjugate addition of Grignard reagents to enones. Reprinted w...

Scheme 49: A collection of commonly encountered hydrogenation reactions.

Figure 7: The ThalesNano H-Cube® continuous-flow hydrogenator.

Scheme 50: Chemoselective reduction of an α,β-unsaturated ketone using the H-Cube® reactor.

Scheme 51: Incorporation of Lindlar’s catalyst into the H-Cube® reactor for the reduction of an alkyne.

Scheme 52: Continuous-flow semi-hydrogenation of alkyne 208 to 209 using SACs with H-Cube® system.

Figure 8: The standard setups for tube-in-tube gas–liquid reactor units.

Scheme 53: Homogeneous hydrogenation of olefins using a tube-in-tube reactor setup.

Scheme 54: Recyclable heterogeneous flow hydrogenation system.

Scheme 55: Leadbeater’s reverse tube-in-tube hydrogenation system for olefin reductions.

Scheme 56: a) Hydrogenation using a Pd-immobilised microchannel reactor (MCR) and b) a representation of the i...

Scheme 57: Hydrogenation of alkyne 238 exploiting segmented flow in a Pd-immobilised capillary reactor.

Scheme 58: Continuous hydrogenation system for the preparation of cyrene (241) from (−)-levoglucosenone (240).

Scheme 59: Continuous hydrogenation system based on CSMs developed by Hornung et al.

Scheme 60: Chemoselective reduction of carbonyls (ketones over aldehydes) in flow.

Scheme 61: Continuous system for the semi-hydrogenation of 256 and 258, developed by Galarneau et al.

Scheme 62: Continuous synthesis of biodiesel fuel 261 from lignin-derived furfural acetone (260).

Scheme 63: Continuous synthesis of γ-valerolacetone (263) via CTH developed by Pineda et al.

Scheme 64: Continuous hydrogenation of lignin-derived biomass (products 265, 266, and 267) using a sustainable...

Scheme 65: Ru/C or Rh/C-catalysed hydrogenation of arene in flow as developed by Sajiki et al.

Scheme 66: Polysilane-immobilized Rh–Pt-catalysed hydrogenation of arenes in flow by Kobayashi et al.

Scheme 67: High-pressure in-line mixing of H2 for the asymmetric reduction of 278 at pilot scale with a 73 L p...

Figure 9: Picture of the PFR employed at Eli Lilly & Co. for the continuous hydrogenation of 278 [287]. Reprinted ...

Scheme 68: Continuous-flow asymmetric hydrogenation using Oppolzer's sultam 280 as chiral auxiliary.

Scheme 69: Some examples of industrially important oxidation reactions in the F&F industry. CFL: compact fluor...

Scheme 70: Gold-catalysed heterogeneous oxidation of alcohols in flow.

Scheme 71: Uozumi’s ARP-Pt flow oxidation protocol.

Scheme 72: High-throughput screening of aldehyde oxidation in flow using an in-line GC.

Scheme 73: Permanganate-mediated Nef oxidation of nitroalkanes in flow with the use of in-line sonication to p...

Scheme 74: Continuous-flow aerobic anti-Markovnikov Wacker oxidation.

Scheme 75: Continuous-flow oxidation of 2-benzylpyridine (312) using air as the oxidant.

Scheme 76: Continuous-flow photo-oxygenation of monoterpenes.

Scheme 77: A tubular reactor design for flow photo-oxygenation.

Scheme 78: Glucose oxidase (GOx)-mediated continuous oxidation of glucose using compressed air and the FFMR re...

Scheme 79: Schematic continuous-flow sodium hypochlorite/TEMPO oxidation of alcohols.

Scheme 80: Oxidation using immobilised TEMPO (344) was developed by McQuade et al.

Scheme 81: General protocol for the bleach/catalytic TBAB oxidation of aldehydes and alcohols.

Scheme 82: Continuous-flow PTC-assisted oxidation using hydrogen peroxide. The process was easily scaled up by...

Scheme 83: Continuous-flow epoxidation of cyclohexene (348) and in situ preparation of m-CPBA.

Scheme 84: Continuous-flow epoxidation using DMDO as oxidant.

Scheme 85: Mukayama aerobic epoxidation optimised in flow mode by the Favre-Réguillon group.

Scheme 86: Continuous-flow asymmetric epoxidation of derivatives of 359 exploiting a biomimetic iron catalyst.

Scheme 87: Continuous-flow enzymatic epoxidation of alkenes developed by Watts et al.

Scheme 88: Engineered multichannel microreactor for continuous-flow ozonolysis of 366.

Scheme 89: Continuous-flow synthesis of the vitamin D precursor 368 using multichannel microreactors. MFC: mas...

Scheme 90: Continuous ozonolysis setup used by Kappe et al. for the synthesis of various substrates employing ...

Scheme 91: Continuous-flow apparatus for ozonolysis as developed by Ley et al.

Scheme 92: Continuous-flow ozonolysis for synthesis of vanillin (2) using a film-shear flow reactor.

Scheme 93: Examples of preparative methods for ajoene (386) and allicin (388).

Scheme 94: Continuous-flow oxidation of thioanisole (389) using styrene-based polymer-supported peroxytungstat...

Scheme 95: Continuous oxidation of thiosulfinates using Oxone®-packed reactor.

Scheme 96: Continuous-flow electrochemical oxidation of thioethers.

Scheme 97: Continuous-flow oxidation of 400 to cinnamophenone (235).

Scheme 98: Continuous-flow synthesis of dehydrated material 401 via oxidation of methyl dihydrojasmonate (33).

Scheme 99: Some industrially important transformations involving Grignard reagents.

Scheme 100: Grachev et al. apparatus for continuous preparation of Grignard reagents.

Scheme 101: Example of fluidized Mg bed reactor with NMR spectrometer as on-line monitoring system.

Scheme 102: Continuous-flow synthesis of Grignard reagents and subsequent quenching reaction.

Figure 10: Membrane-based, liquid–liquid separator with integrated pressure control [52]. Adapted with permission ...

Scheme 103: Continuous-flow synthesis of 458, an intermediate to fluconazole (459).

Scheme 104: Continuous-flow synthesis of ketones starting from benzoyl chlorides.

Scheme 105: A Grignard alkylation combining CSTR and PFR technologies with in-line infrared reaction monitoring....

Scheme 106: Continuous-flow preparation of 469 from Grignard addition of methylmagnesium bromide.

Scheme 107: Continuous-flow synthesis of Grignard reagents 471.

Scheme 108: Preparation of the Grignard reagent 471 using CSTR and the continuous process for synthesis of the ...

Scheme 109: Continuous process for carboxylation of Grignard reagents in flow using tube-in-tube technology.

Scheme 110: Continuous synthesis of propargylic alcohols via ethynyl-Grignard reagent.

Scheme 111: Silica-supported catalysed enantioselective arylation of aldehydes using Grignard reagents in flow ...

Scheme 112: Acid-catalysed rearrangement of citral and dehydrolinalool derivatives.

Scheme 113: Continuous stilbene isomerisation with continuous recycling of photoredox catalyst.

Scheme 114: Continuous-flow synthesis of compound 494 as developed by Ley et al.

Scheme 115: Selected industrial applications of DA reaction.

Scheme 116: Multistep flow synthesis of the spirocyclic structure 505 via employing DA cycloaddition.

Scheme 117: Continuous-flow DA reaction developed in a plater flow reactor for the preparation of the adduct 508...

Scheme 118: Continuous-flow DA reaction using a silica-supported imidazolidinone organocatalyst.

Scheme 119: Batch vs flow for the DA reaction of (cyclohexa-1,5-dien-1-yloxy)trimethylsilane (513) with acrylon...

Scheme 120: Continuous-flow DA reaction between 510 and 515 using a shell-core droplet system.

Scheme 121: Continuous-flow synthesis of bicyclic systems from benzyne precursors.

Scheme 122: Continuous-flow synthesis of bicyclic scaffolds 527 and 528 for further development of potential ph...

Scheme 123: Continuous-flow inverse-electron hetero-DA reaction to pyridine derivatives such as 531.

Scheme 124: Comparison between batch and flow for the synthesis of pyrimidinones 532–536 via retro-DA reaction ...

Scheme 125: Continuous-flow coupled with ultrasonic system for preparation of ʟ-ascorbic acid derivatives 539 d...

Scheme 126: Two-step continuous-flow synthesis of triazole 543.

Scheme 127: Continuous-flow preparation of triazoles via CuAAC employing 546-based heterogeneous catalyst.

Scheme 128: Continuous-flow synthesis of compounds 558 through A3-coupling and 560 via AgAAC both employing the...

Scheme 129: Continuous-flow photoinduced [2 + 2] cycloaddition for the preparation of bicyclic derivatives of 5...

Scheme 130: Continuous-flow [2 + 2] and [5 + 2] cycloaddition on large scale employing a flow reactor developed...

Scheme 131: Continuous-flow preparation of the tricyclic structures 573 and 574 starting from pyrrole 570 via [...

Scheme 132: Continuous-flow [2 + 2] photocyclization of cinnamates.

Scheme 133: Continuous-flow preparation of cyclobutane 580 on a 5-plates photoreactor.

Scheme 134: Continuous-flow [2 + 2] photocycloaddition under white LED lamp using heterogeneous PCN as photocat...

Figure 11: Picture of the parallel tube flow reactor (PTFR) "The Firefly" developed by Booker-Milburn et al. a...

Scheme 135: Continuous-flow acid-catalysed [2 + 2] cycloaddition between silyl enol ethers and acrylic esters.

Scheme 136: Continuous synthesis of lactam 602 using glass column reactors.

Scheme 137: In situ generation of ketenes for the Staudinger lactam synthesis developed by Ley and Hafner.

Scheme 138: Application of [2 + 2 + 2] cycloadditions in flow employed by Ley et al.

Scheme 139: Examples of FC reactions applied in F&F industry.

Scheme 140: Continuous-flow synthesis of ibuprofen developed by McQuade et al.

Scheme 141: The FC acylation step of Jamison’s three-step ibuprofen synthesis.

Scheme 142: Synthesis of naphthalene derivative 629 via FC acylation in microreactors.

Scheme 143: Flow system for rapid screening of catalysts and reaction conditions developed by Weber et al.

Scheme 144: Continuous-flow system developed by Buorne, Muller et al. for DSD optimisation of the FC acylation ...

Scheme 145: Continuous-flow FC acylation of alkynes to yield β-chlorovinyl ketones such as 638.

Scheme 146: Continuous-flow synthesis of tonalide (619) developed by Wang et al.

Scheme 147: Continuous-flow preparation of acylated arene such as 290 employing Zr4+-β-zeolite developed by Kob...

Scheme 148: Flow system applied on an Aza-FC reaction catalysed by the thiourea catalyst 648.

Scheme 149: Continuous hydroformylation in scCO2.

Scheme 150: Two-step flow synthesis of aldehyde 655 through a sequential Heck reaction and subsequent hydroform...

Scheme 151: Single-droplet (above) and continuous (below) flow reactors developed by Abolhasani et al. for the ...

Scheme 152: Continuous hydroformylation of 1-dodecene (655) using a PFR-CSTR system developed by Sundmacher et ...

Scheme 153: Continuous-flow synthesis of the aldehyde 660 developed by Eli Lilly & Co. [32]. Adapted with permissio...

Scheme 154: Continuous asymmetric hydroformylation employing heterogenous catalst supported on carbon-based sup...

Scheme 155: Examples of acetylation in F&F industry: synthesis of bornyl (S,R,S-664) and isobornyl (S,S,S-664) ...

Scheme 156: Continuous-flow preparation of bornyl acetate (S,R,S-664) employing the oscillating flow reactor.

Scheme 157: Continuous-flow synthesis of geranyl acetate (666) from acetylation of geraniol (343) developed by ...

Scheme 158: 12-Ttungstosilicic acid-supported silica monolith-catalysed acetylation in flow.

Scheme 159: Continuous-flow preparation of cyclopentenone 676.

Scheme 160: Two-stage synthesis of coumarin (90) via acetylation of salicylaldehyde (88).

Scheme 161: Intensification process for acetylation of 5-methoxytryptamine (677) to melatonin (678) developed b...

Scheme 162: Examples of macrocyclic musky odorants both natural (679–681) and synthetic (682 and 683).

Scheme 163: Flow setup combined with microwave for the synthesis of macrocycle 686 via RCM.

Scheme 164: Continuous synthesis of 2,5-dihydro-1H-pyrroles via ring-closing metathesis.

Scheme 165: Continuous-flow metathesis of 485 developed by Leadbeater et al.

Figure 12: Comparison between RCM performed using different routes for the preparation of 696. On the left the...

Scheme 166: Continuous-flow RCM of 697 employed the solid-supported catalyst 698 developed by Grela, Kirschning...

Scheme 167: Continuous-flow RORCM of cyclooctene employing the silica-absorbed catalyst 700.

Scheme 168: Continuous-flow self-metathesis of methyl oleate (703) employing SILP catalyst 704.

Scheme 169: Flow apparatus for the RCM of 697 using a nanofiltration membrane for the recovery and reuse of the...

Scheme 170: Comparison of loadings between RCMs performed with different routes for the synthesis of 709.
N-tert-Butanesulfinyl imines in the asymmetric synthesis of nitrogen-containing heterocycles
- Joseane A. Mendes,
- Paulo R. R. Costa,
- Miguel Yus,
- Francisco Foubelo and
- Camilla D. Buarque
Beilstein J. Org. Chem. 2021, 17, 1096–1140, doi:10.3762/bjoc.17.86

- hand, natural amino acids proline and hydroxyproline are functionalized pyrrolidines that have found great application in organic synthesis as chiral organocatalysts in stereoselective processes [93][94]. Cyclizations involving a position in the starting chiral imine Arylation of chiral sulfinyl imines

Graphical Abstract

Scheme 1: General strategy for the enantioselective synthesis of N-containing heterocycles from N-tert-butane...

Scheme 2: Methodologies for condensation of aldehydes and ketones with tert-butanesulfinamides (1).

Scheme 3: Transition models for cis-aziridines and trans-aziridines.

Scheme 4: Mechanism for the reduction of N-tert-butanesulfinyl imines.

Scheme 5: Transition models for the addition of organomagnesium and organolithium compounds to N-tert-butanes...

Scheme 6: Synthesis of 2,2-dibromoaziridines 15 from aldimines 14 and bromoform, and proposed non-chelation-c...

Scheme 7: Diastereoselective synthesis of aziridines from tert-butanesulfinyl imines.

Scheme 8: Synthesis of vinylaziridines 22 from aldimines 14 and 1,3-dibromopropene 23, and proposed chelation...

Scheme 9: Synthesis of vinylaziridines 27 from aldimines 14 and α-bromoesters 26, and proposed transition sta...

Scheme 10: Synthesis of 2-chloroaziridines 28 from aldimines 14 and dichloromethane, and proposed transition s...

Scheme 11: Synthesis of cis-vinylaziridines 30 and 31 from aldimines 14 and bromomethylbutenolide 29.

Scheme 12: Synthesis of 2-chloro-2-aroylaziridines 36 and 32 from aldimines 14, arylnitriles 34, and silyldich...

Scheme 13: Synthesis of trifluoromethylaziridines 39 and proposed transition state of the aziridination.

Scheme 14: Synthesis of aziridines 42 and proposed state transition.

Scheme 15: Synthesis of 1-substituted 2-azaspiro[3.3]heptanes, 1-phenyl-2-azaspiro[3.4]octane and 1-phenyl-2-a...

Scheme 16: Synthesis of 1-substituted 2,6-diazaspiro[3.3]heptanes 48 from chiral imines 14 and 1-Boc-azetidine...

Scheme 17: Synthesis of β-lactams 52 from chiral imines 14 and dimethyl malonate (49).

Scheme 18: Synthesis of spiro-β-lactam 57 from chiral (RS)-N-tert-butanesulfinyl isatin ketimine 53 and ethyl ...

Scheme 19: Synthesis of β-lactam 60, a precursor of (−)-batzelladine D (61) and (−)-13-epi-batzelladine D (62)...

Scheme 20: Rhodium-catalyzed asymmetric synthesis of 3-substituted pyrrolidines 66 from chiral imine (RS)-63 a...

Scheme 21: Asymmetric synthesis of 1,3-disubstituted isoindolines 69 and 70 from chiral imine 67.

Scheme 22: Asymmetric synthesis of cis-2,5-disubstituted pyrrolidines 73 from chiral imine (RS)-71.

Scheme 23: Asymmetric synthesis of 3-hydroxy-5-substituted pyrrolidin-2-ones 77 from chiral imine (RS)-74.

Scheme 24: Asymmetric synthesis of 4-hydroxy-5-substituted pyrrolidin-2-ones 80 from chiral imines 79.

Scheme 25: Asymmetric synthesis of 3-pyrrolines 82 from chiral imines 14 and ethyl 4-bromocrotonate (81).

Scheme 26: Asymmetric synthesis of γ-amino esters 84, and tetramic acid derivative 86 from chiral imines (RS)-...

Scheme 27: Asymmetric synthesis of α-methylene-γ-butyrolactams 90 from chiral imines (Z,SS)-87 and ethyl 2-bro...

Scheme 28: Asymmetric synthesis of methylenepyrrolidines 92 from chiral imines (RS)-14 and 2-(trimethysilylmet...

Scheme 29: Synthesis of dibenzoazaspirodecanes from cyclic N-tert-butanesulfinyl imines.

Scheme 30: Stereoselective synthesis of cyclopenta[c]proline derivatives 103 from β,γ-unsaturated α-amino acid...

Scheme 31: Stereoselective synthesis of alkaloids (−)-angustureine (107) and (−)-cuspareine (108).

Scheme 32: Stereoselective synthesis of alkaloids (−)-pelletierine (112) and (+)-coniine (117).

Scheme 33: Synthesis of piperidine alkaloids (+)-dihydropinidine (122a), (+)-isosolenopsin (122b) and (+)-isos...

Scheme 34: Stereoselective synthesis of the alkaloids(+)-sedamine (125) from chiral imine (SS)-119.

Scheme 35: Stereoselective synthesis of trans-5-hydroxy-6-substituted-2-piperidinones 127 and 129 from chiral ...

Scheme 36: Stereoselective synthesis of trans-5-hydroxy-6-substituted ethanone-2-piperidinones 132 from chiral...

Scheme 37: Stereoselective synthesis of trans-3-benzyl-5-hydroxy-6-substituted-2-piperidinones 136 from chiral...

Scheme 38: Stereoselective synthesis of trans-5-hydroxy-6-substituted 2-piperidinones 139 from chiral imine 138...

Scheme 39: Stereoselective synthesis of ʟ-hydroxypipecolic acid 145 from chiral imine 144.

Scheme 40: Synthesis of 1-substituted isoquinolones 147, 149 and 151.

Scheme 41: Stereoselective synthesis of 3-substituted dihydrobenzo[de]isoquinolinones 154.

Scheme 42: Enantioselective synthesis of alkaloids (S)-1-benzyl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline (...

Scheme 43: Enantioselective synthesis of alkaloids (−)-cermizine B (171) and (+)-serratezomine E (172) develop...

Scheme 44: Stereoselective synthesis of (+)-isosolepnosin (177) and (+)-solepnosin (178) from homoallylamine d...

Scheme 45: Stereoselective synthesis of tetrahydroquinoline derivatives 184, 185 and 187 from chiral imines (RS...

Scheme 46: Stereoselective synthesis of pyridobenzofuran and pyridoindole derivatives 193 from homopropargylam...

Scheme 47: Stereoselective synthesis of 2-substituted 1,2,5,6-tetrahydropyridines 196 from chiral imines (RS)-...

Scheme 48: Stereoselective synthesis of 2-substituted trans-2,6-disubstituted piperidine 199 from chiral imine...

Scheme 49: Stereoselective synthesis of cis-2,6-disubstituted piperidines 200, and alkaloid (+)-241D, from chi...

Scheme 50: Stereoselective synthesis of 6-substituted piperidines-2,5-diones 206 and 1,7-diazaspiro[4.5]decane...

Scheme 51: Stereoselective synthesis of spirocyclic oxindoles 210 from chiral imines (RS)-53.

Scheme 52: Stereoselective synthesis of azaspiro compound 213 from chiral imine 211.

Scheme 53: Stereoselective synthesis of tetrahydroisoquinoline derivatives from chiral imines (RS)-214.

Scheme 54: Stereoselective synthesis of (−)-crispine A 223 from chiral imine (RS)-214.

Scheme 55: Synthesis of (−)-harmicine (228) using tert-butanesulfinamide through haloamide cyclization.

Scheme 56: Stereoselective synthesis of tetraponerines T1–T8.

Scheme 57: Stereoselective synthesis of phenanthroindolizidines 246a and (−)-tylophorine (246b), and phenanthr...

Scheme 58: Stereoselective synthesis of indoline, tetrahydroquinoline and tetrahydrobenzazepine derivatives 253...

Scheme 59: Stereoselective synthesis of (+)-epohelmin A (258) and (+)-epohelmin B (260) from aldimine (RS)-79.

Scheme 60: Stereoselective synthesis of (−)-epiquinamide (266) from chiral aldimine (SS)-261.

Scheme 61: Synthesis synthesis of (–)-hippodamine (273) and (+)-epi-hippodamine (272) using chiral sulfinyl am...

Scheme 62: Stereoselective synthesis of (+)-grandisine D (279) and (+)-amabiline (283).

Scheme 63: Stereoselective synthesis of (−)-epiquinamide (266) and (+)-swaisonine (291) from aldimine (SS)-126....

Scheme 64: Stereoselective synthesis of (+)-C(9a)-epi-epiquinamide (294).

Scheme 65: Stereoselective synthesis of (+)-lasubine II (298) from chiral aldimine (SS)-109.

Scheme 66: Stereoselective synthesis of (−)-epimyrtine (300a) and (−)-lasubine II (ent-302) from β-amino keton...

Scheme 67: Stereoselective synthesis of (−)-tabersonine (310), (−)-vincadifformine (311), and (−)-aspidospermi...

Scheme 68: Stereoselective synthesis of (+)-epohelmin A (258) and (+)-epohelmin B (260) from aldehyde 313 and ...

Scheme 69: Total synthesis of (+)-lysergic acid (323) from N-tert-butanesulfinamide (RS)-1.
Synthetic accesses to biguanide compounds
- Oleksandr Grytsai,
- Cyril Ronco and
- Rachid Benhida
Beilstein J. Org. Chem. 2021, 17, 1001–1040, doi:10.3762/bjoc.17.82

- neat for 30 min, or 24 h diluted in an apolar solvent. Both of these conditions proved very efficient even for hindered reagents and displayed good to excellent yields (Scheme 39B). In the search for new biguanide-based organocatalysts for the transesterification of vegetable oils, Glasovac et al

Graphical Abstract

Figure 1: Tautomeric forms of biguanide.

Figure 2: Illustrations of neutral, monoprotonated, and diprotonated structures biguanide.

Figure 3: The main approaches for the synthesis of biguanides. The core structure is obtained via the additio...

Scheme 1: The three main preparations of biguanides from cyanoguanidine.

Scheme 2: Synthesis of butylbiguanide using CuCl2 [16].

Scheme 3: Synthesis of biguanides by the direct fusion of cyanoguanidine and amine hydrochlorides [17,18].

Scheme 4: Synthesis of ethylbiguanide and phenylbiguanide as reported by Smolka and Friedreich [14].

Scheme 5: Synthesis of arylbiguanides through the reaction of cyanoguanidine with anilines in water [19].

Scheme 6: Synthesis of aryl- and alkylbiguanides by adaptations of Cohn’s procedure [20,21].

Scheme 7: Microwave-assisted synthesis of N1-aryl and -dialkylbiguanides [22,23].

Scheme 8: Synthesis of aryl- and alkylbiguanides by trimethylsilyl activation [24,26].

Scheme 9: Synthesis of phenformin analogs by TMSOTf activation [27].

Scheme 10: Synthesis of N1-(1,2,4-triazolyl)biguanides [28].

Scheme 11: Synthesis of 2-guanidinobenzazoles by addition of ortho-substituted anilines to cyanoguanidine [30,32] and...

Scheme 12: Synthesis of 2,4-diaminoquinazolines by the addition of 2-cyanoaniline to cyanoguanidine and from 3...

Scheme 13: Reactions of anthranilic acid and 2-mercaptobenzoic acid with cyanoguanidine [24,36,37].

Scheme 14: Synthesis of disubstituted biguanides with Cu(II) salts [38].

Scheme 15: Synthesis of an N1,N2,N5-trisubstituted biguanide by fusion of an amine hydrochloride and 2-cyano-1...

Scheme 16: Synthesis of N1,N5-disubstituted biguanides by the addition of anilines to cyanoguanidine derivativ...

Scheme 17: Microwave-assisted additions of piperazine and aniline hydrochloride to substituted cyanoguanidines ...

Scheme 18: Synthesis of N1,N5-alkyl-substituted biguanides by TMSOTf activation [27].

Scheme 19: Additions of oxoamines hydrochlorides to dimethylcyanoguanidine [49].

Scheme 20: Unexpected cyclization of pyridylcyanoguanidines under acidic conditions [50].

Scheme 21: Example of industrial synthesis of chlorhexidine [51].

Scheme 22: Synthesis of symmetrical N1,N5-diarylbiguanides from sodium dicyanamide [52,53].

Scheme 23: Synthesis of symmetrical N1,N5-dialkylbiguanides from sodium dicyanamide [54-56].

Scheme 24: Stepwise synthesis of unsymmetrical N1,N5-trisubstituted biguanides from sodium dicyanamide [57].

Scheme 25: Examples for the synthesis of unsymmetrical biguanides [58].

Scheme 26: Examples for the synthesis of an 1,3-diaminobenzoquinazoline derivative by the SEAr cyclization of ...

Scheme 27: Major isomers formed by the SEAr cyclization of symmetric biguanides derived from 2- and 3-aminophe...

Scheme 28: Lewis acid-catalyzed synthesis of 8H-pyrrolo[3,2-g]quinazoline-2,4-diamine [63].

Scheme 29: Synthesis of [1,2,4]oxadiazoles by the addition of hydroxylamine to dicyanamide [49,64].

Scheme 30: Principle of “bisamidine transfer” and analogy between the reactions with N-amidinopyrazole and N-a...

Scheme 31: Representative syntheses of N-amidino-amidinopyrazole hydrochloride [68,69].

Scheme 32: First examples of biguanide syntheses using N-amidino-amidinopyrazole [66].

Scheme 33: Example of “biguanidylation” of a hydrazide substrate [70].

Scheme 34: Example for the synthesis of biguanides using S-methylguanylisothiouronium iodide as “bisamidine tr...

Scheme 35: Synthesis of N-substituted N1-cyano-S-methylisothiourea precursors.

Scheme 36: Addition routes on N1-cyano-S-methylisothioureas.

Scheme 37: Synthesis of an hydroxybiguanidine from N1-cyano-S-methylisothiourea [77].

Scheme 38: Synthesis of an N1,N2,N3,N4,N5-pentaarylbiguanide from the corresponding triarylguanidine and carbo...

Scheme 39: Reactions of N,N,N’,N’-tetramethylguanidine (TMG) with carbodiimides to synthesize hexasubstituted ...

Scheme 40: Microwave-assisted addition of N,N,N’,N’-tetramethylguanidine to carbodiimides [80].

Scheme 41: Synthesis of N1-aryl heptasubstituted biguanides via a one-pot biguanide formation–copper-catalyzed ...

Scheme 42: Formation of 1,2-dihydro-1,3,5-triazine derivatives by the reaction of guanidine with excess carbod...

Scheme 43: Plausible mechanism for the spontaneous cyclization of triguanides [82].

Scheme 44: a) Formation of mono- and disubstituted (iso)melamine derivatives by the reaction of biguanides and...

Scheme 45: Reactions of 2-aminopyrimidine with carbodiimides to synthesize 2-guanidinopyrimidines as “biguanid...

Scheme 46: Non-catalyzed alternatives for the addition of 2-aminopyrimidine derivatives to carbodiimides. A) h...

Scheme 47: Addition of guanidinomagnesium halides to substituted cyanamides [90].

Scheme 48: Microwave-assisted synthesis of [11C]metformin by the reaction of 11C-labelled dimethylcyanamide an...

Scheme 49: Formation of 4-amino-6-dimethylamino[1,3,5]triazin-2-ol through the reaction of Boc-guanidine and d...

Scheme 50: Formation of 1,3,5-triazine derivatives via the addition of guanidines to substituted cyanamides [92].

Scheme 51: Synthesis of biguanide by the reaction of O-alkylisourea and guanidine [93].

Scheme 52: Aromatic nucleophilic substitution of guanidine on 2-O-ethyl-1,3,5-triazine [95].

Scheme 53: Synthesis of N1,N2-disubstituted biguanides by the reaction of guanidine and thioureas in the prese...

Scheme 54: Cyclization reactions involving condensations of guanidine(-like) structures with thioureas [97,98].

Scheme 55: Condensations of guanidine-like structures with thioureas [99,100].

Scheme 56: Condensations of guanidines with S-methylisothioureas [101,102].

Scheme 57: Addition of 2-amino-1,3-diazaaromatics to S-alkylisothioureas [103,104].

Scheme 58: Addition of guanidines to 2-(methylsulfonyl)pyrimidines [105].

Scheme 59: An example of a cyclodesulfurization reaction to a fused 3,5-diamino-1,2,4-triazole [106].

Scheme 60: Ring-opening reactions of 1,3-diaryl-2,4-bis(arylimino)-1,3-diazetidines [107].

Scheme 61: Formation of 3,5-diamino-1,2,4-triazole derivatives via addition of hydrazines to 1,3-diazetidine-2...

Scheme 62: Formation of a biguanide via the addition of aniline to 1,2,4-thiadiazol-3,5-diamines, ring opening...

Figure 4: Substitution pattern of biguanides accessible by synthetic pathways a–h.
The synthesis of chiral β-naphthyl-β-sulfanyl ketones via enantioselective sulfa-Michael reaction in the presence of a bifunctional cinchona/sulfonamide organocatalyst
- Deniz Tözendemir and
- Cihangir Tanyeli
Beilstein J. Org. Chem. 2021, 17, 494–503, doi:10.3762/bjoc.17.43
- Deniz Tozendemir Cihangir Tanyeli Department of Chemistry, Middle East Technical University, 06800 Ankara, Turkey 10.3762/bjoc.17.43 Abstract Cinchona alkaloid-derived organocatalysts are widely employed in various asymmetric transformations, yielding products with high enantiopurity. In this
- naturally occurring cinchona alkaloids have shown remarkable performance as organocatalysts for stereoselective synthesis in the past decade [1][2][3][4][5][6]. Among them, quinine-derived organocatalysts make a noteworthy appearance in the formation of new stereogenic centres, which can serve as valuable
- building blocks for the construction of more elaborate structures [7][8][9][10][11]. An outstanding class of quinine derived organocatalysts exhibits a bifunctional mode of activation by the incorporation of an acidic unit, such as urea, thiourea, squaramide or sulfonamide moieties, giving rise to the

Graphical Abstract

Scheme 1: Synthesis of organocatalyst 5.

Figure 1: Structures of the screened organocatalysts.

Scheme 2: Proposed transition state for the SMA of 1-thionaphthol to trans-chalcones.

Figure 2: Comparison of the ee values of SMA in the presence of THF and DCM as solvent.
Direct synthesis of anomeric tetrazolyl iminosugars from sugar-derived lactams
- Michał M. Więcław and
- Bartłomiej Furman
Beilstein J. Org. Chem. 2021, 17, 115–123, doi:10.3762/bjoc.17.12
- widely used as organocatalysts. Such moieties are employed in a number of important synthetic transformations, including the aldol reaction [30], Michael addition [31], Mannich reaction [32], and hydrogenation [33]. We plan to test these possibilities in the near future. Stereochemistry and configuration

Graphical Abstract

Scheme 1: Our previous efforts in the field of functionalization of sugar-derived lactams.

Figure 1: Key concepts behind the goal of this work [34].

Scheme 2: Preliminary experiment in search of a procedure for the synthesis of 2-(1H-tetrazol-5-yl)-iminosuga...

Scheme 3: Synthesis of a new class of alkaloid scaffold using the presented methodology.

Scheme 4: Synthesis of a new, chiral 2-(tetrazol-5-yl)-iminosugar based potential organocatalyst.

Scheme 5: Principle behind Woerpel’s model for prediction of the direction of nucleophile addition to oxocarb...

Scheme 6: Difference in conformational stability of glucose- and galactose-derived iminium cations and the maj...

Figure 2: ORTEP structures of compounds 3a and 3e obtained by X-ray analysis. Hydrogen atoms and benzyl group...

Figure 3: Proposed structures of compounds 5a and 2-epi-5a with 1H-1H couplings and NOE effects shown.

Scheme 7: Proposed reaction mechanism for the described Ugi–azide reaction variant.

Scheme 8: Possible pathway for spontaneous imine formation. Values reported are in kcal·mol−1.

Scheme 9: A possible path for tetrazole formation in the described conditions. Values reported are in kcal·mol...
Syntheses of spliceostatins and thailanstatins: a review
- William A. Donaldson
Beilstein J. Org. Chem. 2020, 16, 1991–2006, doi:10.3762/bjoc.16.166
- endpoint. However, Nicolaou’s route is the shortest (6 or 7 steps, 9.8–9.2% yield), while Kitahara’s synthesis is the highest-yielding and does not involve the use of expensive transition metals or organocatalysts. Syntheses to generate the C-14 stereocenter via C–N bond formation Two groups implemented

Graphical Abstract

Figure 1: Structures of spliceostatins/thailanstatins.

Scheme 1: Synthetic routes to protected (2Z,4S)-4-hydroxy-2-butenoic acid fragments.

Scheme 2: Kitahara synthesis of the (all-cis)-2,3,5,6-tetrasubstituted tetrahydropyran.

Scheme 3: Koide synthesis of (all-cis)-2,3,5,6-tetrasubstituted tetrahydropyran.

Scheme 4: Nicolaou synthesis of the (all-cis)-2,3,5,6-tetrasubstituted tetrahydropyran.

Scheme 5: Jacobsen synthesis of the (all-cis)-2,3,5,6-tetrasubstituted tetrahydropyran.

Scheme 6: Unproductive attempt to generate the (all-cis)-tetrahydropyranone 50.

Scheme 7: Ghosh synthesis of the C-7–C-14 (all-cis)-tetrahydropyran segment.

Scheme 8: Ghosh’s alternative route to the (all-cis)-tetrahydropyranone 50.

Scheme 9: Alternative synthesis of the dihydro-3-pyrone 58.

Scheme 10: Kitahara’s 1st-generation synthesis of the C-1–C-6 fragment of FR901464 (1).

Scheme 11: Kitahara 1st-generation synthesis of the C-1–C-6 fragment of FR901464 (1).

Scheme 12: Nimura/Arisawa synthesis of the C-1-phenyl segment.

Scheme 13: Ghosh synthesis of the C-1–C-6 fragment of FR901464 (1) from (R)-glyceraldehyde acetonide.

Scheme 14: Jacobsen synthesis of the C-1–C-7 segment of FR901464 (1).

Scheme 15: Koide synthesis of the C-1–C-7 segment of FR901464 (1).

Scheme 16: Ghosh synthesis of the C-1–C-5 segment 102 of thailanstatin A (7).

Scheme 17: Nicolaou synthesis of the C-1–C-9 segments of spliceostatin D (9) and thailanstatins A (7) and B (5...

Scheme 18: Ghosh synthesis of the C-1–C-6 segment 115 of spliceostatin E (10).

Scheme 19: Fragment coupling via Wittig and modified Julia olefinations by Kitahara.

Scheme 20: Fragment coupling via cross-metathesis by Koide.

Scheme 21: The Ghosh synthesis of spliceostatin A (4), FR901464 (1), spliceostatin E (10), and thailanstatin m...

Scheme 22: Arisawa synthesis of a C-1-phenyl analog of FR901464 (1).

Scheme 23: Jacobsen fragment coupling by a Pd-catalyzed Negishi coupling.

Scheme 24: Nicolaou syntheses of thailanstatin A and B (7 and 5) and spliceostatin D (9) via a Pd-catalyzed Su...

Scheme 25: The Ghosh synthesis of spliceostatin G (11) via Suzuki–Miyaura coupling.
Et3N/DMSO-supported one-pot synthesis of highly fluorescent β-carboline-linked benzothiophenones via sulfur insertion and estimation of the photophysical properties
- Dharmender Singh,
- Vipin Kumar and
- Virender Singh
Beilstein J. Org. Chem. 2020, 16, 1740–1753, doi:10.3762/bjoc.16.146
- abecarnil, tadalafil, cipargamin, yohimbine, etc. which are used in the treatment of various ailments [14][15]. Apart from their pharmaceutical properties, β-carboline derivatives also found various applications in fields such as organocatalysts, as ligands, and fluorescent probes [16][17][18]. Importantly

Graphical Abstract

Figure 1: Representative examples of some commercial drugs and biologically active alkaloids.

Scheme 1: Synthesis of β-carboline-linked 2-nitrochalcones.

Scheme 2: Synthesis of β-carboline-linked benzothiophenone frameworks.

Scheme 3: Comparison of outcome of one-pot vs two-pot approach.

Scheme 4: One-pot synthesis of β-carboline C-1-tethered benzothiophenone derivatives.

Scheme 5: One-pot synthesis of β-carboline C-3-linked benzothiophenone derivatives.

Scheme 6: One-pot synthesis of β-carboline-linked benzothiophene derivative 6C.

Scheme 7: Control experiment in the presence of a radical scavenger.

Figure 2: Proposed reaction mechanism.

Figure 3: Fluorescence spectra of 2aA–nA, 2bB, 2hB, and 6C.

Figure 4: Fluorescence spectra of 4aA–gA, and 4eB.
NHC-catalyzed enantioselective synthesis of β-trifluoromethyl-β-hydroxyamides
- Alyn T. Davies,
- Mark D. Greenhalgh,
- Alexandra M. Z. Slawin and
- Andrew D. Smith
Beilstein J. Org. Chem. 2020, 16, 1572–1578, doi:10.3762/bjoc.16.129
- trifluoromethyl ketones (Figure 1C) [20]. Over the last twenty years, NHCs have been widely exploited as highly efficient organocatalysts that have found use in numerous applications and were the subject of many extensive reviews [21][22][23][24][25][26]. Among the most common reactive intermediates generated

Graphical Abstract

Figure 1: Organocatalytic enantioselective aldol approaches using trifluoroacetophenone derivatives.

Figure 2: NHC-catalyzed approaches to β-lactones using trifluoroacetophenone derivatives.

Scheme 1: Reaction scope with respect to the nucleophile. aIsolated yield of the product in >95:5 dr. bDeterm...

Scheme 2: Reaction scope with respect to the trifluoroacetophenone derivative and α-aroyloxyaldehyde. aIsolat...

Scheme 3: Proposed mechanism.
Oxime radicals: generation, properties and application in organic synthesis
- Igor B. Krylov,
- Stanislav A. Paveliev,
- Alexander S. Budnikov and
- Alexander O. Terent’ev
Beilstein J. Org. Chem. 2020, 16, 1234–1276, doi:10.3762/bjoc.16.107
- as scavengers of C-centered radicals [9] and selective oxidation organocatalysts (for example, in the oxidation of alcohols to the corresponding carbonyl compounds [10][11]). Recently, highly reactive imidoxyl radicals (Figure 1, II) have found a wide application in the processes of hydrogen atom

Graphical Abstract

Figure 1: Imine-N-oxyl radicals (IV) discussed in the present review and other classes of N-oxyl radicals (I–...

Figure 2: The products of decomposition of iminoxyl radicals generated from oximes by oxidation with Ag2O.

Scheme 1: Generation of oxime radicals and study of the kinetics of their decay by photolysis of the solution...

Scheme 2: Synthesis of di-tert-butyliminoxyl radical and its decomposition products.

Scheme 3: The proposed reaction pathway of the decomposition of di-tert-butyliminoxyl radical (experimentally...

Scheme 4: Monomolecular decomposition of the tert-butyl(triethylmethyl)oxime radical.

Scheme 5: The synthesis and stability of the most stable dialkyl oxime radicals – di-tert-butyliminoxyl and d...

Scheme 6: The formation of iminoxyl radicals from β-diketones under the action of NO2.

Scheme 7: Synthesis of the diacetyliminoxyl radical.

Scheme 8: Examples of long-living oxime radicals with electron-withdrawing groups and the conditions for thei...

Figure 3: The electronic structure iminoxyl radicals and their geometry compared to the corresponding oximes.

Figure 4: Bond dissociation enthalpies (kcal/mol) of oximes and N,N-disubstituted hydroxylamines calculated o...

Scheme 9: Examples demonstrating the low reactivity of the di-tert-butyliminoxyl radical towards the substrat...

Scheme 10: The reactions of di-tert-butyliminoxyl radical with unsaturated hydrocarbons involving hydrogen ato...

Scheme 11: Possible mechanisms of reaction of di-tert-butyliminoxyl radical with alkenes.

Scheme 12: Products of the reaction between di-tert-butyliminoxyl radical and phenol derivatives.

Scheme 13: The reaction of di-tert-butyliminoxyl radical with amines.

Scheme 14: Reaction of di-tert-butyliminoxyl radicals with organolithium reagents.

Scheme 15: Cross-dehydrogenative C–O coupling of 1,3-dicarbonyl compounds with oximes under the action of mang...

Scheme 16: Cross-dehydrogenative C–O coupling of 1,3-dicarbonyl compounds with oximes under the action of Cu(BF...

Scheme 17: Oxidative C–O coupling of benzylmalononitrile (47) with 3-(hydroxyimino)pentane-2,4-dione (19).

Scheme 18: The proposed mechanism of the oxidative coupling of benzylmalononitrile (47) with diacetyl oxime (19...

Scheme 19: Oxidative C–O coupling of pyrazolones with oximes under the action of Fe(ClO4)3.

Scheme 20: The reaction of diacetyliminoxyl radical with pyrazolones.

Scheme 21: Oxidative C–O coupling of oximes with acetonitrile, ketones, and esters.

Scheme 22: Intramolecular cyclizations of oxime radicals to form substituted isoxazolines or cyclic nitrones.

Scheme 23: TEMPO-mediated oxidative cyclization of oximes with C–H bond cleavage.

Scheme 24: Proposed reaction mechanism of oxidative cyclization of oximes with C–H bond cleavage.

Scheme 25: Selectfluor/Bu4NI-mediated C–H oxidative cyclization of oximes.

Scheme 26: Oxidative cyclization of N-benzyl amidoximes to 1,2,4-oxadiazoles.

Scheme 27: The formation of quinazolinone 73a from 5-phenyl-4,5-dihydro-1,2,4-oxadiazole 74 under air.

Scheme 28: DDQ-mediated oxidative cyclization of thiohydroximic acids.

Scheme 29: Plausible mechanism of the oxidative cyclization of thiohydroximic acids.

Scheme 30: Silver-mediated oxidative cyclization of α-halogenated ketoximes and 1,3-dicarbonyl compounds.

Scheme 31: Possible pathway of one-pot oxidative cyclization of α-halogenated ketoximes and 1,3-dicarbonyl com...

Scheme 32: T(p-F)PPT-catalyzed oxidative cyclization of oximes with the formation of 1,2,4-oxadiazolines.

Scheme 33: Intramolecular cyclization of iminoxyl radicals involving multiple C=C and N=N bonds.

Scheme 34: Oxidative cyclization of β,γ- and γ,δ-unsaturated oximes employing the DEAD or TEMPO/DEAD system wi...

Scheme 35: Cobalt-catalyzed aerobic oxidative cyclization of β,γ-unsaturated oximes.

Scheme 36: Manganese-catalyzed aerobic oxidative cyclization of β,γ-unsaturated oximes.

Scheme 37: Visible light photocatalytic oxidative cyclization of β,γ-unsaturated oximes.

Scheme 38: TBAI/TBHP-mediated radical cascade cyclization of the β,γ-unsaturated oximes.

Scheme 39: TBAI/TBHP-mediated radical cascade cyclization of vinyl isocyanides with β,γ-unsaturated oximes.

Scheme 40: tert-Butylnitrite-mediated oxidative cyclization of unsaturated oximes with the introduction of an ...

Scheme 41: Transformation of unsaturated oxime to oxyiminomethylisoxazoline via the confirmed dimeric nitroso ...

Scheme 42: tert-Butylnitrite-mediated oxidative cyclization of unsaturated oximes with the introduction of a n...

Scheme 43: Synthesis of cyano-substituted oxazolines from unsaturated oximes using the TBN/[RuCl2(p-cymene)]2 ...

Scheme 44: Synthesis of trifluoromethylthiolated isoxazolines from unsaturated oximes.

Scheme 45: Copper-сatalyzed oxidative cyclization of β,γ-unsaturated oximes with the introduction of an azido ...

Scheme 46: TBHP-mediated oxidative cascade cyclization of β,γ-unsaturated oximes and unsaturated N-arylamides.

Scheme 47: Copper-сatalyzed oxidative cyclization of unsaturated oximes with the introduction of an amino grou...

Scheme 48: TEMPO-mediated oxidative cyclization of unsaturated oximes followed by elimination.

Scheme 49: Oxidative cyclization of β,γ-unsaturated oximes with the introduction of a trifluoromethyl group.

Scheme 50: Oxidative cyclization of unsaturated oximes with the introduction of a nitrile group.

Scheme 51: Oxidative cyclization of β,γ-unsaturated oximes to isoxazolines with the introduction of a nitrile ...

Scheme 52: Oxidative cyclization of β,γ-unsaturated oximes to isoxazolines with the introduction of a sulfonyl...

Scheme 53: Oxidative cyclization of β,γ- and γ,δ-unsaturated oximes to isoxazolines with the introduction of a...

Scheme 54: Oxidative cyclization of β,γ-unsaturated oximes to isoxazolines with the introduction of a thiocyan...

Scheme 55: PhI(OAc)2-mediated oxidative cyclization of oximes with C–S and C–Se bond formation.

Scheme 56: PhI(OAc)2-mediated oxidative cyclization of unsaturated oximes accompanied by alkoxylation.

Scheme 57: PhI(OAc)2-mediated cyclization of unsaturated oximes to methylisoxazolines.

Scheme 58: Oxidative cyclization-alkynylation of unsaturated oximes.

Scheme 59: TEMPO-mediated oxidative cyclization of C-glycoside ketoximes to C-glycosylmethylisoxazoles.

Scheme 60: Silver-сatalyzed oxidative cyclization of β,γ-unsaturated oximes with formation of fluoroalkyl isox...

Scheme 61: Oxidative cyclization of β,γ-unsaturated oximes with the formation of haloalkyl isoxazolines.

Scheme 62: Cyclization of β,γ-unsaturated oximes into haloalkyl isoxazolines under the action of the halogenat...

Scheme 63: Synthesis of haloalkyl isoxazoles and cyclic nitrones via oxidative cyclization and 1,2-halogen shi...

Scheme 64: Electrochemical oxidative cyclization of diaryl oximes.

Scheme 65: Copper-сatalyzed cyclization and dioxygenation oximes containing a triple C≡C bond.

Scheme 66: Photoredox-catalyzed sulfonylation of β,γ-unsaturated oximes by sulfonyl hydrazides.

Scheme 67: Oxidative cyclization of β,γ-unsaturated oximes with introduction of sulfonate group.

Scheme 68: Ultrasound-promoted oxidative cyclization of β,γ-unsaturated oximes.
Aldehydes as powerful initiators for photochemical transformations
- Maria A. Theodoropoulou,
- Nikolaos F. Nikitas and
- Christoforos G. Kokotos
Beilstein J. Org. Chem. 2020, 16, 833–857, doi:10.3762/bjoc.16.76
- aliphatic acids and the coupling of the residual chain with various electrophiles. Metal-based catalysts are common in reactions that require a high redox potential for a single electron transfer (SET) procedure to take place. On the other hand, even if organocatalysts have lower redox potentials, they are

Graphical Abstract

Scheme 1: Norrish type I and II dissociations.

Scheme 2: Proposed radical pair formation after the photolysis of benzaldehyde (8).

Scheme 3: Aldehydes in the Paterno–Büchi reaction.

Scheme 4: 2,3-Diazabicyclo[2.2.1]hept-2-ene (DBH).

Scheme 5: Dissociation pathways of benzaldehyde.

Scheme 6: Reactions that lead to polarized products detectable by CIDNP.

Scheme 7: MMA (26), DEABP (27), and Michler’s ketone (28).

Scheme 8: Radical intermediates of DEABP.

Scheme 9: Photoinitiated polymerization of monomeric MMA (26) using the quinoxalines 32 and benzaldehyde (8).

Scheme 10: Acetone (4) and formaldehyde (35) as photografting initiators.

Scheme 11: Photografting by employing acetaldehyde (36) as the photoinitiator.

Scheme 12: Proposed photolysis mechanism for aliphatic ketones 44 and formaldehyde (35).

Scheme 13: Initiator 50, reductant 51, and benzaldehyde derivatives 52–54 for the polymerization of the methac...

Scheme 14: Proposed mechanism of the photomediated atom transfer radical polymerization employing the benzalde...

Scheme 15: cis/trans isomerization employing triplet states of photosensitizers.

Scheme 16: Salicylaldehyde (68) forms an internal hydrogen bond.

Scheme 17: Olefin isomerization via energy transfer from a carbonyl compound.

Scheme 18: Mechanistic pathways for the Paterno–Büchi reaction.

Scheme 19: Isomeric oxetanes formed after photochemical addition of aryl aldehydes to 2-butenes.

Scheme 20: Rotation of the C3–C4 bond of the biradical intermediate may lead to all four conformations.

Scheme 21: Photolysis products of benzaldehyde (8) in different solvents. a) In benzene or ethanol. b) In hex-...

Scheme 22: N-tert-Butylbenzamide formation proceeds via a benzoyl radical.

Scheme 23: Photochemical pinacol coupling.

Scheme 24: Photochemical ATRA catalyzed by 4-anisaldehyde (52).

Scheme 25: Proposed triplet sensitization mechanism of the ATRA reaction in the presence of 4-anisaldehyde (52...

Scheme 26: Benzaldehyde-mediated photoredox CDC reaction: compatible amides and ethers.

Scheme 27: Photoredox cross-dehydrogenative coupling (CDC) conditions and proposed reaction mechanism.

Scheme 28: Optimized conditions for the photoredox merger reaction.

Scheme 29: Proposed mechanism for the C(sp3)–H alkylation/arylation of ethers.

Scheme 30: Substrate scope for the photochemical alkylation of ethers.

Scheme 31: C(sp3)–H Functionalization of N-containing molecules.

Scheme 32: Substrate scope for the photochemical alkylation of N-containing molecules.

Scheme 33: Additional products yielded by the photochemical alkylation reaction of N-containing molecules.

Scheme 34: C(sp3)–H functionalization of thioethers.

Scheme 35: Proposed mechanism for the C(sp3)–H alkylation/arylation of N-containing molecules and thioethers.

Scheme 36: Hydroacylation using 4-cyanobenzaldehyde (53) as the photoinitiator.

Scheme 37: Selectivity for the formation of the α,α-disubstituted aldehydes.

Scheme 38: Substrate scope for the photochemical addition of aldehydes to Michael acceptors.

Scheme 39: Proposed mechanism for the hydroacylation of Michael acceptors using 4-cyanobenzaldehyde (53) as th...

Scheme 40: Catalytic arylation of aromatic aldehydes by aryl bromides in which the reaction product acts as th...

Scheme 41: Proposed mechanism for the catalytic arylation of benzaldehydes by aryl bromides in which the react...

Scheme 42: Functionalization of the chiral cyclobutanes 180.

Scheme 43: Optimized reaction conditions and proposed mechanism for the sulfonylcyanation of cyclobutenes.
Preparation of 2-phospholene oxides by the isomerization of 3-phospholene oxides
- Péter Bagi,
- Réka Herbay,
- Nikolett Péczka,
- Zoltán Mucsi,
- István Timári and
- György Keglevich
Beilstein J. Org. Chem. 2020, 16, 818–832, doi:10.3762/bjoc.16.75
- ]. Among the P-heterocycles with various ring size, the five-membered derivatives received special attention due to their optimal bond angles and shapes [13][14]. Besides their application as ligands [13][15], the five-membered heterocycles may also be valuable organocatalysts in reactions involving a P
- (III)–P(V) redox cycle, such as in catalytic Wittig-, aza-Wittig-, Staudinger-, Appel- and other reactions [16][17][18][19][20][21]. The ring strain of the four- and five-membered derivatives makes them highly susceptible to deoxygenation, thus they are ideal organocatalysts in P(III)–P(V) redox

Graphical Abstract

Figure 1: Examples for catalytically or biologically active molecules containing five-membered P-heterocyclic...

Scheme 1: Comparison of the isomerization of 1-phenyl-3-phospholene oxide (5), 1-phenyl-3-methyl-3-phospholen...

Scheme 2: Three possible reaction mechanisms considered in the theoretical studies for the isomerization of 3...

Figure 2: The full time experimental kinetic curves (a); The initial part of the kinetic curves of 1c–f and 1h...

Scheme 3: Computed reaction mechanism of the 3-phospholene oxide (1) 2-phospholene oxide (4) isomerization un...

Scheme 4: Computed reaction mechanism of the 3-phospholene oxide (1) 2-phospholene oxide (4) isomerization un...
Aerobic synthesis of N-sulfonylamidines mediated by N-heterocyclic carbene copper(I) catalysts
- Faïma Lazreg,
- Marie Vasseur,
- Alexandra M. Z. Slawin and
- Catherine S. J. Cazin
Beilstein J. Org. Chem. 2020, 16, 482–491, doi:10.3762/bjoc.16.43
- , the presence of an N-atom in the amidine structure leads to opportunities as ligands and organocatalysts [6][7][8]. N-Sulfonylamidines and N-sulfonylimidates are members of a specific class of these amidines. One initial methodology developed for the formation of sulfonylamidines was based on the

Graphical Abstract

Scheme 1: Formation of sulfonyltriazoles and sulfonamidines.

Figure 1: Catalytic systems used in this study.

Scheme 2: Synthetic access to complexes 4–6 [30].

Scheme 3: Variation of sulfonylazides. Reaction conditions: phenylacetylene (0.5 mmol), sulfonyl azide (0.6 m...

Scheme 4: Variation of alkynes. Reaction conditions: alkyne (0.5 mmol), tosyl azide (0.6 mmol), diisopropylam...

Scheme 5: Variation of the amine substrate. Reaction conditions: phenylacetylene (0.5 mmol), tosyl azide (0.6...

Scheme 6: Reactivity of “non-sulfonyl” azide [33]. Reaction conditions: phenylacetylene (0.5 mmol), benzyl azide ...

Scheme 7: Reactivity of diphenylphosphoryl azide. Reaction conditions: phenylacetylene (0.5 mmol), diphenylph...

Scheme 8: Proposed mechanism for the formation of sulfonamidine.

Scheme 9: Stoichiometric reaction between 6 and 8.

Scheme 10: Synthesis of copper-acetylide intermediate A via [Cu(Cl)(Triaz)].

Scheme 11: Catalytic reaction involving copper-acetylide complex A.
Copper-catalyzed enantioselective conjugate addition of organometallic reagents to challenging Michael acceptors
- Delphine Pichon,
- Jennifer Morvan,
- Christophe Crévisy and
- Marc Mauduit
Beilstein J. Org. Chem. 2020, 16, 212–232, doi:10.3762/bjoc.16.24
- -substitution of enals [21]. By using (R)-BINAP (L2)/CuTC in combination with chiral prolinol derivatives L4–6 as organocatalysts, various α,β-functionalized aldehydes were synthesized in good isolated yields (57–74%) and remarkable enantioselectivity (99%) from diethylzinc or dimethylzinc as nucleophiles and

Graphical Abstract

Scheme 1: Competitive side reactions in the Cu ECA of organometallic reagents to α,β-unsaturated aldehydes.

Scheme 2: Cu-catalyzed ECA of α,β-unsaturated aldehydes with phosphoramidite- (a) and phosphine-based ligands...

Scheme 3: One-pot Cu-catalyzed ECA/organocatalyzed α-substitution of enals.

Scheme 4: Combination of copper and amino catalysis for enantioselective β-functionalizations of enals.

Scheme 5: Optimized conditions for the Cu ECAs of R2Zn, RMgBr, and AlMe3 with α,β-unsaturated aldehydes.

Scheme 6: CuECA of Grignard reagents to α,β-unsaturated thioesters and their application in the asymmetric to...

Scheme 7: Improved Cu ECA of Grignard reagents to α,β-unsaturated thioesters, and their application in the as...

Scheme 8: Catalytic enantioselective synthesis of vicinal dialkyl arrays via Cu ECA of Grignard reagents to γ...

Scheme 9: 1,6-Cu ECA of MeMgBr to α,β,γ,δ-bisunsaturated thioesters: an iterative approach to deoxypropionate...

Scheme 10: Tandem Cu ECA/intramolecular enolate trapping involving 4-chloro-α,β-unsaturated thioester 22.

Scheme 11: Cu ECA of Grignard reagents to 3-boronyl α,β-unsaturated thioesters.

Scheme 12: Cu ECA of alkylzirconium reagents to α,β-unsaturated thioesters.

Scheme 13: Conversion of acylimidazoles into aldehydes, ketones, acids, esters, amides, and amines.

Scheme 14: Cu ECA of dimethyl malonate to α,β-unsaturated acylimidazole 31 with triazacyclophane-based ligand ...

Scheme 15: Cu/L13-catalyzed ECA of alkylboranes to α,β-unsaturated acylimidazoles.

Scheme 16: Cu/hydroxyalkyl-NHC-catalyzed ECA of dimethylzinc to α,β-unsaturated acylimidazoles.

Scheme 17: Stereocontrolled synthesis of 3,5,7-all-syn and anti,anti-stereotriads via iterative Cu ECAs.

Scheme 18: Stereocontrolled synthesis of anti,syn- and anti,anti-3,5,7-(Me,OR,Me) units via iterative Cu ECA/B...

Scheme 19: Cu-catalyzed ECA of dialkylzinc reagents to α,β-unsaturated N-acyloxazolidinones.

Scheme 20: Cu/phosphoramidite L16-catalyzed ECA of dialkylzincs to α,β-unsaturated N-acyl-2-pyrrolidinones.

Scheme 21: Cu/(R,S)-Josiphos (L9)-catalyzed ECA of Grignard reagents to α,β-unsaturated amides.

Scheme 22: Cu/Josiphos (L9)-catalyzed ECA of Grignard reagents to polyunsaturated amides.

Scheme 23: Cu-catalyzed ECA of trimethylaluminium to N-acylpyrrole derivatives.
The use of isoxazoline and isoxazole scaffolding in the design of novel thiourea and amide liquid-crystalline compounds
- Itamar L. Gonçalves,
- Rafaela R. da Rosa,
- Vera L. Eifler-Lima and
- Aloir A. Merlo
Beilstein J. Org. Chem. 2020, 16, 175–184, doi:10.3762/bjoc.16.20
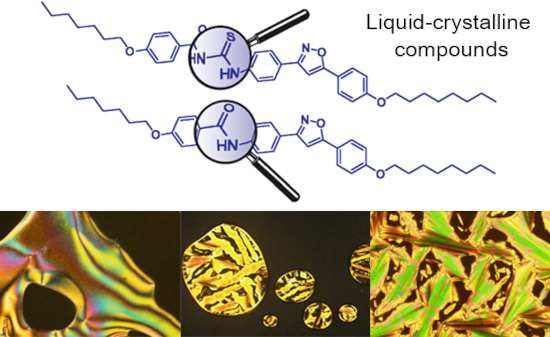
- components, which may be substituted symmetrically or unsymmetrically [1]. Applications of this scaffold include complexing agents in anion sensors [2] organocatalysts [3], intermediates in heterocycle synthesis [1] and development of compounds with pharmacological effects [4]. Considering the importance of

Graphical Abstract

Scheme 1: Amines 3, 4, 8, 9, 12 and 13 installed on 5-membered isoxazoline and isoxazole rings.

Scheme 2: Synthesis of acylisoxazolinylthioureas 17a–c and acylisoxazolylthioureas 18a–c. (i) SOCl2, reflux, ...

Scheme 3: Synthesis of amides. Part A: (i) SOCl2, reflux; (ii) KOCN, acetone, reflux; (iii) amines 3, 4, 8 an...

Figure 1: Optical textures observed on POM for thioureas 17a (a), 17b (b), 18a (c), 18b (d) and 18c (e,f). Al...

Figure 2: Optical textures observed on POM of amide 19. (a) Fan-shaped focal conic texture of the SmA mesopha...

Figure 3: DSC curves for the thiourea 17a (A), amide 19 (B) and 20 (C) upon the first heating and cooling cur...

Figure 4: TGA analysis for thiourea 18c; and amides 20 and 22.
The reaction of arylmethyl isocyanides and arylmethylamines with xanthate esters: a facile and unexpected synthesis of carbamothioates
- Narasimhamurthy Rajeev,
- Toreshettahally R. Swaroop,
- Ahmad I. Alrawashdeh,
- Shofiur Rahman,
- Abdullah Alodhayb,
- Seegehalli M. Anil,
- Kuppalli R. Kiran,
- Chandra,
- Paris E. Georghiou,
- Kanchugarakoppal S. Rangappa and
- Maralinganadoddi P. Sadashiva
Beilstein J. Org. Chem. 2020, 16, 159–167, doi:10.3762/bjoc.16.18
- of xanthate esters with amines [18]. Furthermore, many methods have also been reported for the synthesis of cyclic thiocarbamates, and these include reactions of isothiocyanates with aldehydes in the presence of organocatalysts [19][20], reactions of vicinal diols with potassium thiocyanate [21

Graphical Abstract

Scheme 1: Synthesis of carbamothioates from xanthate esters and benzyl isocyanides.

Figure 1: Substrate scope for the synthesis of carbamothioates. Reaction conditions for methods A and B: sodi...

Figure 2: ORTEP diagram of O-benzyl (4-fluorobenzyl)carbamothioate (4c).

Figure 3: Rotamers of thionocarbamates 4 (top) and computer-minimized structures of 4c (bottom).

Scheme 2: Proposed general reaction mechanism for the formation of carbamothioates (e.g., 4a) from xanthate e...

Figure 4: Optimized geometries of the reactants, transition states, intermediates, and products of the propos...

Figure 5: Relative energies of the reactants, transition states (TS1–TS3), and intermediates (Int1–Int3) of t...
Why do thioureas and squaramides slow down the Ireland–Claisen rearrangement?
- Dominika Krištofíková,
- Juraj Filo,
- Mária Mečiarová and
- Radovan Šebesta
Beilstein J. Org. Chem. 2019, 15, 2948–2957, doi:10.3762/bjoc.15.290
- Bratislava, Mlynská dolina, Ilkovičova 6, 84215 Bratislava, Slovakia 10.3762/bjoc.15.290 Abstract A range of chiral hydrogen-bond-donating organocatalysts was tested in the Ireland–Claisen rearrangement of silyl ketene acetals. None of these organocatalysts was able to impart any enantioselectivity on the
- rearrangements. Furthermore, these organocatalysts slowed down the Ireland–Claisen rearrangement in comparison to an uncatalyzed reaction. The catalyst-free reaction proceeded well in green solvents or without any solvent. DFT calculations showed that the activation barriers are higher for reactions involving
- hydrogen-donating organocatalysts and kinetic experiments suggest that the catalysts bind stronger to the starting silyl ketene acetals than to transition structures thus leading to inefficient rearrangement reactions. Keywords: DFT calculations; green solvents; H-bonding catalysts; Ireland–Claisen

Graphical Abstract

Scheme 1: Ireland–Claisen rearrangement of allyl esters 1a–c.

Scheme 2: Ireland–Claisen rearrangement of 1c mediated by tertiary amines.

Figure 1: Organocatalysts used in this study. Conditions: typical procedure: 1. Et3N (4.9 equiv), DCM, −60 °C...

Scheme 3: Solvent-free Ireland–Claisen rearrangement of cinnamyl esters.

Figure 2: ωB97X-D/6-31G* calculated uncatalyzed Ireland–Claisen rearrangement of 1c. Charges on allylic oxyge...

Figure 3: ωB97X-D/6-31G* calculated Schreiner thiourea (12)-catalyzed Ireland–Claisen rearrangement of 1c. Ch...

Figure 4: ωB97X-D/6-31G* calculated Ph-thiourea (top) and squaramide-catalyzed (bottom) Ireland–Claisen rearr...

Figure 5: a) Rate of product formation; b) reaction profile without catalyst determined by 1H NMR.
A review of asymmetric synthetic organic electrochemistry and electrocatalysis: concepts, applications, recent developments and future directions
- Munmun Ghosh,
- Valmik S. Shinde and
- Magnus Rueping
Beilstein J. Org. Chem. 2019, 15, 2710–2746, doi:10.3762/bjoc.15.264

- transformations using various catalysts as chiral sources has been studied in greater detail than the other strategies mentioned in this review. As per our classification, we further categorize this class into metal catalysts, enzyme catalysts and organocatalysts. Metal catalysts: In 1992, Amundsen and co-workers
- chiral enolates. Organocatalysts: From the outset of electroorganic chemistry, chemists have devoted substantial effort towards applying organocatalysts in electroorganic synthesis. Recent advances integrating organocatalysis and electroorganic synthesis were elegantly presented by Boydston and Ogawa in
- their review article [72]. In this section, we will be presenting a concise description of the use of organocatalysts as chiral inductors in electroorganic synthesis. In 2008, Page, Marken and their group reported a method for electricity-driven asymmetric organocatalytic epoxidation. The percarbonate

Graphical Abstract

Figure 1: General classification of asymmetric electroorganic reactions.

Scheme 1: Asymmetric reduction of 4-acetylpyridine using a modified graphite cathode.

Scheme 2: Asymmetric hydrogenation of ketones using Raney nickel powder electrodes modified with optically ac...

Scheme 3: Asymmetric reduction of prochiral activated olefins with a poly-ʟ-valine-coated graphite cathode.

Scheme 4: Asymmetric reduction of prochiral carbonyl compounds, oximes and gem-dibromides on a poly-ʟ-valine-...

Scheme 5: Asymmetric hydrogenation of prochiral ketones with poly[RuIII(L)2Cl2]+-modified carbon felt cathode...

Scheme 6: Asymmetric hydrogenation of α-keto esters using chiral polypyrrole film-coated cathode incorporated...

Scheme 7: Quinidine and cinchonidine alkaloid-induced asymmetric electroreduction of acetophenone.

Scheme 8: Asymmetric electroreduction of 4- and 2-acetylpyridines at a mercury cathode in the presence of a c...

Scheme 9: Enantioselective reduction of 4-methylcoumarin in the presence of catalytic yohimbine.

Scheme 10: Cinchonine-induced asymmetric electrocarboxylation of 4-methylpropiophenone.

Scheme 11: Enantioselective hydrogenation of methyl benzoylformate using an alkaloid entrapped silver cathode.

Scheme 12: Alkaloid-induced enantioselective hydrogenation using a Cu nanoparticle cathode.

Scheme 13: Alkaloid-induced enantioselective hydrogenation of aromatic ketones using a bimetallic Pt@Cu cathod...

Scheme 14: Enantioselective reduction of ketones at mercury cathode using N,N'-dimethylquininium tetrafluorobo...

Scheme 15: Asymmetric synthesis of an amino acid using an electrode modified with amino acid oxidase and elect...

Scheme 16: Asymmetric oxidation of p-tolyl methyl sulfide using chemically modified graphite anode.

Scheme 17: Asymmetric oxidation of unsymmetric sulfides using poly(amino acid)-coated electrodes.

Scheme 18: Enantioselective, electocatalytic oxidative coupling on TEMPO-modified graphite felt electrode in t...

Scheme 19: Asymmetric electrocatalytic oxidation of racemic alcohols on a TEMPO-modified graphite felt electro...

Scheme 20: Asymmetric electrocatalytic lactonization of diols on TEMPO-modified graphite felt electrodes.

Scheme 21: Asymmetric electrochemical pinacolization in a chiral solvent.

Scheme 22: Asymmetric electroreduction using a chiral supporting electrolyte.

Scheme 23: Asymmetric anodic oxidation of enol acetates using chiral supporting electrolytes.

Scheme 24: Kinetic resolution of primary amines using a chiral N-oxyl radical mediator.

Scheme 25: Chiral N-oxyl-radical-mediated kinetic resolution of secondary alcohols via electrochemical oxidati...

Scheme 26: Chiral iodoarene-mediated asymmetric electrochemical lactonization.

Scheme 27: Os-catalyzed electrochemical asymmetric dihydroxylation of olefins using the Sharpless ligand and i...

Scheme 28: Asymmetric electrochemical epoxidation of olefins catalyzed by a chiral Mn-salen complex.

Scheme 29: Asymmetric electrooxidation of 1,2-diols, and amino alcohols using a chiral copper catalyst.

Scheme 30: Mechanism of asymmetric electrooxidation of 1,2-diols, and amino alcohols using a chiral copper cat...

Scheme 31: Enantioselective electrocarboxylation catalyzed by an electrogenerated chiral [CoI(salen)]− complex....

Scheme 32: Asymmetric oxidative cross coupling of 2-acylimidazoles with silyl enol ethers.

Scheme 33: Ni-catalyzed asymmetric electroreductive cleavage of allylic β-keto ester 89.

Scheme 34: Asymmetric alkylation using a combination of electrosynthesis and a chiral Ni catalyst.

Scheme 35: Mechanism of asymmetric alkylation using a combination of electrosynthesis and a chiral Ni catalyst....

Scheme 36: Asymmetric epoxidation by electrogenerated percarbonate and persulfate ions in the presence of chir...

Scheme 37: α-Oxyamination of aldehydes via anodic oxidation catalyzed by chiral secondary amines.

Scheme 38: The α-alkylation of aldehydes via anodic oxidation catalyzed by chiral secondary amines.

Scheme 39: Mechanism of α-alkylation of aldehydes via anodic oxidation catalyzed by chiral secondary amines.

Scheme 40: Electrochemical chiral secondary amine-catalyzed intermolecular α-arylation of aldehydes.

Scheme 41: Mechanism of electrochemical chiral secondary amine-catalyzed intermolecular α-arylation of aldehyd...

Scheme 42: Asymmetric cross-dehydrogenative coupling of tertiary amines with simple ketones via an electrochem...

Scheme 43: Electroenzymatic asymmetric reduction using enoate reductase.

Scheme 44: Assymetric reduction using alcohol dehydrogenase as the electrocatalyst.

Scheme 45: Asymmetric electroreduction catalyzed by thermophilic NAD-dependent alcohol dehydrogenase.

Scheme 46: Asymmetric epoxidation of styrene by electrochemical regeneration of flavin-dependent monooxygenase....

Scheme 47: Asymmetric electroreduction using a chloroperoxidase catalyst.

Scheme 48: Asymmetric electrochemical transformation mediated by hydrophobic vitamin B12.

Scheme 49: Diastereoselective cathodic reduction of phenylglyoxalic acids substituted with amines as chiral au...

Scheme 50: Ni-catalyzed asymmetric electroreductive cross coupling of aryl halides with α-chloropropanoic acid...

Scheme 51: Electrochemical Mannich addition of silyloxyfuran to in situ-generated N-acyliminium ions.

Scheme 52: Stereoselective electroreductive homodimerization of cinnamates attached to a camphor-derived chira...

Scheme 53: Diastereoselective electrochemical carboxylation of chiral α-bromocarboxylic acid derivatives.

Scheme 54: Electrocatalytic stereoselective conjugate addition of chiral β-dicarbonyl compounds to methyl viny...

Scheme 55: Stereoselective electrochemical carboxylation of chiral cinnamic acid derivatives under a CO2 atmos...

Scheme 56: Electrochemical diastereoselective α-alkylation of pyrrolidines attached with phosphorus-derived ch...

Scheme 57: Electrogenerated cyanomethyl anion-induced synthesis of chiral cis-β-lactams from amides bearing ch...

Scheme 58: Diastereoselective anodic oxidation followed by intramolecular cyclization of ω-hydroxyl amides bea...

Scheme 59: Electrochemical deprotonation of Ni(II) glycinate containing (S)-BPB as a chiral auxiliary: diaster...

Scheme 60: Enantioselective electroreductive coupling of diaryl ketones with α,β-unsaturated carbonyl compound...

Scheme 61: Asymmetric total synthesis of ropivacaine and its analogues using a electroorganic reaction as a ke...

Scheme 62: Asymmetric total synthesis of (−)-crispine A and its natural enantiomer via anodic cyanation of tet...

Scheme 63: Asymmetric oxidative electrodimerization of cinnamic acid derivatives as key step for the synthesis...
1,5-Phosphonium betaines from N-triflylpropiolamides, triphenylphosphane, and active methylene compounds
- Vito A. Fiore,
- Chiara Freisler and
- Gerhard Maas
Beilstein J. Org. Chem. 2019, 15, 2603–2611, doi:10.3762/bjoc.15.253
- intermediates that can be transformed intra- or intermolecularly into a wide array of acyclic, carbocyclic and heterocyclic structures. In these processes, the tertiary phosphanes can act as nucleophilic organocatalysts (for reviews, see [1][2][3][4] or be incorporated in the (pre-)final products, typically as

Graphical Abstract

Scheme 1: Stable betaines I–IV.

Scheme 2: Reactions of N-triflyl-propiolamides 1 with N- and P-nucleophiles. Tf = SO2CF3.

Scheme 3: Synthesis of betaines 3 by a three-component reaction. N-triflylpropiolamides 1: R = Ph (a), 4-Cl-C6...

Figure 1: Phosphonium betaines 3 prepared. An E:Z ratio of 100:0 means that only the E-isomer was observed in...

Scheme 4: Unexpected synthesis of E-3o.

Scheme 5: Betaines 3 from propiolic acid chlorides.

Scheme 6: Two mechanistic scenarios for the formation of betaines 3.

Figure 2: Solid-state structure of E-3a (ORTEP plot), two symmetry-independent molecules in the triclinic uni...

Figure 3: Solid-state structure of E-3b·CH2Cl2 (ORTEP plot); CH2Cl2 solvate molecule not shown.

Figure 4: Solid-state structure of E-3e·H2O·CH2Cl2 (ORTEP plot). The CH2Cl2 solvate molecule is disordered. H...

Figure 5: Solid-state structure of Z-3e (ORTEP plot).

Scheme 7: Resonance structures describing the bonding in 1,2-oxaphospholes/1,5-betaines 8.
α-Photooxygenation of chiral aldehydes with singlet oxygen
- Dominika J. Walaszek,
- Magdalena Jawiczuk,
- Jakub Durka,
- Olga Drapała and
- Dorota Gryko
Beilstein J. Org. Chem. 2019, 15, 2076–2084, doi:10.3762/bjoc.15.205
- separated by column chromatography. Photooxygenation of 3,4-diphenylbutanal (1) with chiral organocatalysts The highly enantioselective synthesis of 3,4-diphenylbutanal (1), according to the procedure developed by Melchiorre, requires the use of noncommercially available sterically bulky silyl ethers [16
- was subjected to photooxygenation in the presence of various organocatalysts: amide 15, imidazolidinone 16, diarylprolinol silyl ethers (S)-17 and (S)-18 which proved the most effective in the photooxygenation of achiral 3-phenylpropanal (Table 1) [14]. The prolinamide-catalyzed reaction furnished
- . All solvents and chemicals used in the syntheses were of reagent grade and were used without further purification. Aldehydes 1 [16][30], 2 [17], 3 [18] and organocatalysts 14 [31], 15 [32], 16 [33] were prepared by known procedures. Silyl ethers of diarylprolinols 17 and 18 were obtained from Sigma

Graphical Abstract

Scheme 1: Asymmetric α-photooxygenation of chiral aldehydes.

Scheme 2: α-Photooxygenation of β-substituted aldehydes.

Scheme 3: Synthesis and α-photooxygenation of 3,4-diphenylbutanal (1).

Scheme 4: Stereoselective α-photooxygenation of 3,4-diphenylbutanal (1) with 1O2.

Scheme 5: Schematic representation of the in situ methodology and preferred conformation of diols with Mo2 co...

Figure 1: ECD spectra of diols syn-6 and anti’-6 recorded a) with 19 in DMSO and b) in acetonitrile compared ...

Scheme 6: Asymmetric synthesis of 3,4-diphenylbutane-1,2-diol.
Fluorinated azobenzenes as supramolecular halogen-bonding building blocks
- Esther Nieland,
- Oliver Weingart and
- Bernd M. Schmidt
Beilstein J. Org. Chem. 2019, 15, 2013–2019, doi:10.3762/bjoc.15.197
- reaction and as organocatalysts in Diels–Alder reactions [27]. The group of Metrangolo also reported halogen bonding-promoted catalysis in water by exploiting a halogen bonding amino acid, which combines excellent donor properties with good water solubility [28], in addition to their seminal contributions

Graphical Abstract

Scheme 1: a) Azobenzenes A1–3 employed in this study. b) U-shaped anthracene halogen bond acceptor bearing tw...

Figure 1: Electrostatic potential map at different isodensity values (B3LYP/ def2/TZVP/DGZVP optimized geomet...

Figure 2: Top: Vertical electronic absorption spectra of a) A2 and b) A3, calculated using TD-B3LYP/def2-TZVP...

Figure 3: a) Space-filling model of U1···A2. The kinked alignment of both the lutidine units of U1 and the az...
A novel three-component reaction between isocyanides, alcohols or thiols and elemental sulfur: a mild, catalyst-free approach towards O-thiocarbamates and dithiocarbamates
- András György Németh,
- György Miklós Keserű and
- Péter Ábrányi-Balogh
Beilstein J. Org. Chem. 2019, 15, 1523–1533, doi:10.3762/bjoc.15.155
- discovered, including HIV-1 reverse transcriptase inhibition activity [6][7][8][9][10][11]. Moreover, their utilization as highly regio- and stereoselective organocatalysts in specific types of chemical tranformations [12][13][14][15][16][17] was introduced, as well. More recently, O-thiocarbamates have been

Graphical Abstract

Scheme 1: Synthetic routes to O-thiocarbamates and dithiocarbamates.

Scheme 2: Substrate scope of isocyanides. aReaction conditions: 1 (1 mmol), S8 (2 mmol), 2a (2mmol), NaH (2 m...

Scheme 3: Substrate scope of alcohols. Reaction conditions: 1a (1 mmol), S8 (2 mmol), 2 (2mmol), NaH (2 mmol)...

Scheme 4: Substrate scope of thiols. Reaction conditions: 1a (1 mmol), S8 (1.2 mmol), 4 (2 mmol), NaOH (2 mmo...

Scheme 5: Scaled-up synthesis for 3a.

Scheme 6: Multicomponent domino synthesis of quinazolinone 7.

Scheme 7: Control experiments.

Scheme 8: Proposed mechanism.
A heteroditopic macrocycle as organocatalytic nanoreactor for pyrroloacridinone synthesis in water
- Piyali Sarkar,
- Sayan Sarkar and
- Pradyut Ghosh
Beilstein J. Org. Chem. 2019, 15, 1505–1514, doi:10.3762/bjoc.15.152
- efficient organocatalysts due to their variable interacting sites. In this context, substituted calixarenes, cyclodextrins, etc., substances known for their ability to encapsulate organic guest molecules, have been explored as organocatalysts over the last two decades [19][20][21][22][23][24]. Another class
- environmentally oriented efficient organocatalysts with prominence “on-water conditions” [30][31]. It is known that an important condition for an organic reaction in water is the aggregation of the reactants and the catalyst by hydrophobic forces. Further, the potential of hydrogen-bond donation may be a vital

Graphical Abstract

Figure 1: Bis-amido-tris-amine macrocycle BATA-MC.

Figure 2: (a) Number distribution plot with particle size in DLS, (b) SEM image and (c) TEM image showing the...

Figure 3: Dependence of the yield of compound 4a on the reaction time using BATA-MC.

Figure 4: Yields of product 4a at different catalyst loading.

Scheme 1: BATA-MC-catalyzed synthesis of 4,5-dihydropyrrolo[2,3,4-kl]acridinones.

Scheme 2: BATA-MC-catalyzed synthesis of pyrrolo[2,3,4-kl]acridinone derivatives.

Figure 5: X-ray single crystal structure of 4d (CCDC 1898008).

Scheme 3: Probable mechanism illustrated for the synthesis of 4a using BATA-MC. For the sake of simplicity, w...

Figure 6: Representation of BATA-MC nanoreactor.

Figure 7: The reusability of the nanoreactor for the synthesis of 4a.
Synthesis of non-racemic 4-nitro-2-sulfonylbutan-1-ones via Ni(II)-catalyzed asymmetric Michael reaction of β-ketosulfones
- Alexander N. Reznikov,
- Anastasiya E. Sibiryakova,
- Marat R. Baimuratov,
- Eugene V. Golovin,
- Victor B. Rybakov and
- Yuri N. Klimochkin
Beilstein J. Org. Chem. 2019, 15, 1289–1297, doi:10.3762/bjoc.15.127
- , which allows to obtain chiral cyclic sulfones with high enantioselectivity [10][11][12]. Also non-racemic cyclic sulfones can be obtained by the Diels–Alder reaction, catalyzed by chiral Lewis acids or organocatalysts. Rh- and Cu-catalyzed CH-insertion reactions occurring at moderate or high
- reactions. Thus, ketonitrosulfones were obtained with good enantiomeric excesses via asymmetric addition of α-nitrosulfones to enones in the presence of organocatalysts [41]. Asymmetric addition of β-ketosulfones to nitroalkenes was implemented using various organocatalysts [42]. The reaction of
- ketosulfones with nitroalkenes in the presence of organocatalysts shows high enantioselectivity, however, leads to a mixture of diastereomers. For the catalytic activation of β-ketosulfones by metal complexes chiral Lewis acids may be considered as an alternative way to carry out the asymmetric Michael

Graphical Abstract

Figure 1: Рharmacologically active sulfones.

Figure 2: Structures of the ligands L1–L8.

Figure 3: Evolution of the conversion of 5 and diastereomeric composition of the products of reaction of 5a w...

Figure 4: Time profile of epimerization and retro-Michael reaction of (2R,3S)-8a in chloroform-d solution.

Figure 5: ORTEP diagram of (2R,3S)-8d.

Scheme 1: The proposed mechanism of asymmetric addition of β-ketosulfones to nitroalkenes.

Scheme 2: Transition state models for asymmetric addition of β-ketosulfones to nitroalkenes.






